How much energy is given off in the reaction?
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter18: Glycolysis
Section: Chapter Questions
Problem 14P: Energetic of Fructose-1 ,6-bis P Hydrolysis (Integrates with Chapter 3.) The standard free energy...
Related questions
Question
Question: How much energy is given off in the reaction?
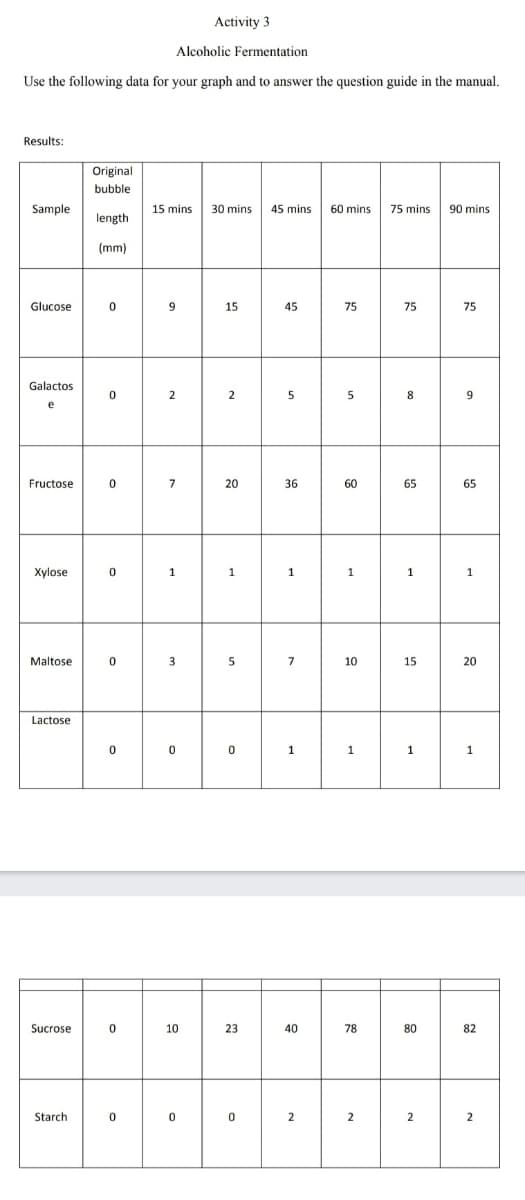
Transcribed Image Text:Activity 3
Alcoholic Fermentation
Use the following data for your graph and to answer the question guide in the manual.
Results:
Original
bubble
Sample
15 mins
30 mins
45 mins
60 mins
75 mins
90 mins
length
(mm)
Glucose
15
45
75
75
75
Galactos
2
2
5
8
9
e
Fructose
7
20
36
60
65
65
Xylose
1
1
1
1
1
1
Maltose
3
7
10
15
20
Lactose
1
1
Sucrose
10
23
40
78
80
82
Starch
2
2
2
2

Transcribed Image Text:ACTIVITY NO 3
ALCOHOLIC FERMENTATION
Introduction
The conversion of carbohydrate such as sugar into an acid or alcohol is called
fermentation. Fermentation that uses yeast is termed as alcoholic fermentation.
Glucose --------→ carbon dioxide and alcohol + energy
The enzyme in yeast catalyzes alcoholic fermentation which actually occurs as a two-
step process. This process takes place under anaerobic conditions such as in the absence of
oxygen liberating a small amount of energy. Some carbohydrates are not hydrolyzed to
simple sugars by yeast under anaerobic conditions due to the lack of the specific enzymes
needed to catalyze the reaction. Fermentation of simple sugars by yeast has different rates of
reaction.
1. The first step involves the decarboxylation of pyruvate from glycolysis to
acetaldehyde acted by the enzyme pyruvate decarboxylase.
2. The second step is the reduction of acetaldehyde which is catalyzed by alcohol
dehydrogenase producing ethanol and carbon dioxide as the end products.
Objectives: At the end of the activity, the student can:
1. Compares the rate of fermentation of the different samples used.
2. Identify which samples are readily fermented, fermented with difficulty
and not fermented at all.
Reagents: (8 ml each sample) 10% glucose, fructose, galactose, maltose, lactose, sucrose,
xylose, starch solutions, 32 ml -10% yeast solution
Materials: 8 plastic cups , 8 test tubes (10 ml), dropper, ruler, 10 ml graduated cylinder,
Test tube rack
Procedure:
1. Obtain 8 clean test tubes and label them.
2. Fill each tube 1/3 full with yeast suspension.
3. Add sample to completely fill the tube.
4. Hold filled tube upright.
5. Place plastic cup on over the tube.
6. Quickly invert the plastic cup. Be careful not to tip the tube. If procedure is correctly
done, only a small air bubble will be present in the upper end of the tube.
7. Measure the length of the air bubble if present. Note its length in millimeter. Record
length as original bubble.
8. Examine the tubes every 15 minute interval and measure length of any bubble as they
develop and increase in size. Subtract the length of the original bubble from all new
measurements to yield net bubble length. Continue observation over a 90 minute period.
9. Plot your data in a graph. Use only 1 graph for all 8 samples
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Concepts of Biology
Biology
ISBN:
9781938168116
Author:
Samantha Fowler, Rebecca Roush, James Wise
Publisher:
OpenStax College

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax