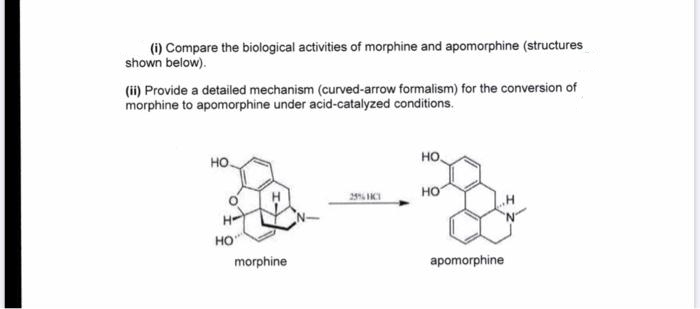
(i) Compare the biological activities of morphine and apomorphine (structures shown below). (ii) Provide a detailed mechanism (curved-arrow formalism) for the conversion of morphine to apomorphine under acid-catalyzed conditions. но. HO но HO morphine apomorphine
Q: Write a mechanism for the formation of cyclic ketal under acid catalysis in benzene. The catalysis…
A: The cyclic ketals or acetal are more stable than acyclic ketals or acetal . Acyclic ketals or acetal…
Q: Write the mechanisms of reaction for the formation of acetanilide from aniline and acetic anhydride
A:
Q: H3C Ph. CH3 Ö 6
A:
Q: Pyridine undergoes electrophilic aromatic substitution preferentially at the 3 position as…
A:
Q: 3, Suggest reactions to accomplish the following transformations in the forward synthesis direction.…
A: The retro-synthetic methodology deals with the schematic representation of the synthesis of produces…
Q: You can see that it is an azo dye, can't you? Knowing that an azo compound is produced for the…
A: Being an antibacterial drug, the Prontosil also called as rubiazol contains the sulfonamide group.…
Q: ® 1,2 Dibromopentane with sodium amide in liquid ammonia. (Predict the product, show reaction)
A: Given reaction is reaction between 1,2 dibromopentane and sodium amide in liquid ammonia.
Q: Using the principles for writing mechanisms and the four common mechanistic steps, write mechanisms…
A: The given reaction is:
Q: Propose a mechanism for the sulfonation of pyridine, and point out why sulfonationoccurs at the…
A: The chemical equation for the sulfonation of pyridine is shown below.
Q: Write the detailed mechanisms for the following transformations.
A:
Q: Give a rearrangement reaction equation to synthesize N-ethyl-2-methyl-buthylamide from the…
A:
Q: The ketone shown was prepared in a three-step sequence from ethyl trifluoroacetate. The first step…
A: Ethyl Trifluoroacetate on reaction with NH3 give amide. Amides on reaction with Grignard reagent…
Q: 2. Outline the synthesis steps for the preparation of the following aromatic amines (with…
A:
Q: Although useful in agriculture as a soil fumigant, methyl bromide is an ozone-depleting chemical,…
A: The mechanism for the formation of methyl bromide from methanol and hydrogen bromide has to be given…
Q: Outline a mechanism for the dehydration of 2-methylcyclohexanol catalyzed by phosphoric acid.…
A: Mechanism : Dehydration of alcohol under acidic condition follows E1 reaction mechanism. It is…
Q: 3. Suggest convenient methods of preparation of each of the following compounds from readily…
A: Interpretation - To prepare the each of following compounds which is given above in the question by…
Q: 4. Predict the biotransformation and conjugation metabolite of below molecule
A: Biotransformation is the alteration of molecule without breaking the bonds.
Q: Treatment of salicylaldehyde (2-hydroxybenzaldehyde) with bromine in glacial acetic acid at 0°C…
A:
Q: Pyrrole undergoes electrophilic aromatic substitution preferentially at the 2 position as…
A:
Q: dimedone reacts with each other, and then with the aminotriazole to form compound 1a in the presence…
A: 1- Benzaldehyde and dimedone both react with each other in the presence of acid because in the…
Q: When 2,2-dibromo-1-phenylpropane is heated overnight with sodium amide at 150 °C, the major product…
A:
Q: Define the Mechanism - Conversion of Acid Chlorides to CarboxylicAcids ?
A: Acid chlorides: Also known as Acyl chloride is one of the derivatives of acid in which OH group is…
Q: Starting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to…
A:
Q: Outline a mechanism for the dehydration of 4-methylcyclohexanol catalyzed by phosphoric acid.
A: To write a mechanism for the dehydration of 4-methylcyclohexanol catalyzed by phosphoric acid.…
Q: Predict the products of reactions of carboxylic acids with alcohols, amines, diazomethane,reducing…
A: Since you have posted multiple questions and have not specified which part you want us to solve, we…
Q: Q1- Formulate clearly the general reaction and the mechanism of the (Sulfonation of Phenol) ?
A:
Q: 4.- Provide an example of the use of (S)-proline as organocatalyst, and mention how this catalyst…
A:
Q: Suggest a retrosynthetic analysis and synthesis for these molecule. OH но, HO.
A: The retrosynthesis is done for any molecule by looking the several factors like electronic…
Q: OMe OMe
A:
Q: The acyl group is a protecting group for amines which can be deprotected by treatment with sodium…
A:
Q: Define the Mechanism - Conversion of Carboxylic Acids to Amides with DCC
A:
Q: When cyclohexanol is dehydrated to cyclohexene, a gummy green substance forms onthe bottom of the…
A: Cyclohexene is prepared on dehydration of cyclohexanol using acid catalysts like phosphoric acid.…
Q: 3 Give mechanisms for the acid-catalyzed and base-promoted alpha-halogenation ofketones. Explain why…
A: The mechanisms for the acid-catalyzed alpha-halogenation of ketones is,
Q: Q6:- Rank the following substances in order of increasing acidity, Justify your choice. benzoic…
A: Acidity It is the tendency of a molecule to donate H+ Inductive effect It is the effect due to the…
Q: 2. Given the following sequence of reactions, draw the structure of products A and B and write a…
A: The primary amine reacts with the ester to form an amide. This reaction goes via a tetrahedral…
Q: Q2. Outline the possible reaction pathways with propose mechanism for the formation of Nylon 6 from…
A: Polymer can be defined as the chemical compound that is formed by the combination of one or more…
Q: Q5:- Give structures and names of the principal organic reactants and products of the following…
A:
Q: Anilinium ion, PhNH3+ , Catalyze semicarbazone formation from benzaldhyde much more effectively than…
A: The semicarbazone is formed when semicarbazide is added to the carbonyl compound, they form imine…
Q: Proline betaine is a putative osmoprotectant in plants and bacteria, helping to prevent dehydration…
A: Given compound:
Q: i. Identify the products obtained when compound 1 is hydrolyzed in the presence of an acid. ii.…
A: The hydrolysis of the above give compound will result in the formation of the following product.
Q: O2N 02 N H2 S04
A: The given compound get rearranged in the presence of an acid catalyst. The compound contains a…
Q: Please show the electron-flow mechanism of the general synthesis of Sulfonamides,this involves…
A: Here we have to draw the mechanism of general synthesis of sulfonamide-
Q: Doxaprost, an orally active bronchodilator patterned after the natural prostaglandins , is…
A:
Step by step
Solved in 2 steps with 1 images

- Explain how benzaldehyde and dimedone reacts with each other, and then with the aminotriazole to form compound 1a in the presence of an acid catalyst. Provide a detailed reaction mechanism. During the development of the optimized procedure for the experiment, it was found out that compound 1b can also be produced from the same set of starting materials. Propose a detailed reaction mechanism for the formation of 1b. Explain your answer. What factor/s may drive the formation of 1b over 1a?Doxaprost, an orally active bronchodilator patterned after the natural prostaglandins , is synthesized in the following series of reactions starting with ethyl 2-oxocyclopentanecarboxylate. Except for the Nef reaction in Step 8, we have seen examples of all other types of reactions involved in this synthesis. Q. Write equations to show that Step 6 can be brought about using either methanol or diazomethane (CH2N2) as a source of the -CH3 in the methyl ester.3. Benzalacetophenone can be nitrated to give the mononitro derivative.(a) Provide the structure of the mononitro derivative.(b) If a second nitro group is introduced, where would it enter? Give the structure of theproduct.
- To synthesize trans-cinnamic acid from benzaldehyde (PhCHO) and acetic anhydride (Ac2O) under basic, refluxing conditions to exemplify the Perkin condensation. 1. Draw the complete chemical reaction to be carried out. 2. Provide a complete arrow-pushing mechanism for the synthesis of trans-cinnamic acid. 3. Briefly describe the role of water, Na2CO3, and HCl in the isolation of the product. Draw the structure of the product at basic pH (after Na2CO3 addition) and at acidic pH (after HCl addition).Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Propose structural formulas for compounds (2) and (3).Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estrone
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.How many chiral centers are present in estrone?Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Show how your proposals for compounds (2) and (3) can be converted to compound (1). (Note: In the course of developing this synthesis, Tietze discovered that vinylic bromides and iodides are more reactive in Heck reactions than are aryl bromides and iodides.)Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q. In the course of the double Heck reactions, two new chiral centers are created. Assume in compound (3), the precursor to rings C and D of estrone, that the fusion of rings C and D is trans and that the angular methyl group is above the plane of the ring. Given this stereochemistry, predict the stereochemistry of…
- Please show the electron-flow mechanism of the general synthesis of Sulfonamides,this involves predicting major and by-products using electronic and structural effects. The arrow push mechanism must be shown.Suggest a reasonable synthetic strategy for synthesizing 3,5-dichlorobenzyl amine from benzaldehyde.Provide the amine and coupling component that can be used as starting materials to synthesize the following dyes



