Hybridization Nết Dipole? Steric Number Overall Electron Pair Geometry Formula Lewis Bond Angle(s) Shape Stucture of central atom Fatrahediall 109.5 Yes NH3 3 5+3=8 H -N-H 4 Bent SP CH4 BrFs BrF3
Hybridization Nết Dipole? Steric Number Overall Electron Pair Geometry Formula Lewis Bond Angle(s) Shape Stucture of central atom Fatrahediall 109.5 Yes NH3 3 5+3=8 H -N-H 4 Bent SP CH4 BrFs BrF3
Chapter22: Bulk Electrolysis: Electrogravimetry And Coulometry
Section: Chapter Questions
Problem 22.6QAP
Related questions
Question
Using the first image as an example, what would the second table look like filled in?
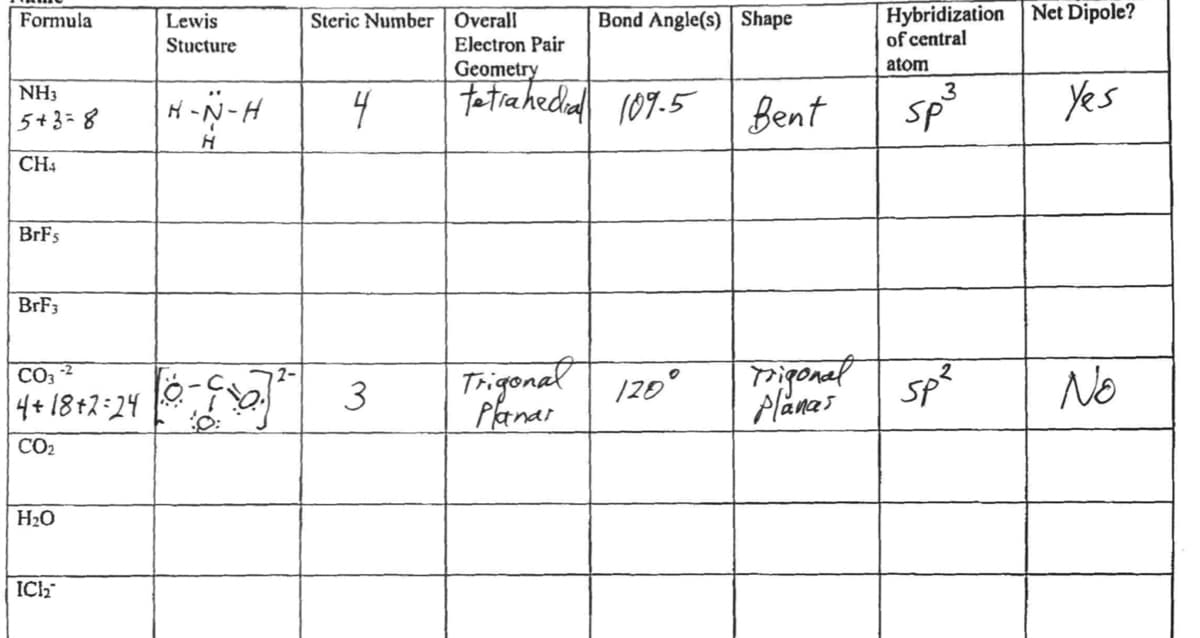
Transcribed Image Text:Net Dipole?
Steric Number | Overall
Electron Pair
Geometry
Hybridization
of central
Formula
Lewis
Bond Angle(s) Shape
Stucture
atom
Fatiahediad 109.5
3
SP
Yes
NH3
H -N-H
4
Bent
5+3=8
CH4
BRF3
Trigonal 120°
Planar
Triponal
sp?
Planai
No
CO3
4+18+2-24
3
CO2
H20
ICh
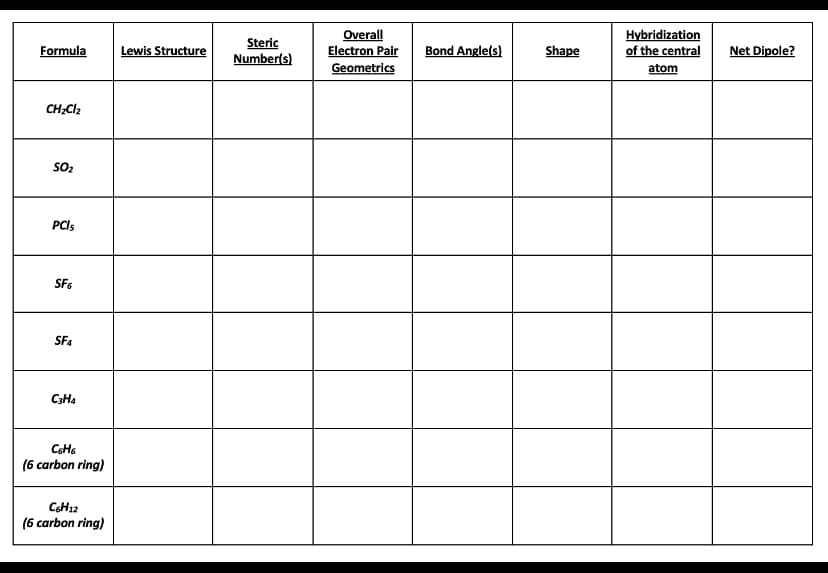
Transcribed Image Text:Overall
Electron Pair
Hybridization
Steric
Number(s)
Formula
Lewis Structure
Bond Angle(s)
Shape
of the central
Net Dipole?
Geometrics
atom
CH;Cl2
SO2
PCIS
SF6
C3H4
(6 carbon ring)
CSH12
(6 carbon ring)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning