Q: Toluene and pyridine form ideal solutions. Consider a solution of toluene and pyridine prepared at…
A: Given, At 25 °C, the vapour pressure of toluene = 28 torr And the vapour pressure of pyridine =…
Q: In this experiment, if you decreased the mass of the benzoic acid and kept the same amount of lauric…
A: The melting point of lauric acid and benzoic acid is 43.8oC and 122oC respectively it means at the…
Q: Solutes. 1 M 1 bar none of these O mole fraction of 1
A: Concentration = moles / Volume in litre = Mol/L = 1M Therefore,…
Q: Calculate the mole fraction of CH2O and C3H8O in a solution containing equal masses of both.
A: Molar mass of CH2O = 30 g/mole Molar mass of C3H8O = 60 g/mole
Q: above
A: According to the Henry's law, Solubility of gas depends on partial pressure of gas of the partial…
Q: At a given temperature, pentane with a vapor pressure of 624 torr is mixed with hexane with a vapor…
A: Raoult's law for solution of volatile liquids is P = P0 * x Where P = partial pressure and P0 =…
Q: Calculate the vapor-pressure lowering, AP, of an aqueous solution that is 2.30 m in aluminum…
A: Given information, For 1 kg of H2O, there are 2.30 moles of aluminium chloride, AlCl3 nA = 55.5093…
Q: Determine the molecular weight of the given. Show complete detail of solution. A. Potassium…
A: Molecular weight is the sum of the atomic mass of the individual element present in that particular…
Q: Calculate the mole fraction of each solute and solvent: 583 g of H2SO4 in 1.50 kg of water—the acid…
A: Given: Mass of H2SO4 = 583.0 g Mass of H2O = 1.50 kg = 1500 g Molar mass of H2SO4 = 98.079 g/mol…
Q: 1. Calculate the Theoretical Freezing Point 2. Calculate the Molality
A:
Q: 2. 100ml of sodium hydroxide solution is required 20ml of 0.5 N sulfuric acid solution to neutralize…
A: Answer: This question is based on acid-base titration where with the help the known concentration of…
Q: phosphoric acid is usually obtained as an 85% phosphoric acid solution. if it is 15 M, calculate the…
A: Given information, 85% phosphoric acid solution Concentration = 15 M
Q: Which aqueous solution has a boiling point closer to its predicted/calculated value, 0.030 m KBr or…
A: Check the ∆Tb value for both solution and check which one has higher value.
Q: A 0.045 m solution (consisting of nonvolatile, nonelectrolyte solute) is experimentally found to…
A: Given: Molality of the solution = 0.045 m Freezing point depression = 0.080°C
Q: calculate the amount of sodium chloride required to prepare 40mL of a concentrated solutionso that…
A:
Q: The number of equivalents of solute present in half a liter of a solution of concentration (0.25…
A: Concentration = 0.25Volume of solution = 0.5 LNo of equivalents of solute = To be determined
Q: A 1.6M NaOH basic solution has a density of 1.52g/mL. Calculate the mole fraction of the solvent.
A: Given data, Molarity of NaOH = 1.6M Density of Solution = 1.52g/mL The molar mass of NaOH = 40g/mol
Q: What mass of benzoic acid (C7H6O2) must be used to prepare a 500 mL solution at 0.440 M…
A:
Q: Given these values, find the initial mole fraction and initial mass % of the solute (NaCl) Solvent…
A: Mole fraction of the solute can be written as - Mole fraction of solute NaCl = moles of solute/moles…
Q: It is desired to make a 0.1M solution by adding 500grams of copper sulfate pentahydrate crystals.…
A:
Q: What is the mole fractions of hydrochloric acid (HCI)and water in a 20% aqueous HCI solution?
A: Solution You know that your resolution is two hundredth acid by mass, which suggests that each one…
Q: A solution is made by mixing equal masses of methanol, CH,O, and ethanol, C, H,0. Determine the mole…
A:
Q: The magnitude of boiling point elevation is _________ the freezing point depression for a certain…
A: Correct answer is :- Always less than
Q: Calculate the molarity and mole fraction of acetone in 1.00-m solution of acetone (CH3COCH3) in…
A: Given- The concentration of the solution of acetone in ethanol = 1.00 m 1 mol of acetone in 1 kg of…
Q: t-Butanol has a freezing point of 25.82 oC and a freezing point depression constant of 8.37 oC/m.…
A: Given, Freezing point of t-Butanol = Tf0 = 25.82 oC Freezing point depression constant of t-Butanol…
Q: Assume that when you were transferring the mass of sodium chloride you measured to Test Tube 2, a…
A: Molality = Number of mol of solute/Mass of solvent in Kg Number of mol = Mass/molar mass Freezing…
Q: Prove the statement that an alternative way to express Henry's law of gas solubility is to say that…
A: Henry law explains the solubility of a gas in liquid solution by partial pressure and mole fraction…
Q: The vapor in equilibrium above a hexane-pentane solution at 25°C has a mole fraction of hexane equal…
A: As per Dalton's law, the mole fraction of the component in the vapor phase is given by the…
Q: mole fraction
A:
Q: What will be the freezing point of the resulting solution if a non-electrolyte solution was…
A:
Q: How much water should be added to 14mL of 15% alcohol solution to reduce the concentration to 7%?
A: Given, V1 = 14 ml M1 = 15% V2 = ? M2 = 7 %
Q: One mole of solute X is present for every 25 moles of solution. If the vapor pressure of pure water…
A: Given: The number of moles of solute is 1 mol and the moles of the solution is 25 mol. The vapor…
Q: Rank the following aqueous solutions from lowest predicted boiling point to highest. In the case of…
A: Boiling point is the temperature at which the vapour pressure is equal to the atmospheric pressure.
Q: What is the composition of a methanol (CH, OH) - propanol (CH,CH,CH, OH) solution that has a vapor…
A: Let mole fraction of methanol in liquid phase = Xm By Raoult's law : P = Pm° Xm + Pp° ( 1-Xm)…
Q: Calculate the mole fraction of the ionic species KCl in the solution.
A: Number of atoms, ions and molecules present on a given amount can be determined by “Mole Concept”.…
Q: Which of the following solutes would raise the boiling point of 1.00 kg of water more one mole of…
A: The elevation in boiling point can be given as follows: where ΔTb: Elevation in boiling point…
Q: Calculate the total pressure of an ideal solution consisting of 0.678 mol of benzene and 0.414 mol…
A:
Q: A solution is made by mixing equal masses of methanol, CH,0, and ethanol, C,H,0. Determine the mole…
A: Mole fraction is defined as moles of that component divided by the total moles of each component in…
Q: Raoult's Law relates the vapor pressure of the solvent above the solution to its mole fraction in…
A: The correct option is (c) According to roult's law, a solvent's vapour pressure in a solution is…
Q: Calculate the mole fraction of benzene in solution containing 30% by mass in carbon tetrachteride.
A: Given :- percent by mass of benzene in solution = 30% To calculate :- mole fraction of benzene
Q: For an aqueous solution of HF, determine the van't Hoff factor assuming 0% ionization.
A: For an aqueous solution of HF, assuming 0% ionization means it does not dissociate into ions .…
Q: 9. Calculate the mass percent of sodium bicarbonate in the solution. 10. Calculate the mole fraction…
A: For the solution of question number 9 and question number 10 we will need to find the mass and mole…
Q: If 2.0 moles of a compound A dissolve in 4.0 moles of another substance B, the mole fraction of B…
A: Given, Moles of compound A = 2.0 Moles of compound B = 4.0Mole fraction a component is…
Q: initial mole fraction
A: Molar mass of NaCl=58.44 Molar mass of water=18g Mole fraction of each solute = Number of moles of…
Q: Why is the freezing point of a pure solvent constant, while that a solution decrease with steady…
A: The depression is a colligative property in the freezing point of a solution i.e. it depends upon…
Q: Describe the relationship between the law of mass action,solute concentration, and effective…
A: The law of mass action has a relation between K and concentrations of the reactants and product. It…
Q: Determine the percentages of each element in benzoic acid in terms of: 1. percent by weight
A: C6H5COOH Benzoic acid
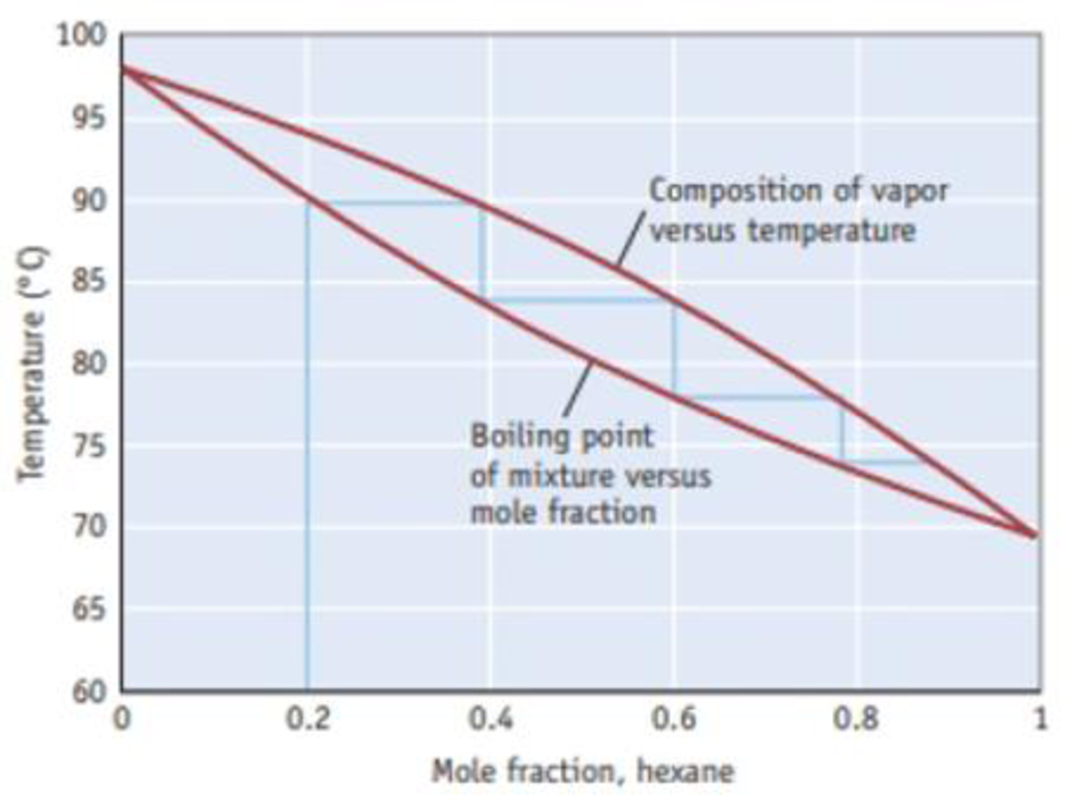
How many theoretical plates are required to produce a solution with a mole fraction of hexane greater than 0.90?
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- . Compatible Polymers A and B are to be blended to achieve a glass-transition temperatureof 60 oC. Tg’s of Polymers A and B are 30 and 85 oC, respectively, using an appropriateequation determine the composition of the blend.Define the relative amounts of liquid and vapor phases in a saturated mixtureare specified by the quality x.A liquid-vapour mixture whose composition is 60 mole % of benzene is contained within a vessel at 60C. What fraction of moles are both methanol and in the liquid phase?
- at a given temprature the vapour pressure ethonal is 44 torr and vapour pressure of methonal is 94 torr. for equmolar mixture the expected vapour pressure of the mixture is?Calculate the change in freezing point of water , with ethalpy of fusion = 6.009 kJ/mole) and the density of ice is 0.9167 g/cc while for that liquid is 0.9999 g/cc for an increase in pressure of 1.00 atm at 273 K.The table below shows temperature/composition data collected for a mixture of methylbenzene (M) and octane (O) at 1 atm. Recall that x stands for the mole fraction in the liquid and y stands for the mole fraction in the vapor in equilibrium. The boiling points for methylbenzene (M) and octane (O) are 110.60C and 125.60C, respectively. Construct the phase diagram with Temperature vs. xM. What is the composition of the vapor in equilibrium with the liquid of composition (a) xM = 0.250 and (b) xO = 0.250. T (0C) 110.9 112.0 114.0 115.8 117.3 119.0 121.1 123.0 xM 0.908 0.795 0.615 0.527 0.408 0.300 0.203 0.097 yM 0.923 0.836 0.698 0.624 0.527 0.410 0.297 0.164
- The vapour pressure of pyridine is 50.0 kPa at 365.7 Kand the normal boi ling point is 388.4 K. W hat is the enthalpyof vaporization of pyridine?Polyethylene glycol 400 (PEG400) is a nonvolatile liquid polymer, which when mixed with water canbe used as a base for parenteral formulations. The designation “400” means that the molecular weightof PEG molecules is approximately 400 g/mol. Preparation with water (ΔHv = 40.7 kJ/mol) requires that the two be mixed at 56 oC, at which the equilibrium vapor pressure of pure water is 16.5 kPa. a. If 105 g of PEG400 are added to 500 g of water at 56 oC, what will the boiling temperature of the solution be?For a given liquid a plot of the lnP versus 1/T gives a straight line with a slope of -3179 K.What is the ΔHvap of this liquid? Your answer should be in kJ/mol and have 3 sig figs
- Crude oil containing 5.0 mole per cent of gasoline is stripped by running the crude down a columnup which live steam is passed, so that 15 kmol of steam are used/100 kmol of oil stripped. Determinethe number of theoretical plates required to reduce the gasoline content to 0.05 mole per cent, assumingthat the crude oil is non-volatile. The vapour–liquid relation of the gasoline in the oil is given by ye = 44x,where ye is the mole fraction in the vapour and x the mole fraction in the liquid. The temperature ismaintained constant by internal heating, so that steam does not condense in the tower.For a given liquid a plot of the lnP versus 1/T gives a straight line with a slope of -4423 K. What is the ΔHvap of this liquid? Your answer should be in kJ/mol and have 3 sig figsCrude oil containing 5.0 mole per cent of gasoline is stripped by running the crude down a columnup which live steam is passed so that 15 kmol of steam are used/100 kmol of oil stripped. Determinethe number of theoretical plates required to reduce the gasoline content to 0.05 mole per cent, assumingthat the crude oil is non-volatile. The vapor–liquid relation of the gasoline in the oil is given by ye = 44x,where ye is the mole fraction in the vapor and x the mole fraction in the liquid. The temperature ismaintained constant by internal heating so that steam does not condense in the tower.

