Identify the structure of the compound having the following molecular formula, IR, and 1H NMR spectra (integrals and multiplicity shown in the boxes: s-singlet, d-doublet, t-triplet, q-quartet, etc.). Label the spectra with the information (functional groups, number of protons, formula, Compound formula. C4H8O Questions to help you decide the structure. What is the
Identify the structure of the compound having the following molecular formula, IR, and 1H NMR spectra (integrals and multiplicity shown in the boxes: s-singlet, d-doublet, t-triplet, q-quartet, etc.). Label the spectra with the information (functional groups, number of protons, formula, Compound formula. C4H8O Questions to help you decide the structure. What is the
Chapter29: Mass Spectrometry
Section: Chapter Questions
Problem 29.5QAP
Related questions
Question
100%
Identify the structure of the compound having the following molecular formula, IR, and 1H NMR spectra (integrals and multiplicity shown in the boxes: s-singlet, d-doublet, t-triplet, q-quartet, etc.). Label the spectra with the information (functional groups, number of protons, formula,
Compound formula. C4H8O
Questions to help you decide the structure.
- What is the element of unsaturation of the molecular formula? Show the math you use:
- What are the functional groups present in this molecule? Show all of them below:
- Draw at least two possible structures that have the required element of unsaturation as well as the observed functional
- Based on the 1H NMR above, what molecular fragmentations do you see
- Draw your final decision of the structure below:
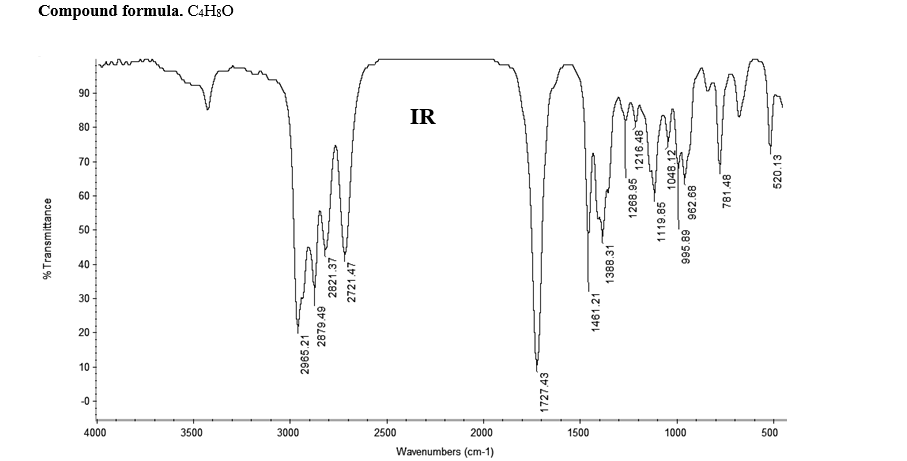
Transcribed Image Text:Compound formula. C4H3O
90
IR
80
70
60
50
40
30
20
10
-0-
4000
3500
3000
2500
2000
1500
1000
500
Wavenumbers (cm-1)
% Transmittance
2965.21
2879.49
2821.37
2721.47
1727.43
1461.21
1388.31
1268.95
1216.48 S
1119.85
1048.12
995.89
962.68
781.48
520.13
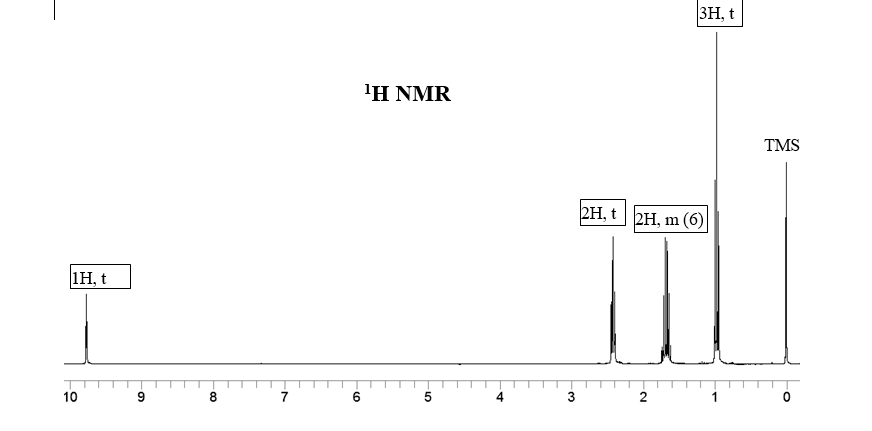
Transcribed Image Text:3H, t
1Η ΝMR
TMS
2H, t 2H, m (6)
1H, t
10
8.
7
4
LO
-CO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning