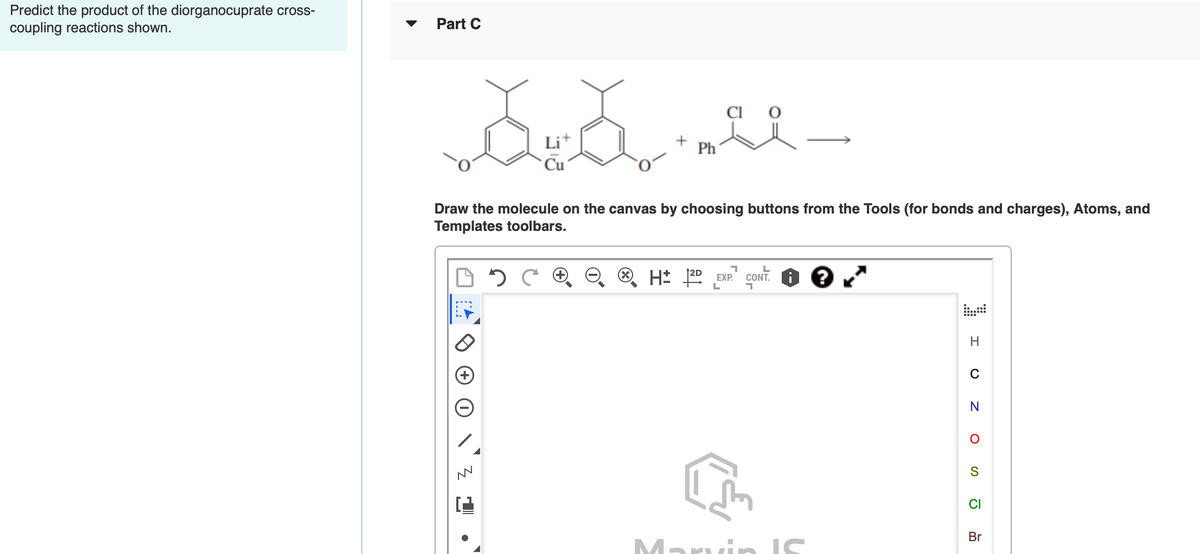
Ictions shown Li+ Ph Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and charges), Atoms, and Templates toolbars. 2C ® O ® H* 2 EXP CONT. H N CI Br
Q: 1. CH3CH20 .C. OCH,CH3 2. Hа CH;CH, CH;CH,O OCH,CH3 add slowly Draw the molecule on the canvas by…
A: The reaction will be:
Q: baamlm9|bM 30 te 00 BK 17 Using electronegativity calculations, tell if one bond in the following…
A: Nonpolar Covalent: This type of bond occurs when there is equal sharing (between the two atoms) of…
Q: The following Lewis dot structure would correspond to which ionic compound? 12- M* O None of these O…
A: The Lewis structure or molecular skeleton is also known as electron dot structure. It is the…
Q: Name each of the following molecular compounds: gSiFA b. IBra
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: The chemical structure of ethylene glycol ((CH), (OH)2) is shown below. Highlight each atom that is…
A: The methylene group is a group where two hydrogen are bonded to carbon atom and the carbon is…
Q: Which of the following gases contain carbon atoms? O C, O CO O NO O HF O N,0 O Ne O NH, ON, ONO, H,…
A: Different gases are given Carbon Containing gases = ?
Q: below as ionic or molecular, based on their valence electrons. NaCI Caz0 CO2 II
A: NaCl -----> Ionic Ca2O -------> Ionic CO2 --------> Molecular
Q: The skeleton of acetic acid is shown. Complete the structure by adding bonds and nonbonding…
A: Given here the skeleton of acetic acid and we are asked to complete thestructure by adding bonds and…
Q: 2) Label the following bonds as polar, non-polar or ionic C-Br Na-Cl H-O…
A:
Q: Which of the following is not a covalent bond? C-C H-Br Na-F 0=0
A: Na-F have ionic bond not covalent bond.
Q: How many carbons are in the following structure? A) 8 B) 10 Br C) II D) 12 E) 13
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Which one of the compounds below is most likely to be ionic? A. CaH2 B. NO2 C. SF4 D. H20
A:
Q: raw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced…
A: The structure of o-nitro aniline:
Q: 5. Which of the following is a noble gas? a. Ne С. О b. N d. H O A O B O C OD
A: Noble gases are the group 18 elements having 8 valence electrons. The general electronic…
Q: Pairs of molecules Турe(s) IMF 1. O=C%3DO and O=C3D0 2. and Na + H' H.
A: Since you have asked a question with multiple subparts, we will answer only first three subparts for…
Q: complete
A: We must maintain four bonds to accommodate all its valance electrons available to pair.
Q: Calculate the molecular mass of the compounds represented by the chemical structures. Round to the…
A:
Q: с. ОН H2O ? acid Draw the molecule on the canvas by choosing buttons from the Tools (for bonds),…
A:
Q: 4. Determine the difference in electronegativity ( JAe.n.|) for each of the following bonds, then…
A:
Q: Part A 1-CH;–Znl (2 equiv.) Draw the molecule on the canvas by choosing buttons from the Tools (for…
A: Both CH2ZnI2 and CH2N2 are used as cyclopropanating agents when reacted with alkenes. Alkynes on…
Q: 15 C,H, 16 C,H,0
A: In a molecular formula all subscript tells us about atoms. So, atom count for C3H8= 11 C2H6O = 9
Q: Which of the following bonds has the most covalent property? C-Cl S-Cl H-Cl Mg-Cl Na-Cl
A: Covalent bond is formed between two atoms by mutual contribution and sharing of one or electrons.…
Q: QUES TION 1 WHICH COMPOUND CONTAINS COVALENT BONDS ? O BF2 O LiF 0 SnFz lO Na H. O All of these 1O…
A: The structure of the given compound are drawn below:
Q: 1) which is not included on the Em spectrum A) Uv B) radio waves C)sound waves.…
A: Hi, we are supposed to answer one question. To get the remaining questions solved please mention the…
Q: ste and smell) natural flavor based on its elementa tion. C 58.80%, H 9.87%, O 31.33% 0₁ H9.791.958…
A: If mass of compound is 100 g. Mass of C = 58.80 g Moles of C = 58.80/12 =4.90 mol Mass of H = 9.87 g…
Q: : ci 1.ci: : :Ci: CI :CI 1 Chlorine molecule Chlorine atom Figure 20-2 Chlorine atom What type of…
A: Here we are asked to identify the type of bond formed when two chlorine atoms comes close to make…
Q: Draw and name the following compounds. Na2[Cu(H2O)2Cl4] [Cr(C2H8N2)3]Cl3, how do you even start with…
A:
Q: For each compound, state whether its bonding is covalent, ionic, or a mixture of covalent and…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Rh Pd Ag Cd In Sn Sb Te | Xe Ir Pt Au Hg TI Pb Bi Po At Rn Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Pu Am Cm Bk…
A: Octet rule means atom with has 8 electrons in valence shell Carbon has 4 valence electron electrons.…
Q: MCQ 150: Bonding pairs of electrons in ammonia are A. 1 B. 2 C. 3 D. 4
A: A bond is formed by sharing 2 electrons between 2 atoms in a covalent bond and each bond comprises…
Q: -20°C Br H2SO4 HO HI РСС
A:
Q: Classify each compound as containing only ionic bonds, only covalent bonds, or containing both types…
A: Ionic bond is formed by the complete transfer of valence electron(s) between atoms. It is a type of…
Q: Determine the correct second resonance structure of thioformate ion? 0=c=S–H ..0 O -1 s=c=0-H H. :
A: Given structure of thioformate ion, HCOS-
Q: CH, 'Br ČH, Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and…
A:
Q: Which of the following contains only covalent bonds? H20 O Ne Cal2 O None of the above NaNO3
A:
Q: her ionic or covalent. (NO) Br4 aPO4
A: Ionic bonds are formed by the transfer of electrons from one atom to another atom Covalent bonds are…
Q: 4. Which compound contains both ionic and covalent bonds? a. PF3 b. KF c. HC;COOH d. MGSO4
A: Ionic compounds are those which are formed by the transference of electrons from one atom to…
Q: Z= Ö: 70. Explain what is wrong with the following Lewis structures, and then correct each one. a.…
A:
Q: Ampyra is a drug used to improve walking in patients w is shown. ート .C. C. H. N. H. How many bonding…
A: Bonding pairs of electrons are those electrons shared by the central atom and any atom to which it…
Q: Classify the bonds below as ionic, nonpolar covalent, or polar covalent. (EN: Cl 3.16; Mg 1.31; H…
A: We are to find whether given molecules has ionic or covalent bonds. Electronegativity : Cl = 3.16 Mg…
Q: MCQ 16: Group IV element that doesn't have strong covalent bonds is A. Carbon (Diamond) B. Tin (Sn)…
A: A covalent bond is formed by sharing of two electrons between two atoms. A covalent bond is formed…
Q: Q3/ Give the chemical structure form A-D. CH Lom HNO ATSO4) DH 2ONH (C) + > (A) Fe/HCI (B) less 5
A:
Q: nules? HH H H BH
A: The compounds given are,
Q: How many valence electrons are in Cl * O ^ - 1
A: The number of valance electrons can be calculated by drawing the lewis structure of the given ion.…
Q: Predict which ions are stable: (a) Br2- (b) C4- (c) Ca+ (d) Ar+ (e) Na+ (f) Cs+
A: The element which acquires noble gas electronic configuration are stable.
Q: Which compounds contain both ionic and covalent bonds? I. BaSO4 II. Ca(NO3): I only b. I and III…
A: Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when…
Q: Which bonds would be covalent bonds? C-Cl C-H Na-O Fe-Cl Zn-S N-O
A: ->Ionic bonds formed between metal and non-metal. ->Covalent bond formed between non-metals.
Q: 1) Based on position on the Periodic Table, what type of bond will be formed between Mg and I ?…
A: A bond between two atoms with the electronegativity difference of >2 is considered to be ionic…
Q: What type of charge do all compounds carry? O positive O varying O neutral O negative
A: A compound is made up of 2 or more than 2 same or different type of atom or ions.There are either…
Q: How are compounds with ionic and covalent bonds alike? A. Both conduct electricity when dissolved in…
A: Covalent compounds formed by sharing of electrons ionic compounds formed by transfer of electrons.
Please help me this question. Thank you!
Step by step
Solved in 3 steps with 1 images

- How is using thiamine as a catalyst rather than cyanide an application of greenchemistry? PROCEDURETo a six inch test tube was added thiamine hydrochloride (0.65 g), followed by water (2mL). The mixture was gently shaken until homogeneous, and then ethanol (7.5 mL)was added. Finally, sodium hydroxide (10%, 1.5 mL) was added and the mixture wasgently stirred to ensure complete mixing.Benzaldehyde (3.8 mL) was then added at room temperature in one portion with gentlestirring. The pH of the resulting reaction mixture was determined and adjusted to >10by dropwise addition of 10% sodium hydroxide. The reaction mixture was then partiallysubmerged in a 65 °C water bath2 and the progress of the reaction was periodicallymonitored by TLC analysis.Once TLC analysis indicated that the reaction was complete, the reaction mixture wascooled to approximately room temperature, and then chilled in an ice-water bath. Theresulting precipitate was collected by vacuum filtration and air-dried. The…Is the Cu detectable at a concentration of 0.10 mg/L in Flame-AAS? Please use data given.What will happen to the estimated vitamin C content of the peach sample if it contains other compounds with reducing property? Explain. https://www.chegg.com/homework-help/questions-and-answers/1-happen-estimated-vitamin-c-content-peach-sample-contains-compounds-reducing-property-exp-q77588726
- Nickel bis-dimethylglyoximate complex was deposited with a weight of (4.869g) with the filter paper from the base medium from adding (10ml) of hot DMG to a salt of nickel chloride at weight (0.78g), find the percentage of nickel ion. Note that the weight of the filter paper (1.30g). At.Wt.: Ni= 58.69 g/mol, C= 12 g/mol, O=16 g/mol, H=1 g/mol, N=14 g/molWhat are the dissociation reaction, including phases, of AICI3?kindly answer this problem and follow this format GIVEN REQUIRED SOLUTION and write regibly thank you so much For the adsorption of a substance A from aqueous solution by charcoal at25oC the freundlich constants are n=3.0 and k=0.50 for y in grams per gramand C in grams per liter. What weight of a W is adsorbed by 2 g of charcoalfrom 1 L of a solution containing originally 2 g of the substance?
- Using the term u of KSP experimental procedure the 6 p.m. As you add 5 ml of .004M AgNo to 5ml of .0025M K2CrO4. Is either of these reagents in excess? If so which one?Generally high temperature is favourable for chemisorption.’ Why?Fluoride Analysis by Ion Selective Electrode Measurable Potential and the Nernst Equation 1) List three factors which would contribure to departure from Nernstian behaviour. 2) Provide as many error sources in calibration of the ISE (up to five). (If u can't provide answer of both than please provide me answer of 2nd )
- If all other variables were kept constant, determine theeffect that the following errors would have on the calculatedpercent yield of the product. Would the yield be expected toincrease, decrease, or would there be no effect? Explainyour reasoning.– The product was insufficiently dried before weighing.– Some of the product was lost during the transfer fromthe Buchner funnel to the evaporating dish.– 7.5 mL of FeCl3 was added instead of 3.0 mL asoutlined in the procedure.– 4.587g of K2C2O4H2O was used instead of exactly4.000g .– The recrystallization step was skipped and theexperiment went straight to vacuum filtration.(Please give clear handwritten answer) 0.1 M HNO3 solution at 298K was electrolyzed in Hittorf cell using platinum electrodes. After electrolysis, 34.2 mg of copper was deposited in coulometer which in series with the Hittorf cell. 50 mL of HNO3 solution was withdrawn from anode compartment after electrolysis and found to have a concentration of 0.0821 M. [Atomic mass Cu = 63]. (i) Find the transference number of H + and NO3 −. (ii) If 50 mL HNO3 solution was run off from cathode compartment, what would its concentration be?At a pH of 10 what is the fraction of EDTA that is in the form Y4-? I got 0.30 but I believe that is wrong.





