How does each of the following changes affect the rate of an E1 reaction? d.) Changing the leaving group from Cl- to Br- e.) Changing the solvent from DMSO to CH3OH
Q: Which of the following alkyl halides would be more reactive in an E2 elimination reaction?
A:
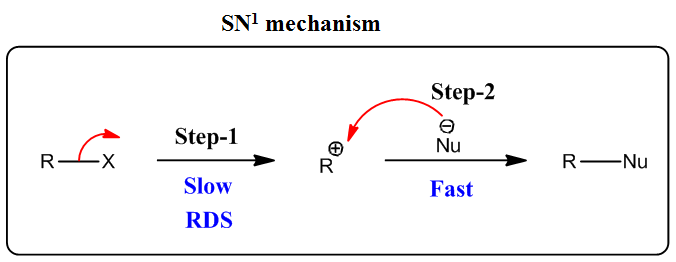
Q: How does the strength of the nucleophile affect an SN1 or SN2 mechanism?
A: Given reaction types, SN1 reaction mechanism SN2 reaction mechanism
Q: what is the difference of the products in an E2 vs E1 reaction?
A:
Q: How does doubling [B:] affect the rate of an E1 reaction?
A: E1 reactions are the unimolecular elimination reaction. This is a two step elimination reaction, in…
Q: Question is attached
A: An elimination reaction is a type of chemical reaction where several atoms either in pairs or groups…
Q: How does changing the leaving group from Cl− to Br− affect the rate of an E1 reaction?
A: In the halogen family, the basicity of the halogen can be given as: F->Cl->Br->I- Fluorine…
Q: Rank the following substrates in order of increasing rate of the E1 reaction. NH2 F OTs D A B
A: E1 reaction : This is a type of elimination reaction is also known as unimolecular elimination…
Q: How does changing the solvent from CH3OH to DMSO affect the rate of an E2 reaction?
A: The reaction in which one molecule is eliminated from reactant compound, such reaction is called…
Q: By what mechanism does the following reaction occur? KI acetone + KBr Br O SN1 E1 O SN2 O E2
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: Which of the following products is most likely to form in the greatest amount if the reaction shown…
A:
Q: Which image correctly depicts the arrow-pushing mechanism of an E2 reaction? Base: :Base Base H2CH…
A:
Q: How does changing the base from −OH to H2O affect the rate of an E2 reaction?
A: The E2 reactions occur in a single step, which has only one transition state. The rate of E2…
Q: Would you expect acetate ion (CH3CO2 -) to be a better nucleophile in an SN2 reaction with an alkyl…
A: The SN2/E2 reaction of alkyl halide is with neutral nucleophile/base, the increasing polarity of the…
Q: In a unimolecular elimination (E1) reaction, the correct order of mechanistic steps is А.…
A: E1 Mechanism Proceeds through loss of a eaving group (LG) and then deprotonation.
Q: How does each of the following changes affect the rate of an E2 reaction? (d, e, and f)
A: E2 reaction is a single step elimination reaction. As this is E2 , so it is bimolecular elimination…
Q: How does changing the leaving group from I− to Br− affect the rate of an E2 reaction?
A: E2 mechanism occurs is a single step concerted reaction with one transition state. The rate at which…
Q: What is NOT step that leads to the product of the Claisen reaction? OCH2CH3 OCH,CH3
A: In Claisen reaction, two esters, each having α-Hydrogen undergo condensation reaction similar to…
Q: 54. Stability of the carbocation intermediate in an SN1 reaction: CH,=CHCH-OH ČH, I. II.CH2=CHCH2OH
A:
Q: Which of the following is the most reactive in an E2 reaction?
A: E2 is bimolecular elimination reaction.
Q: Would you expect acetate ion CH3CO2 to be a better nucleophile in an SN2 reaction with an alkyl…
A: The SN2/E2 reaction of alkyl halide is with neutral nucleophile/base, the increasing polarity of the…
Q: The following reaction takes place several times faster than the reaction of 2-chlorobutane with…
A: The enhances rate of reaction is due to presence of -N..Et2 which undergoes intro molecular SN2…
Q: Why does the leaving group rarely act as a base in SN1 reactions? The ABC step is exothermic and…
A: In a SN1 reaction the leaving group to leave it must be able to accept electron ,if a base remove…
Q: The nucleophile approaches from the opposite side of the carbon with the leaving group in which…
A: It is SN2 reaction. In SN2 reaction i.e nucleophilic substitution biomolecular, one bond is broken…
Q: Why is the mechanism in Figure 28 unlikely? a)The reagent does not have a good leaving group.…
A: In the given reaction the tertiary alkyl bromide reacts with water by biomolecular nucleophilic…
Q: 14. Draw two complete curved arrow mechanisms that accounts for both the products shown in the…
A: Alkyl shift: An alkyl shift occurs when a carbocation does not contain a hydrogen atom that is…
Q: Question attached
A: E1 reaction is unimolecular reaction, where one substituent is removed from the molecule. In a E1…
Q: 5. Which of the following substrates would react fastest by the SN2 mechanism? Br Br Br Br (a) (b)…
A: SN1(Unimolecular nucleophilic substitution reaction): In these types of reactions the rate of…
Q: 4. Describe in table format the following criteria for S,' reactions: (a) strength of nucleophile;…
A: Following are the explanation for SN1 reaction according to given parameters. Dear student, since…
Q: How does doubling [RX] affect the rate of an E1 reaction?
A: The removal of hydrogen halides in a two-step mechanism where the rate of the reaction depends on…
Q: For the dehydrohalogenation (E2) reaction shown, draw the major organic product, including…
A: These are the following important points for E2 Elimination reaction:- * In presence of base,…
Q: From each pair, select the stronger nucleophile. Q.)Cl- or I- in DMSO
A: The species, that make a covalent bond by donates a pair of electrons is known as nucleophile.…
Q: Select which molecule is the better nucleophile in the following pair: a). Br− or Cl− in H2O b).…
A:
Q: What is the major E2 elimination product formed from attached halide?
A: The removal of two or more substituent from the given molecule is defined as elimination reaction.…
Q: Increasing the number of R groups on the carbon with the leaving group forms more highly…
A: Increasing the number of R groups on the carbon with the leaving group forms more highly…
Q: How does changing the solvent from DMSO to CH3OH affect the rate of an E1 reaction?
A: E1 reaction stands for unimolecular elimination reaction. As the name suggests these reactions…
Q: In the E1 reaction, how is the rate affected when the concentration of attacking nucleophile is…
A: Elimination reaction-is type of organic reaction in which two substituents are removed from molecule…
Q: Rank the relative rates of the following alkyl halides in an E1 reaction.
A: Elimination reaction of alkyl halide: Alkyl halide gives an elimination reaction by reacting with a…
Q: Will each of the following reactions follow an El or E2 mechanism? CI HO Он
A:
Q: Draw the major product of this E1 reaction. Ignore any inorganic byproducts. OH
A: Products are present on right hand side and reactants are present on left hand side in a reaction.
Q: If Zaitsev's rule does not apply and the base is bulky then how will the product look?
A: Bulky base like potassium acetate will abstract beta proton from less hindered carbon to form less…
Q: For the following dehydrohalogenation (E2) reaction, draw the Zaitsev product(s), showing the…
A: E2 is a bimolecular elimination reaction. The mechanism of this reaction involves only one step. In…
Q: In the E1 reaction, how is the rate affected when the concentration of attacking nucleophile is…
A: The rate of a reaction helps in the determination of the effect of the change in the concentration…
Q: 1. Assuming no other changes, what is the effect of doubling both the alkyl halide and the…
A: SN1 reactions are Nucleophilic substitution reactions. These are unimolecular reactions. Thus the…
Q: How does each of the following changes affect the rate of an E2 reaction? d. changing the leaving…
A: The explanation is given below-
Q: How does changing the alkyl halide from CH3CH2Br to (CH3)2CHBr affect the rate of an E2 reaction?
A: The nucleophilic substitution reaction of an alkyl halide that involves both the concentration of…
Q: Which of the following pair of "Newman projection --> alkene product" schemes is most accurate for…
A: Answer is explained below.
Q: 3. Which one of these molecules would undergo E2 elimination the most slowly? a) Br b) CI c) CI d)…
A:
Q: Rank the following substrates in order of increasing rate of the SN2 reaction. Br I. A B E
A: Given compounds,
Q: How does changing the halide from (CH3)3CBr to CH3CH2CH2Br affect the rate of an E1 reaction?
A: E1 reaction proceeds with a two step mechanism where the first step is the formation of carbocation…
How does each of the following changes affect the rate of an E1 reaction?
d.) Changing the leaving group from Cl- to Br-
e.) Changing the solvent from DMSO to CH3OH
Step by step
Solved in 2 steps with 1 images

- How does changing the halide from (CH3)3CBr to CH3CH2CH2Br affect the rate of an E1 reaction?How does changing the base from −OH to H2O affect the rate of an E2 reaction?Draw all elimination products that could be formed in an E1 reaction. Ignore the possibility of rearrangements. Ignore any inorganic byproducts.
- Which rearrangement product will form preferentially during the [1,2]-rearrangementof an R-group? Motivate.In the reaction of 1-iodopropane with the hydroxide ion in DMSO how will the rate of reaction change if the concentration of the substrate and nucleophile were each halved? A. The rare will increase by a factor of 4 B. The rate will be reduced by a factor of 1/ 4 C. The rate will be halved. D. The rate will doubleWhen the cleavage of an ether occurs, in step 2, how come the bromide ion does not attack the carbon on the cyclopentane? How come it attacks the CH2 carbon? Does it matter which one is being attacked?



