If the equation produces from data is y = 0.300x - 0.857 and the absorbance for the unknown is 0.429. What would be the concentration of the unknown. Give the answer with 3 decimal places.
If the equation produces from data is y = 0.300x - 0.857 and the absorbance for the unknown is 0.429. What would be the concentration of the unknown. Give the answer with 3 decimal places.
Chapter14: Formula Method
Section: Chapter Questions
Problem 36SST
Related questions
Question
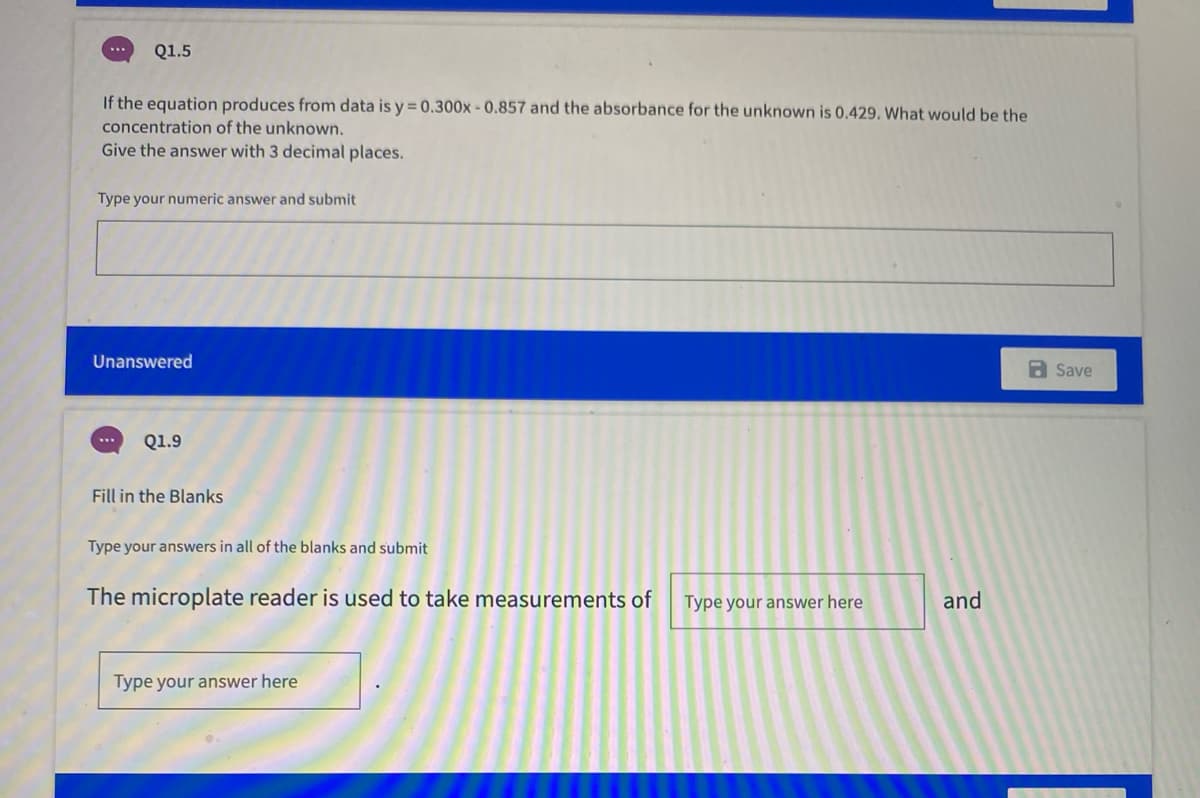
Transcribed Image Text:Q1.5
...
If the equation produces from data is y = 0.300x-0.857 and the absorbance for the unknown is 0.429. What would be the
concentration of the unknown.
Give the answer with 3 decimal places.
Type your numeric answer and submit
Unanswered
a Save
Q1.9
Fill in the Blanks
Type your answers in all of the blanks and submit
The microplate reader is used to take measurements of
Type your answer here
and
Type your answer here
Expert Solution
Step 1
Note: Since you have posted multiple independent questions in the same request, we will solve the first question for you. To get the remaining questions solved, please repost other questions independently.
A spectrophotometric technique is used to measure the absorbance of the unknown solution and it will measure the light quantity passing through the solution.
The standard curve between the absorbance and the concentration of the solution is drawn and the straight-line graph is obtained which is given by the equation, y=mx+b.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



