i need to caluclate a rf value for the tlc plate and i really need help with showing the steps please! Reduction of 4-Nitrobenzaldehyde to 4-Nitrobenzyl Alcohol and Thin Layer Chromatography
i need to caluclate a rf value for the tlc plate and i really need help with showing the steps please! Reduction of 4-Nitrobenzaldehyde to 4-Nitrobenzyl Alcohol and Thin Layer Chromatography
Chemistry for Today: General, Organic, and Biochemistry
9th Edition
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Chapter12: Unsaturated Hydrocarbons
Section: Chapter Questions
Problem 12.29E: What is an important commercial application of hydrogenation?
Related questions
Question
i need to caluclate a rf value for the tlc plate and i really need help with showing the steps please!
Reduction of 4-Nitrobenzaldehyde to 4-Nitrobenzyl Alcohol and Thin Layer Chromatography
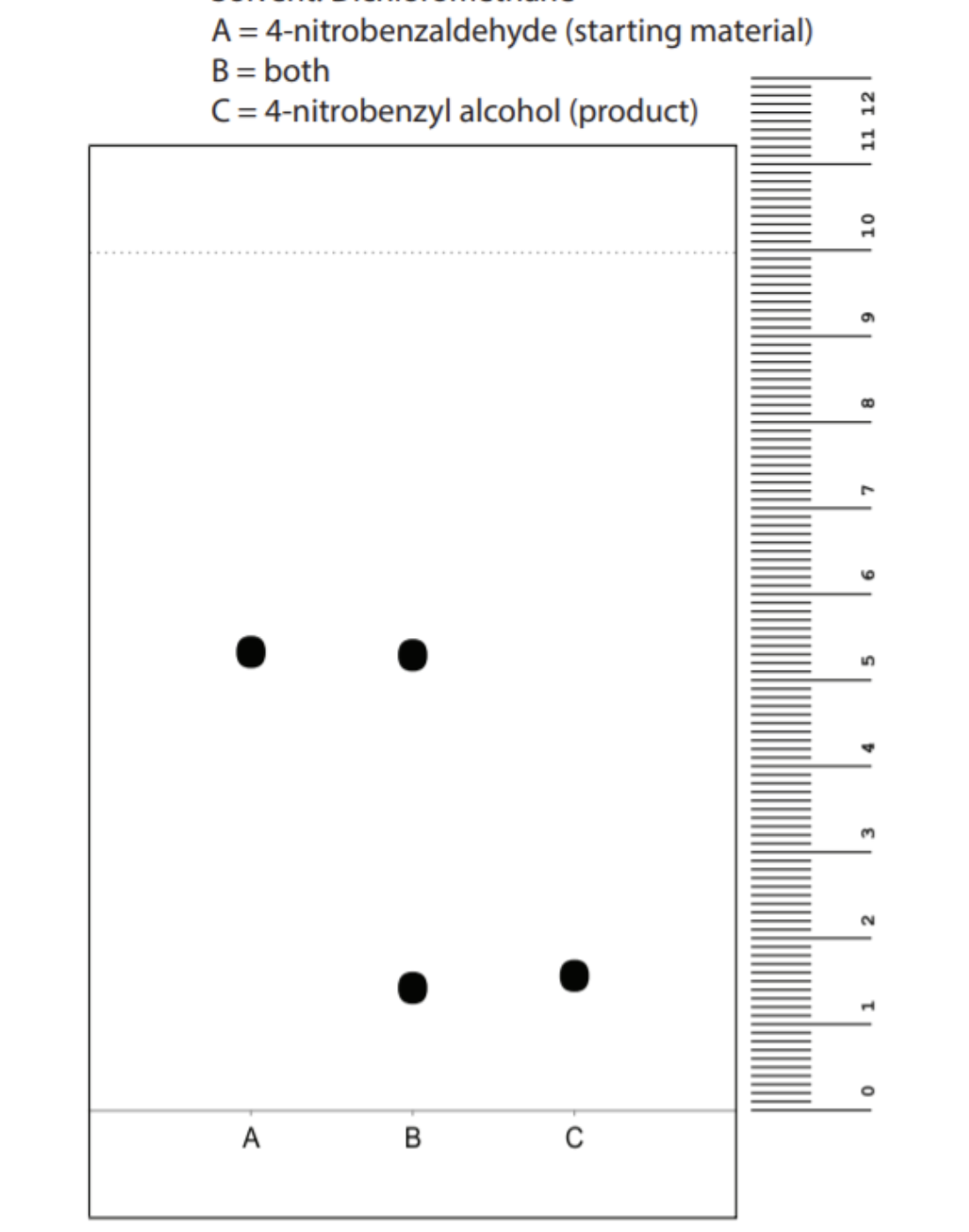
Transcribed Image Text:A = 4-nitrobenzaldehyde (starting material)
B = both
C = 4-nitrobenzyl alcohol (product)
A
B.

Transcribed Image Text:After the reaction has been completed, add about 20 mL of methylene chloride
(dichloromethane/ DCM), and pour the solution into a separatory funnel. Add 20 mL of
water to the separatory funnel. Shake and let the aqueous and organic layers separate.
Place the organic layer (which layer is that?) with your product in an Erlenmeyer flask.
Repeat with another portion of methylene chloride. Combine the organic layers and add a
drying agent. Gravity filter the solution into 100 mL pre-weighed round-bottom flask and
wash the flask and drying agent with an additional 5mL of methylene chloride, also
filtered into your round bottom flask. Evaporate the solvent on a steam bath in the hood
(Erlenmeyer), or use the rotary evaporator (round bottom flask). Weigh the flask again
and calculate the crude yield.
(Be sure all solvent has been removed!)
Purification by recrystallization. Take a melting point of your crude product. Read the
section on recrystallization in your lab text before you come to class. Recrystallization is
a technique for purification of a solid compound. In general, the compound (and any
impurities) is dissolved in a minimal volume of hot solvent (or solvent mixture). This hot
solution is left to cool slowly to room temperature, and then placed in an ice bath. The
solubility of compounds is less in colder solutions, so crystals of compound should form
slowly from the saturated solution. (Since the impurity should be present in small
quantities, the impurity will stay in solution while the compound of interest is
crystallizing). If crystal formation is slow enough, the crystal should be pure compound.
The purified compound is physically separated from solution by filtration. However, if
this crystal formation is fast, impurities may be trapped in the rapidly forming lattice of
the solid crystal. Therefore, it is important to be patient while performing a
recrystallization.
It may help you to see pictures of a movie of the
process.
Recrystallize the product from methylene chloride, and determine the weight percentage
recovery for this purification step once this purified solid has completely dried. If your
TLC of CP1 or CP2 contained multiple spots, take a TLC of the purified compound by
dissolving a few crystals in methylene chloride and eluting with the same solvent as
above (30% ethyl acetate; 70% petroleum ether). Mark this purified product PP.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning