Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter24: Catalytic Carbon-carbon Bond Formation
Section: Chapter Questions
Problem 24.36P
Related questions
Question
iii) please
Transcribed Image Text:v Question Completion Status:
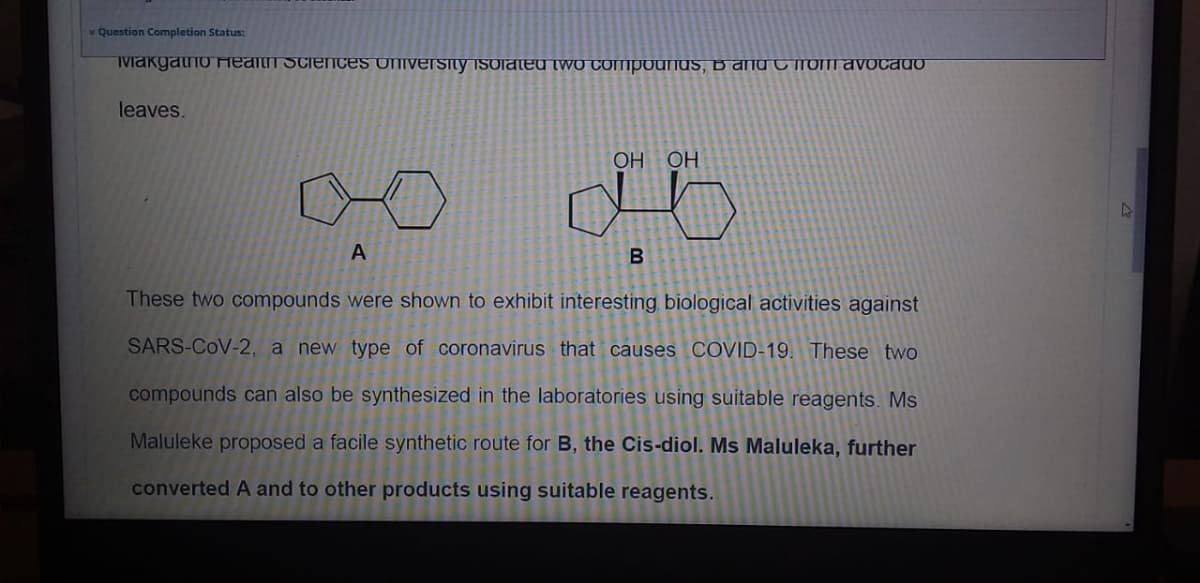
TMakgatno Health SciencES UNIvErsity isoiated two COMpounds, B and C roT avocado
leaves.
НО НО
B
These two compounds were shown to exhibit interesting biological activities against
SARS-CoV-2, a new type of coronavirus that causes COVID-19. These two
compounds can also be synthesized in the laboratories using suitable reagents. Ms
Maluleke proposed a facile synthetic route for B, the Cis-diol. Ms Maluleka, further
converted A and to other products using suitable reagents.
Transcribed Image Text:Question Completion Status:
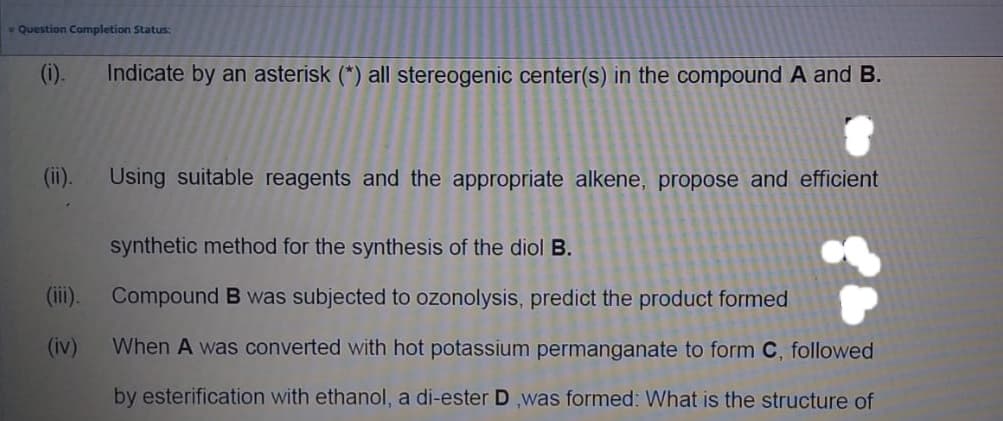
(i).
Indicate by an asterisk (*) all stereogenic center(s) in the compound A and B.
(ii).
Using suitable reagents and the appropriate alkene, propose and efficient
synthetic method for the synthesis of the diol B.
(iii). Compound B was subjected to ozonolysis, predict the product formed
(iv)
When A was converted with hot potassium permanganate to form C, followed
by esterification with ethanol, a di-ester D ,was formed: What is the structure of
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
