Q: A galvanic (voltaic) cell consists of an electrode composed of cadmium in a 1.0 M cadmium ion soluti...
A: Given: Cd2+aq+2e-→Cds; ECd2+|Cd0=-0.403 VCo2+aq+2e-→Co(s): ECo2+|Co0=-0.28 V
Q: a) Which salt has minimum solubility at40°C? 200 100 KNO 160 170 160 b) At what temperature solubili...
A: We give answer from the study of solubility curve.
Q: The sky exhibits the color blue due to the scattering of sunlight by air molecules. The blue light h...
A: a. Calculating the wavelength (in nm) associated with this radiation; Given;
Q: Give the alkene and the reagent that are best suited to synthesize two of the following compounds: C...
A: Organic synthesis.
Q: A 4.00 g sample of KCl is dissolved in 55.0 mL of water. The resulting solution is then added to 41....
A: Given, Mass of KCl = 4.00 g Volume of water = 55.0 mL Volume of CaCl2 = 41.0 mL Molarity of CaCl...
Q: 2. When a 2.00-g strip of zinc metal is placed in an aqueous solution containing 2.50 g of silver ni...
A:
Q: What is the maximum mass of S, that can be produced by combining 86.0 g of each reactant? 8 SO, + 16...
A:
Q: O A. m/z = 168 O B. m/z- 125 OC. m/z = 71 O D. m/z - 57 QUESTION 24 Which of the followings reagents...
A: 23. Correct option : A. 24. SN2 reaction occurs at less sterically crowded carbon. Correct option ...
Q: A medication order for a patient calls for 17 mEq of potassium chloride (MW 74.5, valence 1) in 1000...
A:
Q: bicycle pump inflates a tire whose volume is 664 mL until its internal pressure is 5.56 bar at a tem...
A:
Q: H2SO4. + CH3OH + HBr -
A:
Q: Why is it necessary to use a buret for measuring standard AgNO3 solution in volhard method? 2. What...
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for y...
Q: ncreasing acidity:
A:
Q: 4. Why is it important to keep the temperature below 10C in the synthesis of methyl orange experimen...
A:
Q: Please show solution completely Given compound (b), what is the oxidation state of oxygen? Given co...
A: Oxidation state means how much charge present in the atom in a compound. Magnesium carbonate is an i...
Q: Draw structural formulas for the following compounds (a) (2Z,4Z)-2,4-dibromo-3-methylhepta-2,4-diene...
A:
Q: Combining 0.342 mol Fe,O, with excess carbon produced 11.2 g Fe. Fe,0, + 3C – 2 Fe + 3 CO What is th...
A:
Q: INTRODUCTION: Preparation of Benzoic Acid In organometallic compounds, such as Grignard reagents, th...
A:
Q: Identify the functional group (other than aromatic) in this molecule.
A:
Q: Name the alkene. CH2 H2 H3C `CH3 H2 fu
A: Write IUPAC name of the given structure---
Q: 11) What happens during the propagation step in a polymerization? A) Number of Radicals Increase B) ...
A: What happened during the propagation step in polymerization = ? Options are : a). Number of radicals...
Q: C(s) + H20(g) = H2(g) + CO(g)
A: Answer : C) Kp= Kc(RT)∆ng Where ∆ng= Moles of products in gaseous state - moles of reactant in gase...
Q: Question 19 of 25 The molar solubility of Mg(CN)2 is 1.4 x 10-5 M at a certain temperature. Determin...
A:
Q: What volume of a 0.20 M silver nitrate solution is required to precipitate all the Cl ion in the sol...
A: Given, concentration = 0.20 M Ag+(aq) + Cl-(aq) ------> AgCl(s) According to the stoichiometry...
Q: An experimenter performed a titration of an unknown weak base to determine the identity of the weak ...
A: Weak base is one that does not dissociates completely in aqueous solution. It maintains equilibrium ...
Q: 16. Summarize how C-14 and U-238 dating together can be used to determine fossil ages.
A: Carbon 14 with a lifespan of 5,700 years can only be used to cost the remnants of about 50,000 years...
Q: C4H2O4²- + H2O C4H¾O5²- C4H4Og²- AG° = -3.25 kJ/mol (a) Since AG> 0, Keq is less than 1, and the rea...
A: Change in Gibbs free energy indicates whether the reaction will be spontaneous of non-spontaneous or...
Q: The density of NO2 in a 2.84L tank at 735 torr and 25.0*C is ____g/L.
A:
Q: These amounts of CO(g), H,O(g), CO,(g), and H.(g) are introduced into a 10.00-L flask. The flask is ...
A: Recall, the relation between Qc and Kc and the direction of reaction to attain equilibrium, i) Qc &l...
Q: 3. The haloalkane below is heated in a solution of ethanol and undergoes SN1 and EiS a) Draw the fir...
A: Note: The above alkyl halide is heated with ethanol (weak nucleophile) results E1 products as major ...
Q: A 81.0 mL sample of 0.0500 M HBrO4 is titrated with 0.100 M NaOH solution. Calculate the pH after th...
A: The balanced equation for the reaction between HBrO4 and NaOH is HBrO4 + NaOH --------> NaBrO4 ...
Q: A weather system moving through the American Midwest produced rain with an average pH of 5.05. By th...
A:
Q: flask contains a mixture of N2 and H2 at a total pressure of 5.20 atm. If there are 1.00 mol of H2 a...
A: Given: Total pressure = 5.20 atm No.of mole of H2 = 1 mol No.of mole of N2 = 8 mol Calculate: Partia...
Q: 4. Reaction Na + Cl2 → Type of reaction Balanced equation 5. Reaction Ni + AGNO3 → Type of reaction ...
A:
Q: QUESTION 8 The neutralization constant Kn for the neutralization of acetylsalicylic acid (C gH 80 4)...
A:
Q: Choose the equilibrium constant expression for lead(II) chloride dissolving in water. a) Ksp = [Pb2...
A: The solubility product (Ksp) is the equilibrium constant for the dissolution of a solid substance in...
Q: 5. A 0.1093-g sample of impure NażCO3 was analyzed by the Volhard method. After adding 50.00 mL of 0...
A: Mass of sample = 0.1093 g 50 ml of 0.06911 M AgNO3 Back titrated with 0.05781 M KSCN Volume to reach...
Q: Classify each transformation as substitution, elimination, or addition. HO Br а. OH b. lo С. d. HO. ...
A: In substitution reaction one atom or group is substituted by another atom or group. In elimination r...
Q: H2O2. + HCI H2O2. + HBr 1. ВНз, THF 2. НО, Н202, Н20
A:
Q: State with reasoning, whether the molecule BO is likely to exist.
A: Please find your solution below : Any molecule or compound is likely to exist if it is stable. The m...
Q: What is the percent composition of CCl4?
A:
Q: The rate constant of a reaction is 2.10 ×10−5 L/mol·s at 195°C and 4.90 ×10−3 L/mol·s at 258°C. What...
A:
Q: Propane (C3Hg) reacts with oxygen in the air to produce carbon dioxide and water. In a particular ex...
A:
Q: 1. Identify the acid, base, conjugate acid and conjugate base in the following reaction. HSO4" (aq) ...
A:
Q: M =0.01500 M
A: Initial moles of FeCl3 as, = 2.00M × 10.00mL = 20mmoles
Q: Match the thermodynamic property with its description. ____ heat transfer during a chemical process...
A: Entropy tells the randomness of particles.
Q: Consider the following reaction at T = 298 K and at constant pressure: C2H5OH (g) = C2H4 (g) + H20 (...
A: Given: Standard Gibbs free energy of C2H5OH (g) = -174.8 kJ/mol Standard Gibbs free energy of C2H4 (...
Q: Which one of the following processes would produce a decrease in the entropy of the system? a. boil...
A: We have given that Which one of the following processes would produce a decrease in the entropy of ...
Q: Calculate Keq for the following reaction at 25 °C. A + B C + D AG = -1.45 kcal/mol = AG°= - RT LnKea...
A: Given that: dG = -1.45 kcal/mol T = 25°C = 25°C + 273 = 298 K
Q: A mixture containing only KCl and NaBr is analyzed by the Mohr Method. A 0.3172-g sample is dissolve...
A:
Rank the following carbanions in order of increasing stability:
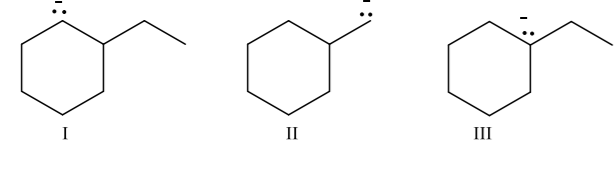
Step by step
Solved in 2 steps with 1 images


