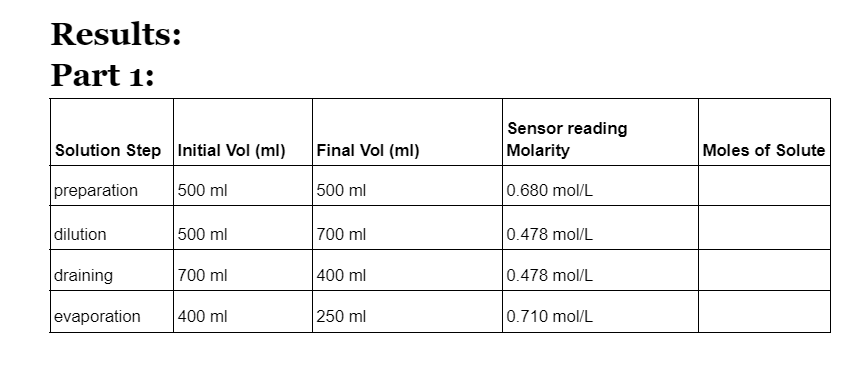
Results: Part 1: Sensor reading Solution Step Initial Vol (ml) Final Vol (ml) Molarity Moles of Solute preparation 500 ml 500 ml 0.680 mol/L dilution 500 ml 700 ml 0.478 mol/L draining 700 ml 400 ml 0.478 mol/L evaporation 400 ml 250 ml 0.710 mol/L
Q: A 15% milk solution is prepared by dissolving g of milk in 0.5L of water.
A:
Q: The interaction of light with a molecule depends on characteristics of the molecule. The presence o...
A: The interaction of light with a substance depends on characteristics of the molecule because every m...
Q: of of of redict whether S1 or Sy2 will be the primary substitution Nal in H20 SN2 LNS HO HO SN1 SN2 ...
A:
Q: thanol is used as disinfectant. 70% of solution is consider a concentration that disrupts biological...
A: Ans. A concentration of about 70% ethyl alcohol is most effective for killing germs. In general, sol...
Q: Predict the products of the electrolysis of an aqueous solution of sodium chloride in cell using pla...
A: Electrolysis is a spontaneous redox process Here we are required to find the product when aqueous so...
Q: By how much had the concentrations changed when the reaction reached equilibrium? The concentration ...
A: A student placed 0.264 mol of PCI3 (g) and 0.180 mol of Cl₂(g) into a 1.00 liter container at 250 °C...
Q: Calculate the [OH-] of 101.40 mL of a buffer initially consisting of 0.1271 M C5H5N and 0.0838 M C5H...
A:
Q: What is the total number of valence electrons represented in the Lewis structures of each of the fol...
A: What is the total number of valence electrons represented in the Lewis structures of each of the fol...
Q: Solve for the mass of NaNO; which is required to saturate 212g of H,O at 50° C.
A: We know that, According to the standard solubility curve, At 50°C,Maximums Mass of NaNO3 soluble in ...
Q: direct answer pls no explanation ANSWER NUMBER 2 ONLY
A: The reaction between solid white phosphorus and oxygen produces solid tetraphosphorusdecoxide.
Q: PRECIPITATION and KSp 2. Calculate the KSP of a. 1.75 x 105 mol of Cd(OH), in a 1.0 L saturated solu...
A:
Q: Balance the following redox equations using the half-reaction method. Please help with a and e bc i’...
A: Balance the reaction:- As2S3 + ClO3- -----> Cl- + H2AsO4- + S P -----> PH3 + H2PO2- Bi(OH)3...
Q: What is the mass of KCIO3 that can be dissolved in 200g of H20 at 90° C?
A:
Q: What is the pH of a 0.500 M solution of trimethylamine (pKb = 4.13)?
A:
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant ...
A: Complete the following table --
Q: Predict the FINAL product for the following synthetic transformation: NH2 HNO, H2SO, NH2 NH2 NH2 O,N...
A:
Q: At 460 °C, the reaction SO2(g) + NO2(g) NO(g) + SO3(g) has Kc = 85.0. Suppose 0.109 mol of SO2, 0.05...
A:
Q: A mixture of nitrogen and oxygen gas is compressed from a volume of 92.0 L to a volume of 85.0 L, wh...
A:
Q: Help with the following questio
A: Given data : Mass of ethanol, M = 301 gram Initial Temperature, T1 = - 3.2 ℃ = (273 - 3.2) K = 269.8...
Q: According to the picture above, Which statement best describes the set of primary air pollutants re-...
A: The primary air pollutants responsible for acid rain is as given below.
Q: 9. NO2 O2N 10.
A:
Q: 4Cr+3o2 -> 2 CrO3 Starting with 1.4 grams of O2, how many moles of Cr2O3 van be produced?
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as...
Q: This energy diagram shows the allowed energy levels of an electron in a certain atom. (Note: the SI ...
A:
Q: For spectrophometric method to be viable, which of the following must be met? a. A high MW complex ...
A: Spectrophotometry is a term which refers to the quantitative analysis of spectra to compare the rela...
Q: CIIBI(CII):CIICII; Br
A:
Q: Balance the equation. Remember to include states of matter. Ni(s) + HCl(aq) → NiCl2(aq) + H2(g)
A:
Q: What is Beer's law? 2. What are the characteristics of Beer's Plot? 3. What are the application of u...
A: Spectroscopy is the study of how light interacts with matter. All molecules absorb and emit light di...
Q: need help writing the theory on a oxidation reduction reaction like the re-dox reaction based on P...
A: Redox reaction is a reaction in which oxidation and reduction takes place simultaneously.
Q: Hydrocarbons are compounds that contain only C and H atoms. When a hydrocarbon reacts with O2, CO2 ...
A: Hydrocarbons are compounds that contain only carbon and hydrogen atoms.
Q: Consider the following reaction: NH4HS(s)=NH3(g)+H2S(g)NH4HS(s)=NH3(g)+H2S(g) An equilibrium mixture...
A:
Q: N204 + 2 NO2 The K, for the reaction above is 2.40 x 10-2 at 1143.15 K. Assume that the initial conc...
A:
Q: Answer: 7. The level of ethyl acetate is determined in colchicines by headspace gas chromatography. ...
A: In the given problem the head space gas chromatography technique is used to determine the amount of ...
Q: Understand how the phase diagram shifts in response to solutes being added to a solvent Understand h...
A: A question based on phase diagram that is to be accomplished.
Q: PRECIPITATION and KSp 3. What concentration of F is necessary to start the precipitation of SrF, fro...
A:
Q: What is Hantaro Nagaoka contribution atomic theory?
A: Atom is the smallest particle which can not be further divided in to any other tiny particle.
Q: 11. From the data in Table 4-1 calculate the heat of combustion of C¿H¿OH(g) t 25°C.
A: The Combustion of ethanol is given by., C2H5OH(g)+3O2(g) ⟶ 2CO2(g) +3H2O(l) Now for the given reacti...
Q: o-chloroiodobenzene 2. 7-ethynyldecatri-1,3,9-triene 3. 2-bromo-2,3-dimethylpetan-2-c 4. 2-bromo-5-e...
A:
Q: Question 1 What is the pH of a 2.19 M solution of methylamine? Report your answer to 2 decimal place...
A: We have to calculate pH of solution.
Q: A chemist measures the enthalpy change AH during the following reaction: H,O(1)→H,O(g) AH=44, kJ Use...
A:
Q: QUESTION 3 Looking at the structure of salicylic acid, it has two different OH groups (carboxylic ac...
A: Answer is given below
Q: What is Henri Becquerel contribution to the atomic theory? Explain in detail.
A: Answer - discovery of radioactivity in uranium by French physicist Henri Becquerel in 1896 forced sc...
Q: Question 1 You will observe a weak acid-strong base titration in this experiment. Select all stateme...
A: Please find your solution below : The acid-base titration is a technique which is used to determine ...
Q: 5. A solution of 0.09 M in Mg²+ and 0.33 M in NH.. What minimum concentration of NH, will cause Mg(O...
A:
Q: Calculate the wavelength of light required to break the bond between the two chlorine atoms in a chl...
A: Given, The wavelength of light required to break the bond between the two chlorine atoms in a chlor...
Q: SeF4 has a tetrahedral structure. Bond angles for IF6+ are 60 °. Selenium tetrafluoride (SeF4) is a...
A:
Q: Which of the following statements is/are true? (Select all that apply) OThe higher the concentration...
A:
Q: = 0.400 M, B] = 1.00 M , and [C] = 0.300 A mixture initially contains A, B, and C in the following c...
A:
Q: TUS V Complete the table below. Be sure each of your answer entries has the correct number of signif...
A:
Q: What will be the equilibrium concentration of H2O?
A:
Q: NO2- ion -- How many lone-pairs of electrons are on the central N atom? AsF5 molecule -- How many l...
A: Given, NO2- ion -- The number of lone-pairs of electrons are on the central N atom is: AsF5 molecul...
Step by step
Solved in 3 steps

- Standardization of Sodium Thiosulfate Solution Primary Standard used: Potassium Dichromate Formula mass of 1o standard: __________________ % Purity of 1o standard: 99.80% Trials 1 2 3 Weight of K2Cr2O7 (g)Weight of K2Cr2O7 (g) 0.0315 0.0331 0.0380 Final Volume Reading Na2S2O3 (ml) 27.50 27.50 34.90 Initial Volume Reading Na2S2O3 (ml) 1.10 0.00 1.00 Net Volume Na2S2O3 used (ml) Molarity of Na2S2O3Density of solution:Trial 1: 1.2 g/mLTrial 2: 1.2 g/mLTrial 3: 1.2 g/mL Average density = 1.2 g/mL What is the relative average deviaion, %?Determine the concentration of linalool in a highly valued lavender essential oil. The peak areas of a series of standard solutions (calibrations standards) acquired from GC-MS (gas chromatography-mass spectrometry) is presented below. Concentration (mg/L) Peak area 0 201 25 5214 50 14658 75 23647 100 32657 125 42652 150 52478 QUESTION: Calculate the concentration of linalool in yourhighly valued lavender essential oil which returns a peak area of 27235.
- I would need help with these questions. The method referenced above was followed by a student and she got the following results: Caffeine Std. Conc. (ppm) Absorbance 100 1.806 50 0.899 40 0.724 30 0.545 20 0.365 10 0.183 Further, she analyzed an unknown sample and she got the following results: Sample # Absorbance 1 0.398 1. Graphically find the concentration in ppm and then calculate in mol/L of Caffeine in Sample #1? 2. Which solvent was used to extract Caffeine and why? Why is it necessary to do extraction three times? 3. Cuvettes used in this experiment were made from which material and why? 4. At what wavelength the Absorbance was measured?EX 2.Given-> Volume of Na+ = 500 ml Molarity of Na+= 0.0100M Molar mass of Na2CO3 = 105.99 gm/mole Millimole of Na+ = molarity × volume Number of millimole = 0.0100 × 500 = 5 millimole Na2CO3 ---> 2Na+ + CO32- Millimole of Na2CO3 = millimole of Na+/2 Millimole of Na2CO3 =5/2 = 2.5 millimole Mole of Na2CO3 = 2.5 × 10-3mole (1 mole = 10^3 millimole) Weight of Na2CO3 required = mole × molar mass = 2.5 × 10-3 × 105.99 =0.26 gm Hence, 0.26 gm Na2CO3 must dissolve in 500 ml of water.A 18 g of unknown organic sample was dissolve in 756 mL of benzene. The boiling point of benzene was increased by 3.36oC. As the first step of analysis, determine the moecular weight of the unknow sample? Kb of benzene= 2.64oC/m Bb of benzene = 80.09 oC density of benzene = 0.874 g/mL at 25 °C Answer in whole number, no units required.
- Trial 1 Trial 2 Trial 3 Initial burette reading (mL) 2.29 1.41 1.95 Molarity of NaOH (M) 0.100 0.100 0.100 Volume of vinegar sample (mL) 5.00 5.00 5.00 Final burette reading (mL) 50.37 49.39 49.84 Table 2. Titration data Trial 1 Trial 2 Trial 3 Initial burette reading (mL) 2.29 1.41 1.95 Molarity of NaOH (M) 0.100 0.100 0.100 Volume of vinegar sample (mL) 5.00 5.00 5.00 Final burette reading (mL) 50.37 49.39 49.84 Expected color at end point Volume of NaOH used (mL) 48.08 47.98 47.89 Compute for the ff: a. Average moles of acetic acid (mol)? b. Average molarity of acetic acid (M)? c. Average molarity of acetic acid (M)?SALT SOLUTION: Trial 1: Volume: 25 mLMass of 25mL solution: 29.824 gDensity: 1.2 g/mL Trial 2:Volume: 25 mLMass of 25 mL solution: 29.855 gDensity: 1.2 g/mL Trial 3:Volume: 25 mLMass of 25 mL solution: 29.816 gDensity: 1.2 g/mL METAL BAR: Method I - Trial 1:Mass of metal bar: 57.063 gVolume of metal bar: 6.7 cm^3Density of bar: 8.5 g/cm^3 Trial 2:Mass of metal bar: 57.063 gVolume of metal bar: 6.0 cm^3Density of bar: 9.5 g/cm^3 Trial 3:Mass of metal bar: 57.063 gVolume of metal bar: 6.0 cm^3Density of bar: 9.5 g/cm^3 Method 2 - Trial 1:Dimensions: diameter = 1.20 cm, height = 4.91 cmMass of metal bar: 57.063 gVolume of metal bar: 5.55 cm^3Density of bar: 10.3 g/cm^3 Trial 2:Dimensions: diameter = 1.20 cm, height = 4.95 cmMass of metal bar: 57.063 gVolume of metal bar: 5.60 cm^3Density of bar: 10.2 g/cm^3 Trial 3:Dimensions: diameter = 1.20 cm, height = 4.95 cmMass of metal bar: 57.063 gVolume of metal bar: 5.60 cm^3Density of bar: 10.2 g/cm^3 QUESTION - From your data, calculate the…From a 10-mL sample, a 1-mL aliquot was taken and diluted to 100mL. From this, a 5-mL aliquot was taken and diluted to 20mL.The final 20mL was found to have a concentration of 0.004M analyte X.What is the concentration of analyte X in the 10-mL sample?
- Table of caffeine standards concentration . Sample Conc, ppm Std1 16 Std2 32 Std3 48 Std4 64 Std5 80 If the volume used to make 100 mL of std 1 is 2 uL what is the concentration in M used to make std calibration curve ? The standards are going to be used to build calibration curve to analyze caffeine in an energy drink. if 500 mL of the energy drink has target of 400 mg , how will you prepare the sample if you need 10 mL for the analysis ? Caffeine MM=194.19 g/mol.Sample AnalysisSource of Water sample:Tap Water Molarity of Na2S2O3 1 2 3 0.0243 0.0245 0.0228 Average Molarity of Na2S2O3 0.0239 Trials 1 2 3 Volume of sample used 100.00 100.00 100.00 Final Volume Reading Na2S2O3 (ml) 6.70 10.00 13.10 Initial Volume Reading Na2S2O3 (ml) 3.40 6.70 10.00 Net VolumeNa2S2O3 used (ml) 3.30 3.30 3.10 mg O2 in sample ppm O2 in sample average ppm O2 in sampleMethodology: Make schematic diagram for the procedure below B. % SO3 determination Dry the soluble sulfate sample at 100° C for 1-2 hours, and cool in desiccator. Weigh out 0.5 – 0.7 g (± 0.3 mg) duplicates sample. Transfer to 400-mL beakers. Dissolve each in 200 mL of dist. H2O + 4 mL of 6 m HCl. Precipitant is prepared by dissolving 1.3 g BaCl2 ∙ H2O in 100 mL water. Filter if solution is not perfectly clear. Heat the BaCl2 solution and sulfate sample solution nearly to boiling, and then add the entire amount to the HOT sulfate sample solution while stirring vigorously. Wash stirring rod with distilled water and include washings in your final mixture. Digest by letting stand 1-2 hours. Overnight standing is acceptable. Filter through an ash less filter paper of fine porosity (Whatman no. 42). Slowly pour the supernatant liquid through the filter paper.

