Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter33: Automated Methods Of Analysis
Section: Chapter Questions
Problem 33.5QAP: Give the advantages and disadvantages of sequential injection analyzers compared to traditional flow...
Related questions
Question
Is the Cu detectable at a concentration of 0.10 mg/L in Flame-AAS? Please use data given.
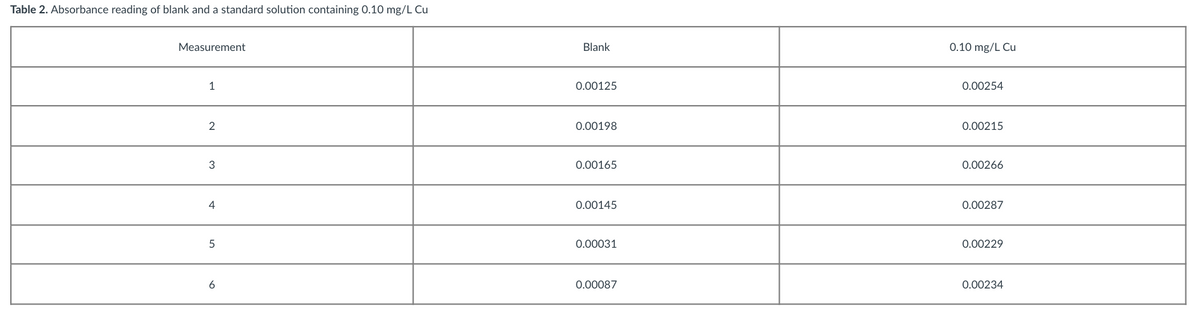
Transcribed Image Text:Table 2. Absorbance reading of blank and a standard solution containing 0.10 mg/L Cu
Measurement
Blank
0.10 mg/L Cu
1
0.00125
0.00254
2
0.00198
0.00215
3
0.00165
0.00266
4
0.00145
0.00287
5
0.00031
0.00229
6
0.00087
0.00234
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning