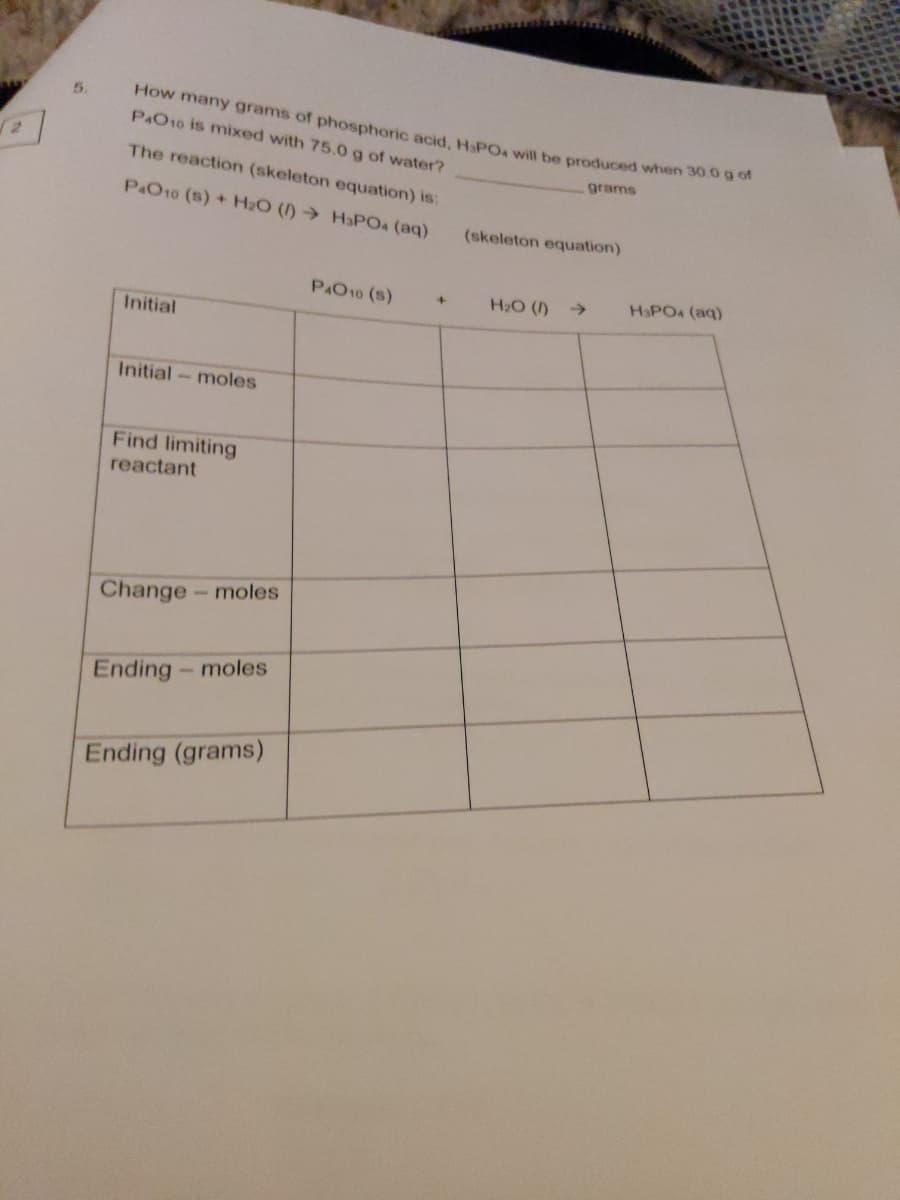
How many grams of phosphoric acid, HPOA will be produced when 30.0 g o P&O10 is mixed with 75.0g of water? 5. The reaction (skeleton equation) is: grams P&O10 (s) + H2O ()→ HaPO4 (aq) (skeleton equation) PAO10 (s) H2O () > HaPO4 (aq) Initial Initial moles Find limiting reactant Change-moles Ending- moles Ending (grams)
Q: gIVEN THE FOLLOWING EQUATION OF THE COMBUSTION OF BUTANE C4H10 + O2 ARROW CO2 + H2O A. WHICH IS…
A: Given C4H10 + 6.5 O2 →…
Q: 11 of 1 Review I Constants Periodic T Butane, C4H10, reacts with oxygen, O2, to form water, H2O, and…
A: Part B) The mass of water produced is to be calculated when 6.96 g of butane reacts with excess…
Q: how many grams CO2 are produced?
A: Given : mass of C2H4 = 75.76 g Mass of O2 = 201.6 g To calculate :- Limiting reactant Mass of…
Q: Balance the reaction shown below: C2H6+O2→CO2+H2OC2H6+O2→CO2+H2O Once balanced, given 0.826…
A: The question is based on the concept of reaction Stoichiometry. It involves calculation of moles of…
Q: Given the following reaction: C3H8+5O2 = 3CO2+4H2O Using dimensional analysis: If 20.0 g of C3H8…
A:
Q: 23 In a universe far, far away, there exists an alien world where they have a different periodic…
A: Instead of the conventional periodic table some alien periodic table is given, wherein name of…
Q: he left side of a balanced chemical equation is shown below K2Cr2O7 + 4 H2SO4 + 3 SeO2 → If 0.600…
A:
Q: After you balance the following equation: n-octene (C8H16) + Potassium permanganate + sulfuric acid…
A: The chemical equation for the given reaction is: C8H16 + KMnO4 + H2SO4 → CO2 + H2O +…
Q: In the reaction between CO2 + H2O --> H2CO3, how many grams of CO2 react with an excess of water to…
A: Mass of H2CO3 = 640 g Molar mass ( MM) of H2CO3 , = 2 × MM of H + MM of C + 3 × MM…
Q: A mixture of 4 moles of H2 and 3 moles of O2 reacts to make water. Identify: limiting reactant (LR),…
A: Given: A mixture of 4 moles of H2 and 3 moles of O2 reacts to make water. We have to identify the…
Q: 1. Methanol, CH3OH, combusts in air to produce carbon dioxide and water vapor according to the…
A:
Q: KMnO4 and H2C2O4 react according to the following balanced equation: 2 KMnO4 + 5 H2C2O4 + 3 H2SO4…
A:
Q: 3.Given the balanced equation below, determine how many grams of KOH would be produced if 11.7 g of…
A: Given, Balanced chemical equation : 2 K + H2O -----> 2 KOH + H2 mass of potassium = 11.7 g Mass…
Q: If you are given 2.50 g C2H4 and 2.75 g of O2 to conduct the combustion reaction written below: C2H4…
A:
Q: Nitrobenzene C6H5NO2, is an important raw material for the dy industry. It is prepared from benzene…
A:
Q: Given the following balanced reaction 4KNO3 (aq) – 2K20 (s) + 2N2 (g) + 502 (g), fill in the blanks…
A: All the details is explained in handwritten solution.
Q: The balanced equation for the decomposition of potassium chlorate is shown below. When carried out…
A:
Q: The gram quantities of both reactants, C2H6 and O2are given for the following balanced reaction.…
A: The reaction is as follows: 2C2H6 + 7 O2------------>6H2O + 4CO2 The given mass of C2H6 = 15 g.…
Q: Equimolar amounts of H2 (9) and Br2 (9) are injected into an evacuated, rigid container, where they…
A: The reaction given is, Given: Mass of Br2 reacting = 1.6 g.
Q: 75.0g ammonia NH3 reacts with 197.0 g fluorine to produce dinitrogen tetrafluoride and hydrogen…
A: Note : As per our guidelines we are supposed to answer first 3 parts of question please submit other…
Q: used to remove exhaled carbon dioxide from the air supply according to the equation…
A: Molar mass of LiOH = 24 g/mol Molar mass of CO2 = 44 g/mol From balanced reaction Using mass-mass…
Q: Determine the limiting reactant in the reaction. Calculate the amount in grams of CO2 from thew…
A:
Q: Be sure to answer all parts. Propane (C3Hg) is a minor component of natural gas and is used in…
A: Part a) given reaction :- C3H8(g) + O2(g) ---> CO2(g) + H2O(g) Part b) given moles of…
Q: 5. for each of the for the reation and state the termg of the numbers of individual molecules and in…
A:
Q: Given the following balanced reaction 4KNO3 (aq) – 2K20 (s) + 2N2 (g) + 502 (g), fill in the blanks…
A: All details explanation in Handwritten solution.
Q: HCI + Na2S -> H2S + NaCl is given. According to this reaction, if 5 moles of hydrochloric acid…
A: Limiting reactant :- A reactant whose number of moles are less than the required number of moles as…
Q: According to the balanced.chemical equation : CaCl2 (aq) + Na2CO3 (aq)CaCO3 (s) + 2NACI (aq) What is…
A:
Q: Balance the following reaction in KOH. What are the coefficients in front of C5H12O2 and KMnO4 in…
A: The correct answer with explanation is given below.
Q: 9. Methyl alcohol (CH3OH) is burned with 20% excess air. How much unburned oxygen in kg-mol-…
A: Air consists of many gaseous components such as oxygen, nitrogen, sulfur dioxide, etc. The main two…
Q: 13. 12 moles CO and 12 moles Fe2O3 react as follows to produce iron and carbon dioxide. Determine…
A: The balance chemical reaction : 3CO + Fe2O3 ---> 2Fe + 3CO2 Number of moles of CO = 12 moles…
Q: Citric acid, C6H8O7, a component of jams, jellies, and fruity soft drinks, is prepared industrially…
A: The complete balanced equation = C12H22O11 + H2O +3O2 →2C6H8O7 + 4H2O Given in the question, Mass of…
Q: 18. Ammonium bicarbonate, which was classically obtained from deer antlers, is still used for baking…
A: This question can be solved by basic mole concept
Q: 4Al2O3+ 9Fe------> 3Fe3O4 +8Al If 25.4g ofAl2O3 is reacted with 10.2g of Fe, determine the limiting…
A: For getting limiting reagent we have to divide by stoichiometry coefficient to the each substance…
Q: Magnesium oxide can be made by heating magnesium metal in the presence of oxygen. When 10.1g of…
A: The balanced chemical reaction between magnesium and oxygen is shown as,
Q: Consider the reaction between 46.0 g of C2H5OH (l) and 32.0 g of O2 (g) producing CO2 and H2O vapor.…
A:
Q: How many moles of Al will be consumed when 0.800 mol of Al2O3 are produced in the following…
A: the relation between the mole of reactant with the mole of product is known as stoichiometric…
Q: At the factory, a new engineer combines of 1500 L of carbon dioxide and 1500 g of ammonia (NH3). His…
A: Given, A new engineer combines of 1500 L of carbon dioxide and 1500 g of ammonia (NH3) We know…
Q: C12H26O2 underwent combustion with 138g of O2. The Balanced equation is…
A: It is clear from the given reaction that 2 moles of C12H26O2 react with 35 moles of O2 to form 24…
Q: Potassium superoxide, KO2, is used in rebreathing gas masks to generate oxygen. KO2(s) + H2O(l)…
A:
Q: Complete the following reaction and balance the reaction. Select all of the following that are…
A: The given reaction is to be completed and balanced using choices of coefficients. The given equation…
Q: 5. When 122.0g of chlorine gas (CL) reacts with 23.00g of solid phosphorus (P4) in the following…
A:
Q: Copper reacts with iodine in a similar reaction from what was done in lab to produce copper(I)…
A: And the given reaction is:- 2Cu(s) + I2(s) ⟶2CuI(s)
Q: 3.Given the balanced equation below, determine how many grams of KOH would be produced if 11.7 g of…
A:
Q: 4. Magnesium undergoes a single displacement reaction with hydrochloric acid. Balanced Equation: How…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: 6.50 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have…
A:
Q: Phosphine, an extremely poisonous and highly reactive gas, will react with oxygen to form…
A:
Q: Given the following balanced reaction 4KNO3 (aq) → 2K2O (s) + 2N2 (g) + 5O2 (g), fill in the blanks…
A:
Q: Sulfur tetrafluoride, SF4, reacts slowly with oxygen, O2, to form sulfur tetrafluoride monoxide,…
A: Balanced chemical equation is an equation where number of different atoms on reactant side is equal…
Q: for each of the unbalanced equations, indicate how many moles of the second reactant would be…
A:
Q: Given the following unbalanced reaction, answer parts a, b, c, and d. (Not all choices will be used)…
A:
Step by step
Solved in 2 steps with 2 images

- and Employees CH STATE GE 90 ENDULERSE IS FOREVER "ON-Tame Name Jodi Rayal Molarity = wt. x1000 3. Calculate the massof Potassium hydrogen phthalate (KHP) to prepare a 250.0 mL solution of 0.1000 M KHP solution. Mol ut. x Voluse the 2500×1000 204.22250 mass Initial Buret Reading Final Buret Reading Vol NaOH Added Moles of NaOH Moles of HCI Volume of unknown HCI solution Concentration of HCI Average concentration of HCI of KHP = 5.11g 4. Complete the table below for the following neutralization reaction of an unknown concentration of hydrochloric acid with 0.100 M sodium hydroxide: Mu spate solution Trial 1 0 11.15 го data and calculate the concentration 15 Trial 2 11.15 22.15 Trial 3 22.15 33.25 abyh 067 LANDINUKASHU TROWTHCENTISSATE ARAD Ma That par މއކނި REMURNAR MISAMARALISERDRY TileZinc and magnesium react with hydrochloric acid to produce the metal chlorides and hydrogen gas. A 10.00 gram smaple of a mixture of Zn and Mg was with the stoichiometric quanitity of HCl. The reaction mixture was then reacted with 156 mL of 3.00M silver nitrate to produce the maximum quantity of silver chloride. First determine the % magnesium in the mixture- then, if 76.0 mL of HCl was added, what was the molarity of the HCl?The aluminum in a 1.200-g sample of impure ammonium aluminum sulfate was precipitated with aqueous ammonia as the hydrous Al2O3.xH2O. The precipitate was filtered and ignited at to give anhydrous Al2O3, which weighed 0.2001 g. Express the result of this analysis in terms of %NH4Al(So4)2 %Al2O3 %Al
- You are supplied with 1150 kg ethanol (C2H5OH), together with methanol (CH3OH), potassium hydroxide (KOH) and ammonia (NH3). You are requested by a customer to produce methyl acetate (CH3COOCH3), potassium acetate (CH3COOK) and ammonium acetate (CH3COONH4) in mass forms. (a) Calculate the mole & mass of acetic acid (CH3COOH) converted from ethanol through excessive oxidation.25.0mL of a 0.515 M K2S solution is mixed with 30.0 mL of 0.833 M HNO3 acid solution to give the following reaction: K2S(aq) + 2HNO3(aq) → 2KNO3(aq) + H2S(g) H2S is an unwanted by-product in a pulp and paper industry. To capture H2S gas, it is bubbled through a NaOH solution to produce Na2S with a yield of 94%. H2S(g) + 2NaOH (aq) → Na2S (aq) + 2H2O(l) The mass of H2S(g) that was processed (in kg) if 10.76 kg of Na2S was collected isPlease find the CO2 amount and show the calculation: Air composition (apprx) = 80% N2 and 20% O2 (mol basis only)Average molecular weight of Air = (0.8 x 28 + 0.2 x 32) = 29 g/mol (apprx) Math Sample:2kg pure charcoal (12C) is to be burnt completely with air. Find the air, CO2 andN2 amount in kg’s.Solution:C + O2 = CO2 (you must use a balanced equation)Therefore from the mole ratio of the reaction we write,C : O2 : CO2 = 1:1:1and from air composition we gotO2 : N2 : Air = 1:4:5 Given, 2 kg C = 2000 g/ (12 g/mol) = 166.67 mole CTherefore, similar mole of O2 required. So equivalent Air supposed to be 5times than the mole amount of O2 and released N2 will be 4 times than therequired O2. Therefore, Air amount = 5 x 166.67 moles= 833.34 moles= (833.33x29/1000) kg= 24.17 kg air Similarly N2 released amount will be = (166.67 x 4 x 28/1000) kg = 18.67 kgFind CO2 amount by yourself! (Isn’t it 7.34 kg?)
- A 244.5-g sample of ground water is analyzed for calcium. The Ca2+ in the sample is first precipitated and filtered-off as NH4CaPO4.7H20. This precipitate is dried and heated, releasing water and ammonia to yield anhydrous calcium pyrophosphate (CaP2O7). The mass of CaP2O, obtained is 0.0419 g. Give the calcium content of the ground water in parts per million (to three significant figures).The reaction mixture intially contains 4.0 g of ferrous ammonium sulfate hexahydrate, Fe(NH4)2(SO4)2 * 6H2O, and 50mL 1M oxalic acid. Determine the limiting reactant,Theoretical yield of the ferrous oxalate dihydrate, and The per cent yield of the ferrous oxalate dihydrate product.Citric acid, C6H8O7, a component of jams, jellies, and fruity soft drinks, is prepared industrially via fermentation of sucrose by the mold Aspergillus niger. The equation representing this reaction is C12H22O11 + H2O + 3O2 --> 2C6H8O7 + 4H2O Using a metric ton(1000kg) of sucrose the expected yield is 1122Kg. On average if only 1036kg is produced, what is the % yield for this reaction?
- A 19.51 ݃ sample of impure methylamine, which contains 72.58% (by mass) of CH3NH2 , isreacted with 30.81 ݃ of pure oxygen gas:4CHଷNHଶ(g) + 9Oଶ(g) ⟶ 4COଶ(g) + 10HଶO(ℓ) + 2Nଶ(g) In another experiment, this impure methylamine was used as follows: An unknown mass of the impure compound is dissolved in enough water to make 500.0 ݉ܮof solution. 20 ݉ܮ of this solution was transferred by pipette to a clean 250 mL volumetric flask andmade up to the mark. The molarity of the CH3NH2 in the final solution was determined to be 0.103 M.Determine the mass of CH3NH2 present in the original amount of impure compound used tomake this solution.Given Active Ingredient: precipitated sulfur (ointment) Raw Materials: 500 g calcium polysulphide and 1.5 kg hydrochloric acid Actual Yield: 343.4g precipitated sulfur Formulation: 250 mg per jar Dosage form: Ointment packaging:100 jars per box Synthesis and Packaging (Need answer)- Balanced Chemical Equation:- % composition by mass of each compound:- Mass to Mass Stoichiometry Calculation:- Limiting Reagent:- Excess Reagent:- Amount (g) in excess: % Yield:- Number of dosage form and packaging that can be produced from stoichiometric solution:A chemist carries out the reaction below and obtains 3.45g of iron Fe (Fe, MW=55.845g/mol). Fill in the blanks below to determine the percent yield of the reaction when 8.30g of Fe2O3 (MW= 159.69g/mol) and 3.50g of CO (MW=28.01G/mol)were used in the reaction. Fe2O3(s) + 3CO(g) = 2Fe(s) + 3CO2(g) Note that for two of the blanks, will be the same answer. 1. What would be the mass in grams of Fe produced based on the mass of Fe2O3 used? answer: (blank) 2. What would be the mass in grams of Fe produced based on the mass of CO used? answer: (blank) 3. What is the theoretical yield of Fe in grams? answer: (blank) 4. What is the limiting reagent? Enter the formula of compound. answer: (blank) 5. What is the percent yield of this reaction? answer: (blank)

