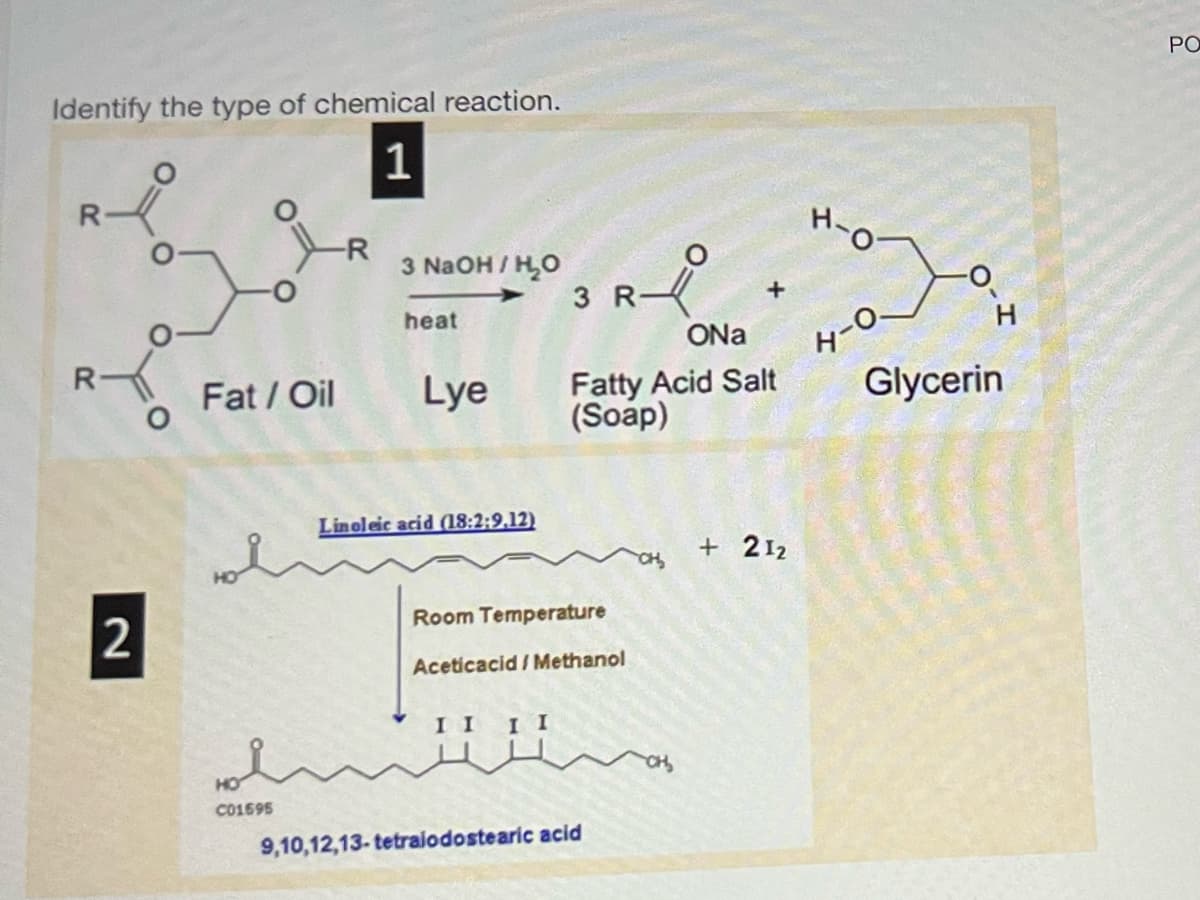
Identify the type of chemical reaction. 1 R R 2 Fat / Oil C01695 -R 3 NaOH/H₂O heat Lye Linoleic acid (18:2:9,12) II II 3 R Room Temperature Aceticacid/Methanol Å 9,10,12,13-tetralodostearic acid ONa Fatty Acid Salt (Soap) x +212 H-O H-O- H Glycerin
Q: Calculate for the initial pH, final pH, and buffer capacity if 0.1 mole of H+ is added to 1L of…
A:
Q: 6. How many mL of 6M HCI solution should be diluted to make 200 mL of a 1.5M solution?
A: Diluted solution can be prepared by diluting the concentrated sample of HCl.
Q: Fill in the blanks: In the peptide, LASALLEDASMA, (Write the full name of the amino acids) has the…
A: The amino acid composition of the given peptide is as follows:…
Q: Which of the following statements about aromatic amino acids is correct? (a) All are strongly…
A: Organic substances known as amino acids include both amino and carboxylic acid functional groups.…
Q: 14. Vitamin P: functional groups, mechanism of action, biological role, sources, daily requirement,…
A: Previously, the term "vitamin P" was applied to a class of plant chemicals known as flavonoids.…
Q: 1. How does the resin facilitate purification in glutathione S-tranferase (GST) affinity? 2.…
A: In Biochemistry, affinity chromatography is a technique used for the purification of protein based…
Q: Using an arrow, draw the site of cleavage for the following peptides that are reacted by: Pepsin…
A: Site specific proteases are enzymes that cleaves polypeptide chains only at specific points. Trypsin…
Q: Qualitative Color Reactions Color Reaction Biuret Ninhydrin Xanthoproteic Millon's Intact Protein…
A: Hi! Thank you for the question. We are authorized to answer four subparts at a time, since you have…
Q: 15. A substrate binds to enzyme active site. a) List the roles of serine, histidine and aspartic…
A: The enzyme has serine, histidine and aspartate in the active site. Serine proteases are protein…
Q: Which of the following statements concerning glycolysis is correct? O The pathway for the process is…
A: The first process in breaking down glucose to release energy for cellular metabolism is called…
Q: Casein is a biological polymer—what are the monomers of this polymer? (General term)
A: Polymers are high molecular weight compounds made up of smaller molecules linked in a chain-like…
Q: Suppose that the thermal denaturation experiment on the protein with the Met to Lys mutation took…
A: pKa is the pH at which a weak acid is 50% dissociated. Changes in pH affect the ionisation of…
Q: This question is about "ANSERINE" not just "SERINE
A: Anserine is a dipeptide composed of beta alanine and methyl histidine. It is found in brain of birds…
Q: Chemistry help
A: pI (isoelectric point) is the PH at which a molecule carries no net charge. Isoelectric focusing is…
Q: 1.) How do enzymes affect the proteins that we ate? 2.) How can co-enzymes affect enzyme activity?
A: Enzymes are bio catalysts that work to catalyse a biochemical reaction by decreasing its activation…
Q: Which of the following statements concerning enzymes is TRUE? a. Enzymes can increase the…
A: Enzymes are biological catalysts that enhance the rate of biochemical reactions. The enzymes are…
Q: Q4.3- For each of the five major protein types in eukaryotic cells 1) provide a specific example of…
A: Proteins can be classified into different types based on their function, chemical nature,…
Q: A peptide has the following amino acid composition: 2 Met, 2 Phe, 2 Glu, 1 Arg, 1 Lys, 1 Val, 1 Leu,…
A: Recall that: Amino acid sequences are written with N-terminal amino acid on the left and C-terminal…
Q: The side chain of which of the following amino acids is most hydrophilic a. Phe b. Val c. Leu d.…
A: The hydrophobicity or hydrophilicity of the amino acids in a protein is quantitatively analysed…
Q: Assumed that you have isolated the protein cytochrome C from a cell lysate. Discuss the advantages…
A: Cytochrome C Cytochrome C is a protein that is loosely bound to the inner membrane of mitochondria…
Q: diabetic patient has a sodium concentration of 141 mmol/L, a glucose concentration of 742 mg/dL and…
A: Osmosis is the movement of water across the semipermeable membrane driven by the difference in…
Q: Choose A if the statement is CORRECT B if the statement is WRONG This amino acid is/has: 6. 7. 8. 9.…
A: Proteins are constituted of twenty naturally occurring amino acids. The amino acids are classified…
Q: Algae are being studied as a source of lipids to be used as source of biodiesel as a liquid fuel.…
A: Given that algae uses H2O, NH3, CO2 in the presence of light to produce a compound CH1.8O0.5N0.2 and…
Q: Select the correct response:
A: Tyrosine gives rise to catecholamines which includes dopamine, norepinephrine, and epinephrine.
Q: In gluconeogenesis, how is glucose-6-phosphate converted to glucose? is converted to glucose by…
A: Gluconeogenesis is a metabolic process that converts non-carbohydrate carbon substrates such…
Q: Peptide mass fingerprinting can be used to identify proteins. It is a method of: a.…
A: The proteins can be identified my various methods such as NMR, X-ray crystallography, protein…
Q: Please answer fast What types of interactions likely promote the dimerization of a protein?
A: Proteins are polymers of amino acids that are joined together through peptide bonds. A peptide is a…
Q: Your supervisor asks to make 350 mL of a 0.75% (w/v) starch solution using a 5% (w/v) stock…
A: Given Values: Concentration of the starch stock solution = 5 % The volume of the final starch…
Q: What does it mean if a compound is said to have “high phosphoryl transfer potential”?
A: ATP is known as the energy currency of the cell. ATPs are required by the cell for various reactions…
Q: referred to feedback inhibition as “Goldilocks regulation”. Describe how feedback inhibition keeps…
A: Biochemical pathways and reactions are occurring constantly in cells. These pathways convert the…
Q: 7. Which of the following statements regarding the regulation of glycogen metabolism is false? A.…
A: In human body, when there is excess of glucose in blood, insulin acts on the hepatocytes and muscle…
Q: what are the chemical methods for glucose determination and their clinical significance?
A: Glucose is the most important carbohydrate fuel of the body. The normal concentration of glucose in…
Q: So when drugs are bound to plasma proteins, the effects are NOT intensified?
A: Drugs are chemical substances that have the capability to improve physical and mental illness.…
Q: A one-to-one protein (P)-ligand (L) complexation (P + L PL) has a equilibrium constant (Kd) value…
A: ∆Cp is the heat capacity at constant pressure. ∆S is change in entropy, degree of randomness. At…
Q: Which of the following statements concerning SDS in SDS-PAGE is INCORRECT? a. SDS is a negatively…
A: SDS-PAGE is an electrophoresis technique used to separate protein molecules. Protein molecules can…
Q: In order to study protein structures and functions, many protein techniques have been developed for…
A: Proteins are composed of twenty standard amino acids attached together via peptide bonds. These…
Q: The side chain of cysteine contains: OA) a hydroxyl group OB) an amine group C) a carboxyl group OD)…
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: In the alpha-helices, which of the following statement is NOT true? a. Backbone carbonyl…
A: Proteins are composed of amino acids that are attached together via peptide bonds. The linear chain…
Q: 4) For various amino acid pairs (for example: F to A, E to R, D to N, V to L, S to W), ask yourself:…
A: Amino acids are biomolecules that have an amino group and a carboxyl group linked to the same carbon…
Q: 2. Biuret Test Color of 3% CuSO4 solution: Sample casein alanine albumin gelatin distilled water 3.…
A: Proteins are large molecules made up of amino acid residues linked via a peptide bond. Amino Acids…
Q: Which of the following substances can deliver electrons to the ETC to help pump out H+ across the…
A: Electron transport chain is a chain of electron carriers that transfer electrons to molecular…
Q: A. Will decreasing the amount of energy needed to make an exergonic reaction occur cause the…
A: The thermodynamics of a chemical reaction focuses on the energetics of the products and the…
Q: 1. How does the resin facilitate purification in glutathione S-tranferase (GST) affinity? 2.…
A: Glutathione S-tranferase (GST) is a large family of proteins that are involved in the detoxification…
Q: Formation of a peptide bond is a. dehydration reaction b. reducing reaction c.…
A: Proteins are composed of twenty naturally occurring amino acid. The amino acids in a protein are…
Q: Why can plant material be substituted? Plants contain high amounts of saturated fats. Certain plants…
A: Introduction : Linoleate - A linoleate is a salt or ester of linoleic acid(which is an organic…
Q: what is the concetration of a lysozyme solution with an absorbance of 0.720 measured at 280 nm(…
A: Measuring the concentration of a substance in solution on the basis of its absorbance forms the…
Q: Which of the following single-stranded DNA molecules are likely to pair with each other to form…
A: DNA are polymers of nucleotides. A nucleotide consists of a nitrogenous base(A, T, G, C) attached to…
Q: Lactate dehydrogenase isoforms contain "H" and "M" subunits. In total, how many subunits are found…
A: - A dehydrogenase is an enzyme that transfers a hydride from one molecule to another. - Enzyme…
Q: Sketch on one reaction rate vs. substrate concentration graph & sketch on one Lineweaver-Burk type…
A: LB plot is double reciprocal plot which gives Km and Vmax values as x intercept and y intercept…
Q: Identify basic structure of the 8 types of membrane proteins shown below Understand how primary…
A: According to the fluid mosaic model of the plasma membrane is a phospholipid bilayer that has…
Step by step
Solved in 2 steps

- Identify the acid on the left and its conjugate base on the right in the following equations:(a) HOCl + H2O ↔ H3O+ + OCl-(b) HONH2 + H2O ↔ HONH3+ + OH-(c) NH4+ + H2O ↔ NH3 + H3O+(d) 2HCO3-2 ↔ H2CO3 + CO3-2 (e) PO4-3 + H2PO4- ↔ 2HPO4-2Define the following terms:a. alditolb. enediolc. epimerizationd. acetale. ketalPlease balance the following half-based reactions: SbH3 → Sb (acidic solution) BrO3- → Br2 (acidic solution) Cl- → ClO2- (basic solution) (PLEASE DONT USE H+ ION INSTEAD SUB H30+ )
- Give full explanation The physcian orders ascorbic acid 0.25mg IM for your patient admitted with an alcohol problem. You have ascorbic acid 500mg/mL. How many milliliters will you administer?Which of the carbohydrate tests (Molisch, Seliwanoff, Benedict, Barfoed, Bial, Iodine Tests) would be positive for maltose? Include description of the color change. Explain in 2-3 sentencesTerpin hydrate contains 1 gr of codeine per fluidounce. How much codeine (in mg) should be added to 8 fluidounces such that each teaspoonful contains 20 mg of codeine? Select: a 946.2 mg b. 427.8 mg c. 518.4 mg d. 6.55 mg. Please type answer note write by hend.
- Match the following descriptions with the correct lipid-based compounds:Define the following terms in 2-3 sentences.a Glycoside b.Glycone c.Aglycone d.Bufadienolide e.CyclopentanoperhydropenanthreneRank the following in order of increasing solubility in water: a triacylglycerol, a diacylglycerol, and a monoacylglycerol, all containing only palmitic acid.



