Identify whether the following fatty acids are saturated or unsaturated. a. CH3(CH2)5CH=CH(CH2)7COOH b. CH3(CH2)7CH-CH(CH2)7COOH c. CH3(CH2) 10COOH d. CH3(CH2) 16COOH
Q: Is the claim "Our product contains no cholesterol" in coconut oils true? Why or why not?
A: Oils is a commonly usable chemical for various uses.Oil is nonpolar chemical substance which is…
Q: Discuss how P. fluorescens can be modified to make it a more effective biocontrol agent against the…
A: Fluorescent Pseudomonads are part of the Plant Growth Promoting Rhizobacteria (PGPR) family of…
Q: 2. Calculate AG for the exchange of 3 Na* by 2 K* by Na*-K* ATPase (in kJ per mole of ATP) under the…
A: Gibbs free energy change for the exchange of 3Na and 2 K by Na-K ATPase It is a thermodynamic…
Q: Enumerate and discuss three disorders that could result from the absence of enzymes
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that…
Q: On a per-carbon basis, where does the largest amount of biologically available energy in…
A: There are two major types of dietary lipids: plant-derived triacylglycerol and animal-derived…
Q: 4. Which of the following statements about the citric acid cycle is INCORRECT? A. It is a cyclic…
A: Metabolic pathways are a series of process which includes chemical reactions occurring in a cell.…
Q: Monosaccharides can be obtained not only from dietary carbohydrates but other major sources include…
A: Monosaccharides is a one of the type of carbohydrate. Monosaccharides are also called as simple…
Q: If a base, such as sodium hydroxide (NaOH) were added to milk, would the protein precipitate? Why or…
A: Milk is a great source of vitamins and minerals, especially calcium. Milk plays a crucial role in…
Q: CH2OH OH HO, CH2OH OH OH OH ÓH Enatiomers Diastereomers O Epimers Anomers
A: When two monosaccharides differ from each other in their configuration around a single specific…
Q: during digestion the pH in the stomach is 2. what does this indicate about the optimum pH of pepsin…
A: Digestion is the process of conversion of complex food substances into simple absorbable forms. The…
Q: Which of the following is the complementary strand of a DNA strand made up of 32% cytosine? A.…
A: Two strands of DNA are complementary to each other , if we know the sequence of one strand, we could…
Q: 3. Recombinant protein is produced by a genetically engineered strain of Escherichia coli during…
A: Disulfide-bonded proteins primarily mature in the oxidative environment of the eukaryotic…
Q: What is phenylketonuria? Discuss its occurrence, symptoms if any, treatments if there are, and any…
A: The pattern of inheritance of a condition caused by a recessive faulty gene copy located on an…
Q: Q1. Many of the components for lipid synthesis need to be "activated", which happens by a variety of…
A: Lipids are non-polar biomolecules, which act as the major components of the plasma membrane, energy…
Q: During plasmid extraction, after the addition of acetate-conta in ing neutralization buffer the…
A: Plasmids are extrachromosomal DNA in the bacterial cells. They are double stranded circular DNA,…
Q: What are the respective associated substances & processes? reduction regeneration RuBP RUBISCO NADPH…
A: Photosynthesis occurs in basically two steps and they are light reaction and dark reaction. In the…
Q: Each proton that moves across the membrane releases about 14 kJ/mol of energy. Given that ATP…
A: The ATP synthase in the inner mitochondrial membrane uses the flow of protons into the mitochondrial…
Q: Translation in eukaryotes and prokaryotes are similar and yet different. From a therapeutic…
A: Translation is the process where mRNA transcript of a particular gene is decoded to give rise to a…
Q: Table Q1(a) shows typical values for the intracellular and extracellular concentrations of the major…
A: All cells have an electrical potential difference or membrane potential across their plasma…
Q: Biological value
A: Protein efficiency ratio: It is defined as a ratio of weight gain of a subject to protein intake.…
Q: The following bond makes bovine pancreatic trypsin inhibitor as one of the stable proteins.…
A: Introduction: Bovine pancreatic trypsin inhibitor (BPTI) binds to trypsin and prevents peptide…
Q: The apo form of an enzyme: (select all that applies) Group of answer choices Is the cofactor-free…
A: Enzymes are biological catalysts that help living organisms speed up chemical reactions. Enzymes can…
Q: II. A 45-ycar-old man complained of rapid weight loss, tachycardia, increased sweating, occasional…
A: A pheochromocytoma is a tumor in the adrenal gland and causes hypersecretion of hormones from this…
Q: - What investigation to differentiate between anemia due to vitamin B12 or folic acid deficiency? is…
A: Vitamin B 12 , is a cyanacobalamin. It is usually produced by the bacteria. There are wide variety…
Q: Do "Enzymatic Browning reaction is intensified with the presence of oxygen?.
A: Introduction Enzymatic browning is a chemical process that we can see in fruits, vegetables etc. .…
Q: The citric acid cycle occurs in the O smooth endoplasmic reticulum. mitochondrial matrix. nucleus.…
A: The TCA cycle, also known as the Krebs cycle, takes place in the mitochondria and generates a…
Q: Movement of water is controlled by which factor?
A: Movement of water occurs through the xylem cells of plants to reach all parts of plant body . As…
Q: Scenario IV. Electron Transport Chain Total Number of ATP Obtained from one Pyruvate Molecule Number…
A: Glucose or carbohydrates or sugars are oxidized through the process of glycolysis. Glycolysis occurs…
Q: Enumerate the 3 Coronavirus strains that caused a large-scale outbreak in humans in the past 2…
A: Coronaviruse is basically a group of RNA viruses that can causes the serious complication to birds…
Q: 30. assicuatuib if 2alpha and 2 beta chains to form adult hemoglobin
A: HbA2 is a gene that in encodes for the alpha globin chain of haemoglobin in humans.
Q: Complete the sentences to explain how the H Match the words in the left column to the appropriate…
A: The electron transport chain is a series of proteins and organic molecules found in the…
Q: Explain the importance of carbohydrates in your body
A: Carbohydrates are among the most important biomolecules needed for the sustenance of life. They are…
Q: Describe the common strategy (steps) for protein sequencing, starting with a biological sample…
A: Proteins are one of the 4 major biomacromolecules. Proteins are the most abundant of the 4…
Q: Please answer both questions (a and b). Benzodiazepines such as diazepam and clonazepam act on…
A: Benzodiazepines are class of psychoactive drugs whose core chemical structure is the fusion of a…
Q: During exercise when the body lacks an adequate supply of oxygen to support energy production, the…
A: When a person does workout, then the body has a tendency to lack a very high amount of oxygen…
Q: Which of the following best describes the mechanism of action of a cardiac glycoside? A)…
A: Cardiac glycosides are the compounds that are important for enhancing the force of heart to increase…
Q: Which post-transcriptional modification is NOT shared by all eukaryotic RNAs? a. Modifying bases…
A: Posttranscriptional modification is the process by which the RNA primary transcript is chemically…
Q: What amino acid side chains can be modified by methyl groups? What is unusual about methyl group…
A: What amino acid side chains can be modified by methyl groups? Answer: Methylation is a process of…
Q: Which is true about eukaryotic cDNA? Choose all that apply. a. it is constructed from mRNA that…
A: Complementary DNA (cDNA) is made from messenger RNA in the laboratory for various purposes. Because…
Q: In terms of structure, what is the commonality between aldolase (glycolyssis) and transaldolase…
A: Aldolase is an enzyme that is involved in glycolysis and it converts fructose-1,6-bisphosphate into…
Q: A) Describe the glycosidic bond (using standard convention) indicated by “Arrow a.” B) Draw the…
A: Sterols are lipids containing a steroid nucleus and an alkyl chain. The steroid nucleus is a 4 ring…
Q: The structure of the dipeptide Gly-Asn is given by The structures of the amino acids Gly and Asn are…
A: Glycine (Gly - 3 letter code ) and Asparagine (Asn) are amino acids. Amino acids are the building…
Q: Angiotensin-converting enzyme inhibitors (ACE inhibitors) are used to treat high blood pressure.…
A: ACE is an enzyme in the Renin-Angiotensin-Aldosterone pathway (RAAS). ACE inhibitors (ACE I)…
Q: Complete the table below regarding the different laboratory tests done for Carbohydrates. Only for…
A: There are different types of tests to check the presence of carbohydrates in different chemical…
Q: To set up a standard curve you would have had to set up a dilution series. Draw up a table showing…
A: Serial dilution is the process of converting a dense solution towards a more useable concentration…
Q: deoxynucleosides?
A: Nucleosides and deoxynucleosides are endogenous metabolites excreted from RNA turnover and DNA…
Q: Which of the following describes a Z-DNA helix? a. It is inhibited by methylation of bases b. It is…
A: DNA is a polymer made up of two polynucleotide chains that coil around each other to form a double…
Q: Which isomer- (R)-arginine or (S)-arginine is D-arginine? O (R)-arginine O (S)-arginine Submit…
A: Isomers are molecules with same molecular formula and different arrangement of atoms. Enantiomers…
Q: HN Histidine NH₂ OH HN + H |+ H-NH H
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that…
Q: Some enzymes require additional ...........................to catalyze reactions effectively. These…
A: Introduction: Enzymes are large biological molecules important for thousands of metabolic processes…
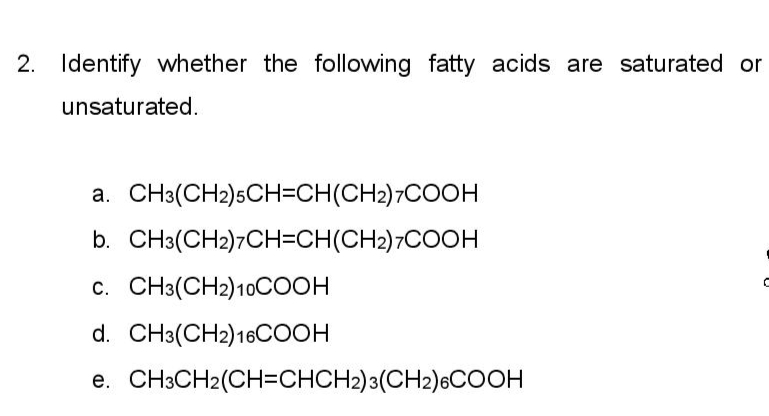
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- 1. Which one is NOT a property of glycerol? a. "backbone" in the simple lipid structure b. undergoes condensation with three molecules of fatty acids c. its OH groups reacts with the fatty functional groups of fatty acids d. it is a water-soluble ester 2. Which one has to bends in the structure? a. palmitic acid b. oleic acid c. linoleic acid d. myristic acid3. One of the triacylglycerols found in corn oil contains palmitic acid, linoleic acid, and linolenic acid. During the production of spreadable margarine, all of the double bonds in this triacylglycerol are converted from cis to trans isomers. Linoleic acid: CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOHLinolenic acid: CH3CH2CH=CHCH2CH=CHCH2CH=CH(CH2)7COOH Palmitic acid: CH3(CH2)14COOH a. Identify the unsaturated fatty acid(s) listed above and provide the associated omega designation. b. Draw an accurate representation of the structure of the triacylglycerol present in spreadable margarine. Circle all ester bonds in the structure you have drawn.1) Draw the structures and provide the systematic names of the following fatty acids (all double bonds are in cis- configuration).a) 24:0b) 18:3(Δ6,9,12)c) 20:3(Δ8,11,14)2) Define and briefly explain the process of saponification.3) Commercial vegetable oils are subjected to partial hydrogenation processing before they are made available on the market;a) Explain the importance of this partial hydrogenation on cooking oil.b) Give two reasons why un-hydrogenated vegetable oil is not suitable for cooking.c) What undesirable effect does partial hydrogenation cause?4) Platelet-activating factor and Prostacyclin are lipids that play roles affecting coagulation.a) Differentiate the roles of each aforementioned lipid with reference to coagulation. b) What specific type of lipids does each molecule belong to?5) Draw the structures of lecithin and cephalin, outlining the general features. 6) Explain the chemical importance of having 2-deoxy-D-ribose in DNA over D-ribose.7) Draw a schematic…
- 4. What is the role of decarboxylation in fatty acid synthesis? Describe another process discussed in this Biochemistry I course that uses decarboxylation for the same purpose.1. Which of the following best describes glycogen? A. a fibrous protein that helps in maintaining cellular structure B. a storage polysaccharide found in plants C. has alpha 1-6 glyosidic bonds. D. is formed by the hydrolysis of glucose molecules. 2. Which part of an amino acid gives it its unique identity? A. the long carbon-hydrogen tails of the molecule B. the carboxyl and amino groups C. the components of its side chain D. the glycerol molecule that forms the backbone of the amino acid1.Why do fat molecules can pack closely together? a. "bends" b. polarity c. length d. linearity 2. Which one is NOT a property of glycerol? a. "backbone" in the simple lipid structure b. undergoes condensation with three molecules of fatty acids c. its OH groups reacts with the fatty functional groups of fatty acids d. it is a water-soluble ester 3. Which one has to bends in the structure? a. palmitic acid b. oleic acid c. linoleic acid d. myristic acid
- 1. What are the differences between unsaturated and saturated fatty acids? 2. What is Biological Value (protein)? 3. What is Chemical Score (protein)? 4. What temperature is your beverage optimal for rehydration?4. Draw the condensed structural formula for the triacylglycerol made from 3 saturated fatty acids that are 24 carbons long Using diagram from 1 as the reactant, draw the full equation (using condensed structural formulas) for this triacylglycerol undergoing saponification, using NaOH2. Draw the condensed structural formula for the triacylglycerol made from 3 saturated fatty acids that are 18 carbons long. Using your diagram from #2 as the reactant, draw the full equation (using condensed structural formulas) for this triacylglycerol undergoing enzyme hydrolysis, using lipase.
- 1. a. Explain why the melting point of palmitic acid (16 carbons, no double bonds) is slightly lower thanthat of stearic acid (18 carbons, no double bonds). Explain why the melting point of oleic acid (18carbons, one double bond) is lower than that of stearic acid b. A mixture of lipids containing phosphatidic acid, cholesterol, testosterone, phosphatidylserine, andphosphatidylethanolamine was applied to a hydrophobic interaction chromatography column. Thecolumn was washed with a high salt buffer, and the lipids were then eluted with decreasing saltconcentrations. In what order would the lipids be eluted from the column? Explain your answer.1. What is the possible identity of the AA? a. Aspartic acid b. Lysine c. Alanine d. Proline 2. What is the isoelectric point of AA? 3. What is the pKa responding to the dissociation of the alpha-carboxylic group?The correct systematic name for the fatty acid below is: a) 3 octadecatrienoic acid b) 9 octadecatrienoic acid c) 3,6,9 octadecatrienoic acid d) 9,12,15 octadecatrienoic acid e) None of the above





