Q: Calculate [H3O+] and the pH of each H₂SO4 solution (Ka2 = 0.012). At approximately what…
A: It is di protic acid So there are two different dissociation constants Ka1 and Ka2
Q: 5. At 25°C, calculate the molar H3O+ and OH-1 concentrations of the following: b. 0.0600 M NH3 c.…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: lowest pKa to highest pKa.
A:
Q: Q: The pOH of a weak acid is (8.27)and pKa=4.95 , So the concentration of this acid is: * O 4E-3M О…
A: For any weak acid, HA, the dissociation occurs as below : HA --> H+ + A- Thus, the…
Q: Which factors impact the relative stability of the two indicated bases, and in what way? O2N A B
A: In the given question we have to show the relative stability of indicated bases. here, base A is…
Q: HCl А b) NBS В + С hv
A: A nucleophile is an electron-rich species and it may have a negative charge or unshared electron…
Q: 0.06 moles of hydrochloric acid is added to a solution that contains 0.24 mol bromoacetic acid and…
A: Given information, 0.06 moles of hydrochloric acid is added to a solution that contains 0.24 mol…
Q: Determine whether the given salt will undergo hydrolysis or not. Write: YES or NO (Note: case &…
A:
Q: Carbonic Acid Buffers and the case of the child that drinks 1L of Cherry Coke® 10 Reasons to NEVER…
A: Buffer:A buffer is a solution of equimolar concentration of weak acid and its conjugate base or weak…
Q: In a 2.31M C2H5NH2 aqueous solution, determine the percentage of ionised C2H5NH2 molecules. Kb is…
A: We have to calculate the percentage ionization of given weak base.
Q: Learning Task 3 dentify the buffer systems below: 1. KH2PO4/ H3PO4 2. NaCIO4 /HCIO4 3. KF /HF 4.…
A: The question is based on the concept of buffer solution. A buffer is a solution which resist any…
Q: what is the equation that best describes what happens H2S dissolves in water? a. H2S (g) --> H2S…
A: We will find the equation that best describes what happens H2S dissolves in water. H2S is a gas.
Q: Rank the following substances in order of increasing acid strength. (1 as least and 4 as most in…
A:
Q: What is the AH° for the following reaction: H2O (1) + CH4 (g) --> CH;OH (I) + H2 (g) Given the AHf…
A: Consider the given reaction is as follows; H2O l + CH4 g → CH3OH l + H2 g The value of ∆H° for the…
Q: 5. At 25°C, calculate the molar H3O+ and OH-1 concentrations of the following: c. 0.200 M C6H5COONa…
A: The Given data for acid, base, salt
Q: provide all steps
A: It is the hydrolysis of the cyclic acetal in the acidic medium.
Q: Calculate the pH of a 0.381 M solution of ethylenediamine (H, NCH, CH,NH,). The pK, values for the…
A:
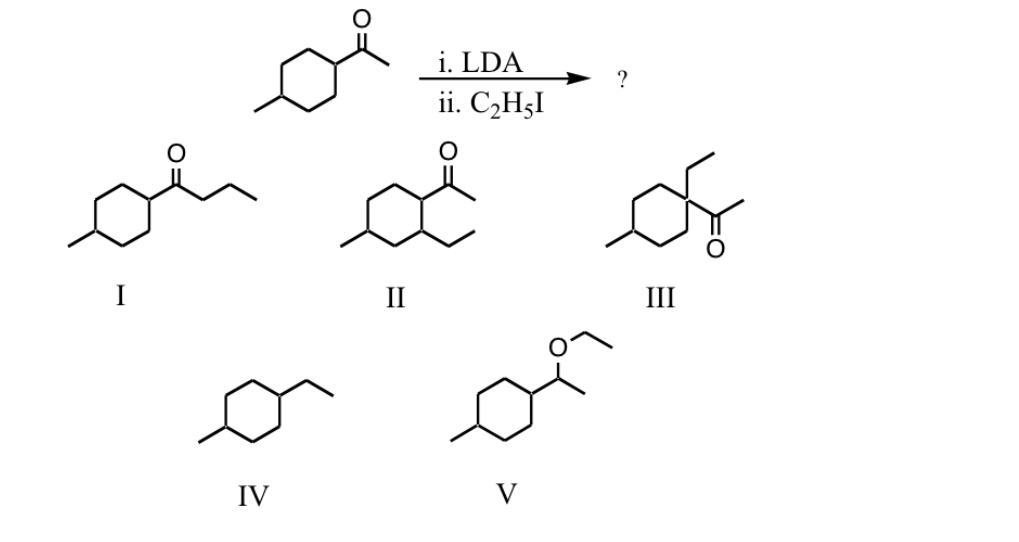
Q: What is the missing reactant? LDA -78°C [enolate salt] ? CH-OH THE I. II. III. IV. CH3-Br H-C-oc,H5…
A: Given reaction is : What is the missing reactant ? Options are :
Q: 5. At 25°C, calculate the molar H3O+ and OH-1 concentrations of the following: a. 0.0300 M C6H5COOH
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: NaOH, DMSO solvent Br Ph Br NaOH, DMSO solvent Ph Br NaOH, DMSO solvent Br LDA, THF solvent
A:
Q: A 0.9182 g sample of CaBr2 is dissolved in enough water to give 500. mL of solution. What is the…
A: Solution: Number of moles present in a solution give the ratio of number of moles of solute to the…
Q: Haw many L Of 2,40 M HCI are required to react completly with 14.0g of Al CoH)s LHCI Al COH); t 3HCI…
A:
Q: OH NH A dnsohe in ether Add NaHCO, walet lav eher byer 1. Brine wash 2. MgSo. 3. evaporation Add HCI…
A: Solution: The given ternary mixture is of ketone, carboxylic acid and an amide with class neutral,…
Q: les of LIOH Would be required to completely neutralize 6.0 H2CO3 + 2LIOH 2H20 + LizCO3
A: In neutralization reaction, an acid and a base react to form water and a salt. It involves the…
Q: i) CH3CH2COH ii) F3CCOH ii) CI3CCH,COH
A: Acids and Bases are defined based on several theories. One of among those theories is Bronsted–Lowry…
Q: Determine whether the given salt will undergo hydrolysis or not. Write: YES or NO (Note: case &…
A: Hydrolysis is the process of the addition of water to a particular compound. The H2O water molecule…
Q: Which of the following will dissolve in a nonpolar solvent? Mark all that apply KOH PCl3 CH4…
A: Given compounds are : a). KOH b). PCl3 c). CH4 d). NH3 e). I2 Which of the…
Q: Determine whether the given salt will undergo hydrolysis or not. Write: YES or NO (Note: case &…
A: Hydrolysis of Calcium Chloride (CaCl2 ) - The term 'Hydrolysis' means reaction with water.…
Q: why is it that we need to take a drug that contains Famotidine Calcium Carbonate Magnesium…
A: FODMAP intolerance is one of the most common causes of bloating and abdominal pain. If you strongly…
Q: 9. NH-acidic properties are shown by? A. Sulfacyl sodium B. Sodium barbital C. Theobromine D.…
A:
Q: 5 ml of a 0.2 M NH3 solution are added to 5 ml of a 0.1 M NHẠCI solution (Kb for NH3 is = 1.8 ×…
A: Answer: Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: II. Determine the most reduced rat compound. Write your answers in yourr Chemical Formula Empin 1.)…
A: Given chemical formula change to Empirical formula--
Q: what is the equation that best describes what happens H2S dissolves in water?
A: H2S is and acid and it behave like arhenius acid when dissolve in water. Arhenius acid - Those…
Q: Ethanoic acid is a weak acid pka value 4.74 at 298K. Calculate the pH value at 298K of: a 0.1M…
A:
Q: what Reagents can conuert it to the product Draw the two major product. Use ph for phenyi Br ).pph3…
A:
Q: CI OMe OMe LDA (3.0 equiv); Mel Me- -OMe
A: In this question, we will draw the reaction mechanism in the presence of LDA LDA is strong base and…
Q: 1. LDA, THF 2. add slowly a) 3. Н.О 1. NaOCH¿CH3 b) 2 2. H,0* 1. LDA, THF 2. add slowly с) `OCH3…
A: Note: According to our guidelines we are supposed to answer only first three subpart. Kindly repost…
Q: 1. H2O NaOH + 2. + H2O → HaPO. 3. Na:COa NaOH +
A: Following are the balanced chemical equations . Since it is a multipart questions,I solved 1st 3…
Q: Fe3+(aq) + SCN–(aq) ⇆ FeSCN2+(aq) Suppose that the solution of NaSCN was accidentally…
A: Equilibrium constant (Keq) :- It is the ratio of concentration of products to the concentration of…
Q: Select the two species below that are not amphoteric: H2O NH3 HCO3- NH4+ Cl- HF
A: Given species are - H2O NH3 HCO3- NH4+ Cl- HF (1) H2O - water is an amphoteric substance ,…
Q: The MAJOR result of the above reaction is which of the following? NaOEt ELOH Br I. П. ÖEt III.
A: The above reaction is a well example of elimination reaction in the presence of strong base to yield…
Q: Which factors impact the relative stability of the two indicated bases, and in what way? O2N A В
A: There are different factor which help in stabilise the species 1) inductive effect is the dragging…
Q: Rank these compounds (attach photo) in terms of increasing acidity IV II > III > IV IV IV > II >…
A:
Q: Which one of the following is the equilibrium reaction of basic C6H5NH2 with H20? Select one: a.…
A: Given Reactant = C6H5NH2 and H2O Product = ?
Q: The concentration of H2S in a saturated aqueous solution at room temperature is approximately 0.1 M.…
A: The given chemical reactions are : H2S(aq) + H2O(l) ⇌ H3O+(aq) + HS−(aq) Ka1 = 8.9 ×…
Q: pnk the campounds în onder of decresling acidity?
A: Acidic nature of aromatic Carboxylic acid (benzoic acid) increases with increasing the number of…
Q: a If an acid, HA, dissolves in water such that the Ka is 70000, what is the pK, of that acid? pKa
A: Applying formula Pka= -log ka
Q: Assignment Score: 45.9% Question 12 of 19 What concentration of CI re [CI¯] =
A: Here is your solution
Q: what ?s mecaniss Br n-Buli PHCN Ph. Et20, -78 °C then aq. HCI 61%
A: In the first step the 3-bromopyridine undergoes Li exchange with n-BuLi. In the next step the anion…
Q: The pKa of Cl3CCO2H is 0.64. What is the pH of a 0.580 M solution? What percentage of the Cl3CCO2H…
A: For weak acid : pH = 0.5 x pKa - 0.5 log C pH = 0.5 x 0.64 - 0.5 x log 0.580 pH = 0.32 + 0.1183 =…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Don't give handwritten answers please otherwise I'll down voPlease make the answer clear to understandCalculate for the theoretical yield of Rochelle salt (MW = 282.83) (a) that will be obtained from 6.6 grams of monohydrated Sodium carbonate (124.01). What is the reacting ratio? Refer to the choices below. (b) that will be obtained from 15g of Potassium bitartrate (MW = 188.10). Refer to the choices below. Rochelle salt: 2KHC4H4O6 + Na2CO3.H2O + 6H2O ---->2 C4H4O6KNa.4H2O +CO2 A 22.55 g B 25.47 g C 30.11 g D 32.64 g



