I Review I Constants I Periodic Table Learning Goal: To understand the arrangement of the periodic table and identify certain groups of elements by name (e.g. halogens, metals, lanthanides etc.). Groups and periods The periodic table is a list of elements arranged by atomic number. As you can see in the image (Figure 1) below, the atomic number for each Columns in the periodic table are called groups, and the rows are called periods. Notice that the groups are labeled 1 to 18 and the periods are labeled 1 to 7. Groups 1, 2, and 13 to 18 are called the main group elements, groups 3 to 12 are the transition elements, and the bottom two rows are called the lanthanides and actinides, respectively. element is the whole number in the cell. The atomic This table gives properties and alternate names for some of the groups: mass, written below the symbol here although it may appear elsewhere in some periodic tables, is the average mass of the isotopes of that element. Group Name Properties 1 Alkali metals React to form 1+ ions. 2 Alkaline earths React to form 2+ ions. 17 Halogens React to form 1- ions. Rare gases 18 (noble gases) Stable and unreactive. (Elements from other groups form ions to achieve the same number of electrons as a noble gas.) Metals vs. nonmetals Notice the heavy zigzag line running diagonally across the right part of the table. This line separates metals (below and to the left of the line) from the nonmetals (above and to the right of the line). Metals tend to lose electrons to form positive ions, whereas nonmetals tend to gain electrons to form negative ions. Figure 1 of 1 atomic number 1.008 atomic mass
I Review I Constants I Periodic Table Learning Goal: To understand the arrangement of the periodic table and identify certain groups of elements by name (e.g. halogens, metals, lanthanides etc.). Groups and periods The periodic table is a list of elements arranged by atomic number. As you can see in the image (Figure 1) below, the atomic number for each Columns in the periodic table are called groups, and the rows are called periods. Notice that the groups are labeled 1 to 18 and the periods are labeled 1 to 7. Groups 1, 2, and 13 to 18 are called the main group elements, groups 3 to 12 are the transition elements, and the bottom two rows are called the lanthanides and actinides, respectively. element is the whole number in the cell. The atomic This table gives properties and alternate names for some of the groups: mass, written below the symbol here although it may appear elsewhere in some periodic tables, is the average mass of the isotopes of that element. Group Name Properties 1 Alkali metals React to form 1+ ions. 2 Alkaline earths React to form 2+ ions. 17 Halogens React to form 1- ions. Rare gases 18 (noble gases) Stable and unreactive. (Elements from other groups form ions to achieve the same number of electrons as a noble gas.) Metals vs. nonmetals Notice the heavy zigzag line running diagonally across the right part of the table. This line separates metals (below and to the left of the line) from the nonmetals (above and to the right of the line). Metals tend to lose electrons to form positive ions, whereas nonmetals tend to gain electrons to form negative ions. Figure 1 of 1 atomic number 1.008 atomic mass
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter5: Atomic Theory : The Nuclear Model Of The Atom
Section: Chapter Questions
Problem 62E
Related questions
Question
Please answer question 11 part C and D
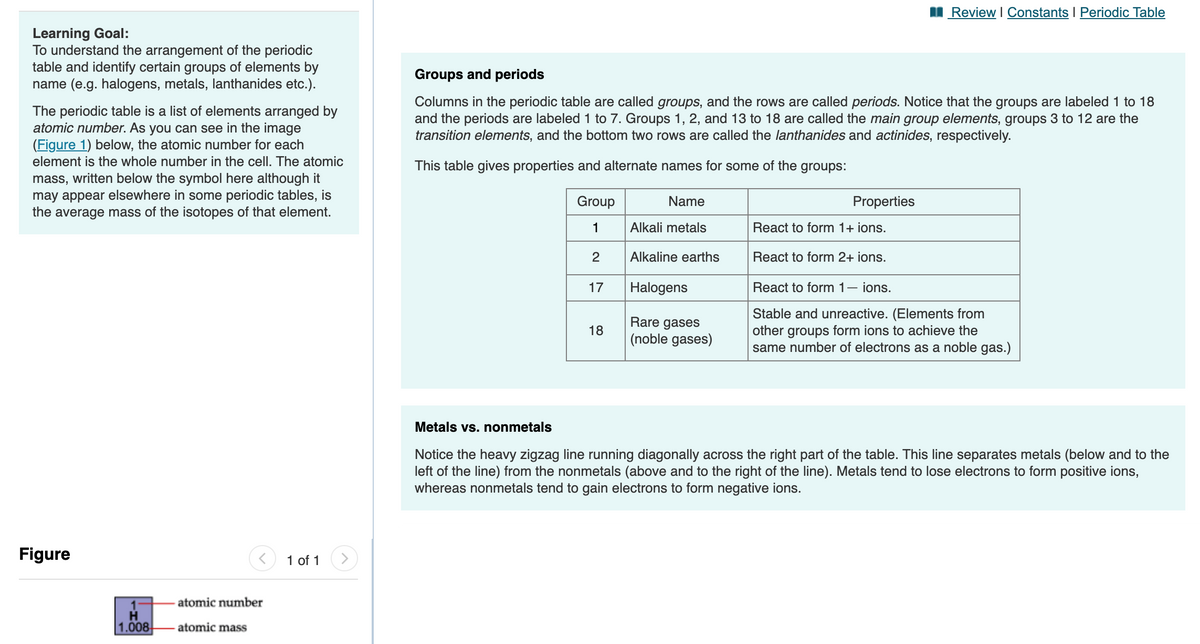
Transcribed Image Text:Review I Constants I Periodic Table
Learning Goal:
To understand the arrangement of the periodic
table and identify certain groups of elements by
name (e.g. halogens, metals, lanthanides etc.).
Groups and periods
The periodic table is a list of elements arranged by
atomic number. As you can see in the image
(Figure 1) below, the atomic number for each
Columns in the periodic table are called groups, and the rows are called periods. Notice that the groups are labeled 1 to 18
and the periods are labeled 1 to 7. Groups 1, 2, and 13 to 18 are called the main group elements, groups 3 to 12 are the
transition elements, and the bottom two rows are called the lanthanides and actinides, respectively.
element is the whole number in the cell. The atomic
This table gives properties and alternate names for some of the groups:
mass, written below the symbol here although it
may appear elsewhere in some periodic tables, is
the average mass of the isotopes of that element.
Group
Name
Properties
1
Alkali metals
React to form 1+ ions.
2
Alkaline earths
React to form 2+ ions.
17
Halogens
React to form 1– ions.
Rare gases
(noble gases)
Stable and unreactive. (Elements from
other groups form ions to achieve the
same number of electrons as a noble gas.)
18
Metals vs. nonmetals
Notice the heavy zigzag line running diagonally across the right part of the table. This line separates metals (below and to the
left of the line) from the nonmetals (above and to the right of the line). Metals tend to lose electrons to form positive ions,
whereas nonmetals tend to gain electrons to form negative ions.
Figure
1 of 1
atomic number
H.
1.008
- atomic mass
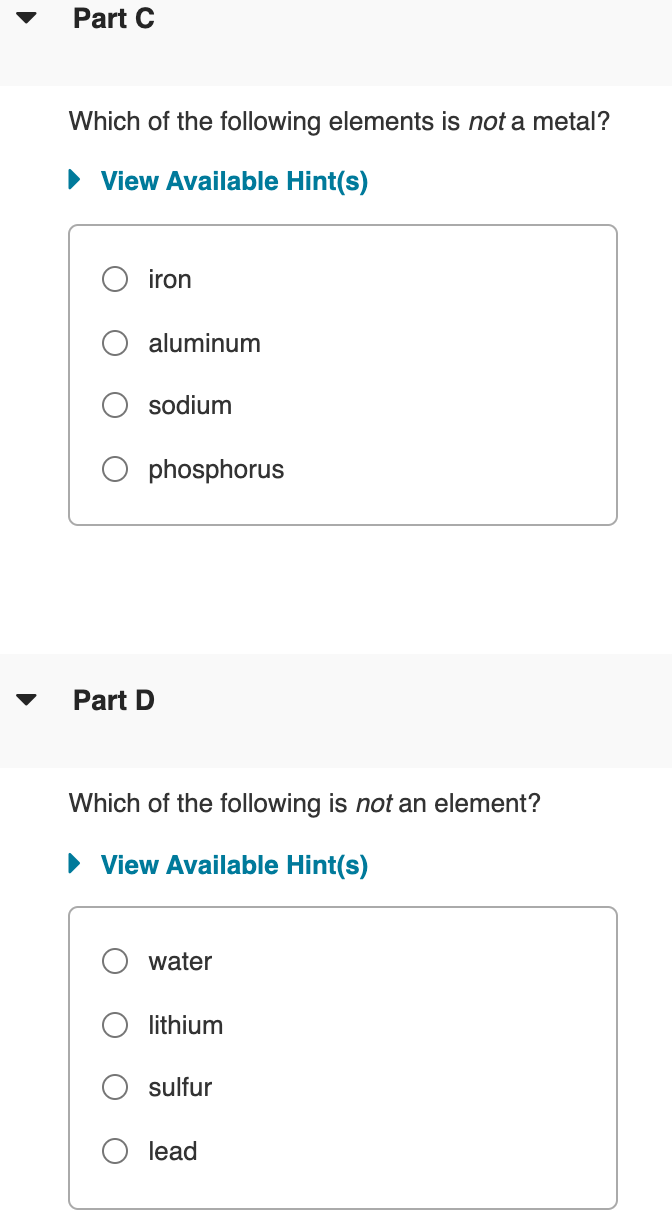
Transcribed Image Text:Part C
Which of the following elements is not a metal?
• View Available Hint(s)
iron
aluminum
sodium
phosphorus
Part D
Which of the following is not an element?
• View Available Hint(s)
water
lithium
sulfur
lead
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning