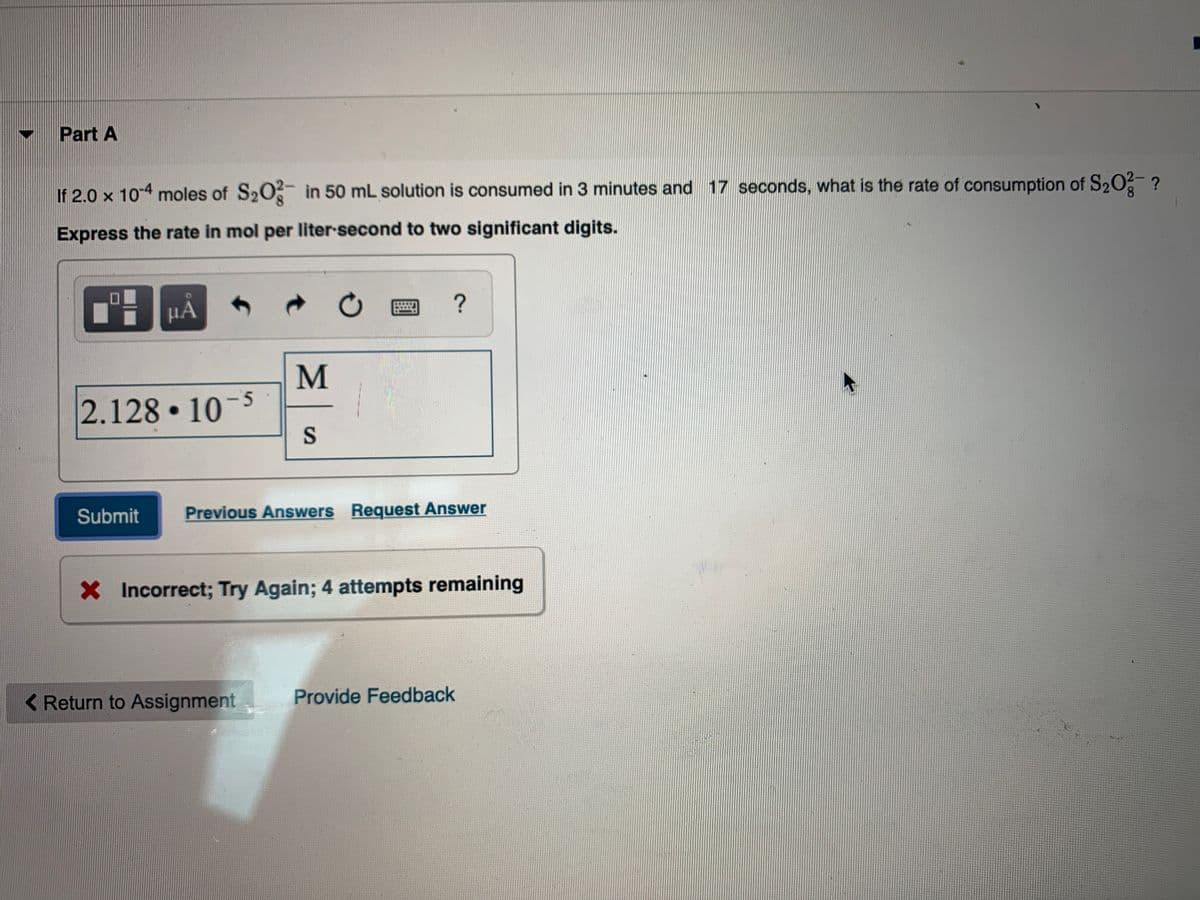
If 2.0 x 104 moles of S20- in 50 mL solution is consumed in 3 minutes and 17 seconds, what is the rate of consumption of S20 ? Express the rate in mol per liter second to two significant digits.
Q: Q25: There are so many microbes in a single mL of culture, it is very difficult to perform one…
A: Given: 2 serial dilutions are performed, each with a value of 1 (dilution)
Q: Sugar decomposes in water at a rate proportional to the amount still unchanged. There were 70 lbs of…
A: Answer: In the problem statement it is said that, 3 hours is the time required for the sugar…
Q: A chemist adds 130.0 mL of a 9.85M silver perchlorate (AgClo,) solution to a reaction flask.…
A:
Q: -1 A geochemist measures the concentration of salt dissolved in Lake Parsons and finds a…
A: Concentration of salt in Lake Parsons was found to be 25.6 g L-1. The average concentration of salts…
Q: 22. Plot a properly labelled graph of Concentration vs. Time in the space provided. Draw a curve to…
A: Given data : Time (s) Concentration(mol/L) 0 1.90 1 1.40 2 1.10 3 0.90 4 0.80 5 0.75…
Q: Determine the volume of concentrated (14.7 M) phosphoric acid is needed to prepare 25.0 L of 3.0 M…
A: Molarity is defined as the ratio of the number of moles of solute to the volume of solution in…
Q: Consider a block of wood having a volume of 125 cm³. If the block of wood burns at a rate of 15…
A:
Q: Chemistry Due to E. coli outbreak in spinach, USDA recommends that we boil spinach in water before…
A:
Q: A group of students were asked to determine the time is took for a single drop of methanol (CHA) to…
A: Option D. 15.0% is correct. Given data set for time take to the evaporation of first drop of…
Q: 2) A student investigated the neutralisation of dilute hydrochloric acid, using an excess of calcium…
A:
Q: * :The concentration of ( mgO) in bone is a-10.2 b-0.2 c-0.3 d-4 :We can write law the work as…
A:
Q: C. PREDICT THE PRODUCTS AND IDENTIFY THE TYPE OF CHEMICAL REACTION. 1. H,0 2. H, + O, → 3. KBr + Cl,…
A:
Q: What specific value from the graph is needed to calculate the molar concentration of the NaOH(aq)?
A: Use the titration formula. If the titrant and analyte have a 1:1 mole ratio, the formula is molarity…
Q: In an experiment, hydrochloric acid reacted with different volumes of sodium thiosulfate in water A…
A: The reaction between HCl and sodium thiosulfate produces a yellow colored precipitate of sulfur.
Q: Calculate initial concentrations added to the test tube, the dilution factor must be taken into…
A:
Q: 2. Al + 3 e 5 Al E° = - 1.67 Brz + 2 e 2 Br E' = + 1.09 E' = + 0.50 V E° = + 0.141 V 3. MnO, + 2 H2O…
A: The spontaneous redox reactions are to be drawn from the given information. A redox reaction is…
Q: For Questions 20-24 consider the following scenario: A 0.8390 g of an unknown sample containing…
A: Your answer is correct. The explanation is given here. The sample containing chloride ion reacts…
Q: [OH-] = 1.0 × 10-7 Express the concentration to two significant figures and include the appropriate…
A: Calculate hydronium ion concentration of the folowing---
Q: O Cr O H,O O Mg Figure I Figure 2 Two samples of Mg(s) of equal mass were placed in equal amounts of…
A: Option C should be the correct answer. As per the given data we are having Mg of equal mass which is…
Q: OH 1.PBO 2. Mg OH
A:
Q: 4. Explain the laboratory rules for waste disposal.
A: Given question demands the all type of waste disposal rules used in the laboratory. We know that, in…
Q: 2. For the reaction, 5 3+ it was found out that at a particular instant, Mn²+ (a 2+ (aq) + MnO4 (aq)…
A:
Q: OMe + BuzSnBr OMe BuSnH "Br AIBN
A: Tributyl tin hydride (Bu3SnH) : Organotin hydrides are very good radical reducing agent , it is…
Q: Sodium phosphate dissociates when it dissolves in water. Na,PO,(aq) - 3 Na+(aq) + PO,3-(aq) Given…
A: As per our guidelines we can only solve first three sub-parts of a question. Please resubmit the…
Q: Federal regulations set an upper limit of 50 parts per million(ppm) of NH3 in the air in a work…
A: (a) The volume of HCl solution is 1.0 × 102 mL which is equal to 0.100 L and the volume of NaOH…
Q: What is the practical importance of extraction in relation to chemistry especially when it comes to…
A: Extraction is important for Isolating important materials from plants .
Q: 27. A seguir é mostrado o espectro de RMN de um éster com fórmula C,H,O,. O espectro no…
A:
Q: 2. A student doing this experiment was puzzled by the fact that while each of the individual…
A: Hazardous Solution contains toxic ,hazardous chemicals that are harmful for nature.
Q: 4. Add 10 ml of 0.85% lactic acid. During normal metabolism the body produces small amounts of…
A: Given: 10 mL of 0.85% lactic acid. Metabolism of the body produces a small amount of lactic acid,…
Q: A certain chemical dissolves in water at a rate proportional to the product of the amount undissoved…
A: Total mass of saturated solution = 100 gm Mass of solute that can be dissolved in solution = 50 gm
Q: Which set is the correct coefficients for the following equations:SbCl3 + Na2S ------ Sb2S3 + NaCl
A:
Q: Give the overall balanced equation. Assume that all concentrations are 1.0M and that all partial…
A: In a balanced chemical reaction atoms of each element on the reactant side and product side must be…
Q: Suppose a vessel contains NH, at a concentration of 0.920M. Calculate the concentration of NH, in…
A: Initial concentration of NH3= 0.920M Concentration of NH3 after 170 seconds = ? Order of reaction =…
Q: Classify the following reactions whether they are homogeneous or heterogeneous: 1- PbCl2 (s) 2NH3…
A: In a reaction, reactant and product both are present. There are many types of reaction…
Q: Sodium carbonate dissociates when it dissolves in water as follows: If the concentration of…
A:
Q: Which set is the correct coefficients for the following equations:B5H9 + O2 -------------- B2 O3 +…
A: Balance the above reaction by hit and trial method. 1st balance B and Hydrogen by multiplying…
Q: OH 1. PBr 2. Mg 1) Bra, light 2) KOC(CH₂) 1) SOCT, 2) LEAID OH
A: #(c): Alcohol when reacts with PBr3 forms alkyl bromide which on reaction with Mg forms Grignard…
Q: Balance the following equations and express the rate of the following reactions in terms of the…
A: Balance equation : Balance equation are those equation in which atom on both sides are equal. Rate :…
Q: A chemist adds 415.0 mL of a 1.78M potassium iodide (KI) solution to a reaction flask. Calculate the…
A: The question deals with the concept of solution preparation. we have to calculate the number of…
Q: Compare: capacity factor, k' I 0.18 0.16 0.14 0.12 0.1 11 0.08 0.06 0.04 0.02 0 the quantity in I is…
A:
Q: A series of BOD tests was run at three different dilutions. The results are shown on the table. What…
A: The answer is attached below-
Q: 3. Now that you know the physical state of each of the reactants and products in this chemical…
A: The substances take part in a reaction are called reactants. The substances formed after a…
Q: Given the line notation: Where X2+ +e X+ Y* +3e Y Pr+ +4e → Pt E =0.5V E =-155V E = 1.20 V Calculate…
A:
Q: After an injection of 5 mg of Drug Y, the concentration of the drug in the bloodstream drops at the…
A: Initial concentration = [ A0 ] = 5 mg Final concentration = [ A ] = 1 mg Rate constant = 0.020…
Q: Give one event that carbon monoxide poisoning may happen
A: Carbon monoxide is the odourless and tasteless gas and it is toxic to animals and humans. Carbon…
Q: 1. Suppose that a home furnace burns propane gas, C;Hy. The air intake of this furnace (the part of…
A: The incomplete combustion reaction of propane gas is as follows: 2C3H8(g)+7O2(g) →…
Q: 2. Fifty cubic meters of water containing 28mg/L solids passes the Gallo River in one minute. There…
A:
Q: Human saliva contains enzymes that start the process of digestion. Human saliva typically has a…
A:
Q: 7) A chemical company developsa new processthatresults in some waste. Their internal studies show…
A: Any chemical substance that causes malignant growth is known as a cancer-causing agent. However,…
Q: Will a reaction take place? Use the exam data sheet. Complete & balance; Classify. C,H+ 0, à…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Please answer super fast and answer all questions and show calculations For the image attached For 1. a Mass of metal: Trial 1 is 35.0228 g Trial 2 is 35.0915 g Trial 3 is 34.0821 g Mass of water: Trial 1 is 20.0177 g Trial 2 is 20.0250 g Trial 3 is 20.0168 g For delta t of water: Trial 1 is 15.5 C Trial 2 is 15.7 C Trial 3 is 15.1 C For delta t of metal Trial 1 is 80.1 C Trial 2 is 80.2 C Trial 3 is 79.5 C For B my calculated Specific heat is: Trial 1 is 0.462 Trial 2 is 0.467 Trial 3 is 0.466Please answer fast it’s very important and urgent I say very urgent so please answer super super fast please For the image attached For 1. a Mass of metal: Trial 1 is 35.0228 g Trial 2 is 35.0915 g Trial 3 is 34.0821 g Mass of water: Trial 1 is 20.0177 g Trial 2 is 20.0250 g Trial 3 is 20.0168 g For delta t of water: Trial 1 is 15.5 C Trial 2 is 15.7 C Trial 3 is 15.1 C For delta t of metal Trial 1 is 80.1 C Trial 2 is 80.2 C Trial 3 is 79.5 C For B my calculated Specific heat is: Trial 1 is 0.462 Trial 2 is 0.467 Trial 3 is 0.466Design a procedure on how you will prepare 1.25 M table sugar (C12H22O11)solution and 1.25 M table salt (NaCl) solution diluted to 1 L water. Perform three trialseach set-up and get the average. Write/ encode your output in short bond paper withthe following parts:Title____________________I. Objectives:II. Materials:III. Procedure (including pictures/diagrams):IV. Data:V. Generalization: (Note: Perform this activity on a separate table/ area at home to ensure accuracyand precision of data. Be careful on handling the solution to avoid spillage.)
- 100 g of soil is leached with a strong solution of Calcium chloride such that all the exchange sites are occupied by Ca2+. The soil is subsequently leached again with a strong solution of magnesium chloride. It is determined that the resulting 100 mL leachate contains 5000 mg of Ca2+. What is the soil CEC (cmolc/kg)? The atomic wt. of Ca is 40 g/mol.please answer in word don't image upload thank you.Show ALL calculations pls. Don't reject if you dont know how to answer I need this. a. What are the average values for vph and vmo? Include calculation of net volume and errorsb. What is/are the component/s of the soda ash sample? c. Calculate the percent composition of the component/s. No need to include error propagation, but you still need to apply Significant Figure rules.Sources of Error Determine the relationship between the observed/apparent value (EX) VERSUS that of the true value (ET) for the quantity being sought by writing either <, >, or = on the space provided TOPIC: Determination of % SO32-Not all solid was transferred during the filtration of the precipitate. EX _____ ET
- 1. Calculate the experimental density of a salt solution and the percent error (same as relative error percent) using some or all the data given below. solubility of NaCl salt in water: 0.357 g/mLmass of empty graduated cylinder: 25.19g mass of graduated cylinder + salt solution: 30.47g total volume of salt solution: 4.98 mLtrue density of salt solution: 1.07 g/mLcan you answer, please? the experimental data are Reference: https://www.youtube.com/watch?v=OOXRkycKEOc&feature=youtu.be and https://www.youtube.com/watch?v=3wLJLm0QLpg&feature=youtu.beA variation of the indicator-dilution method (see preceding problem) is used to measure total blood volume. A known amount of a tracer is injected into the bloodstream and disperses uniformly throughout the circulatory system. A blood sample is then withdrawn, the tracer concentration in the sample is measured, and the measured concentration [which equals (tracer injected)/(total blood volume) if no tracer is lost through blood vessel walls] is used to determine the total blood volume.In one such experiment, 0.60 cm3 of a solution containing 5.00 mg/L of a dye is injected into an artery of a grown man. About 10 minutes later, after the tracer has had time to distribute itself uniformly throughout the bloodstream, a blood sample is withdrawn and placed in the sample chamber of a spectrophotometer. A beam of light passes through the chamber, and the spectrophotometer measures the intensity of the transmitted beam and displays the value of the solution absorbance (a quantity that…
- Benzene is dissolved at a constant initial concentration of 350 mg/l and is contained within a landfill in leachate water above bottom liner of the landfill system. It starts to diffuse through the liner at time t = 0. Using Equation Ci (x,t) = Co erfc x/ 2(D*t)0.5 related narrative, and (error function table), calculate the concentration of benzene at its breakthrough location as it diffuses through a 1.5 meter thick liner (x=1.5 meters represents breakthrough location where liner fails of contaminant is released), after 50 years. Assume D = 10-5 m2/sec (diffusion coefficient in free standing water) and w = 0.7 (coefficient of tortuousity). Clearly present your assumptions and your value of erf, Select the answer range which best fits the answer you calculate. Group of answer choices 315 to 350 mg/l My answer is not in the range of any of the choices my answer. 215 to 314 mg/l 175 to 200 mg/l 370 to 400 mg/lWith the images attached please write 3 paragraphs 1st paragraph: objective stated clearly 2nd paragraph: complete condense method describe 3rd paragraph: analysis explained Please please please answer everything it's very important please answer super super fast NOTE: This does not require hands-on experimentationDesign a procedure on how you will prepare 1.25 M table sugar (C12H22O11)solution and 1.25 M table salt (NaCl) solution diluted to 1 L water. Perform three trialseach set-up and get the average. Write/ encode your output in short bond paper withthe following parts:Title____________________I. Objectives:II. Materials:III. Procedure (including pictures/diagrams):IV. Data:V. Generalization:

