Q: What is the main difference between space group and point group symmetries? Which one do we prefer…
A: Symmetry group: In group theory, a Symmetry group for an object is a group of all the…
Q: Determine the point groups of the following molecule: XeF4
A: A point group express all the symmetry operations that can be execute on a molecule that result in a…
Q: Define the term order in symmetry operations?
A: To find: The definition of order n symmetry operations
Q: For each of the following molecules, list the symmetry elements present and assign its point group.…
A: The symmetry elements and point group for a, b & c = ? Note : As per our company guidelines :…
Q: Please explain Point group symmetry for a molecule with hexagonal symmetry such as bilateral…
A: The hexagonal symmetry means that the molecule must have the C6 rotation symmetry element. It is…
Q: Identify the point groups to which the following objects belong: (i) a sphere, (ii) an isosceles…
A: a. The circle has its origin fixed so that it belongs to the r3 point group which has its origin…
Q: Determine the point groups of the px atomic orbitals, including the signs on the orbital lobes.
A: The point group is basically the group of symmetries in which at least one point is fixed. It…
Q: What point groups and the symmetry elements
A: Solution: To know the point group for given compound or object we need to know about principle axis,…
Q: Determine the point groups for A snowflake .
A: Point groups are determined by getting symmetries by keeping one point fixed. The point group is a…
Q: 4. Describe an object in nature (not a molecule or ion!) whose symmetry corresponds to each of the…
A:
Q: Determine the point groups for a triangular prism .
A: Point group: It is used to detect the all symmetry elements existing in the molecules which helps in…
Q: Find the point groups of all the possible geometrical isomers for the complex MA2B2C2 in which there…
A: Isomers are those molecules that have same molecular formula but differ in their arrangement of…
Q: Determine the point groups for The possible isomers of tribromobenzene .
A: The structure of chemical compound can be represented as structural formula. A structural formula…
Q: Homework : What is the point group of the following. Justify your answer and indicate the main…
A: Point group symmetry of the given elements. As per our guidelines we have to answer first three…
Q: What is the point group of XeOF2 (Xe is the central atom) ?
A: In XeOF2 the central atom Xe undergo SP3d hybridization. Xe contains two lone pairs. So, the…
Q: 2. Draw the structure of borazine and identify its symmetry elementals and operations
A:
Q: What is the main difference between space group and point group symmetries? Which one do we prefer…
A: Following are the differences between space group and point group in a crystal lattice and group…
Q: Determine the point group of SF;Cl.
A: Point group of SF5Cl ?
Q: Can molecules belonging to the point groups D2h or C3h be chiral? Explain your answer.
A:
Q: List all the symmetry elements of the following molecules and determine their point groups. a)…
A: The molecule contains the following symmetry elements: 1,2,4-trichlorobenzene has a plane of…
Q: What is the point group of [Cu(II)(H2O)6]2+?
A: The point group for above complex is
Q: 5. (a) Show that for a Co, point group from the following : (i) EL,? = h = 4 iit All representations…
A: The character table for C2V is given
Q: 1. Draw and write all the symmetry operations (C.. a. S.. E and i) for each molecule or object. 2.…
A:
Q: 3- Determine the geometry of the BrCl3 molecule using the VSEPR theory. Write down the symmetry…
A: Bromine covalently bonds with three chlorine atoms. Three valence electrons of Br is involved in…
Q: What is the point group of cisplatin? H3N, H3N' CI
A: Cisplatin is a coordination complex having square planner structure. In cisplatin, the central metal…
Q: (a) symmetry of each. Draw the possible isomers of Ru(CO)4F2 and determine the point group
A: Soln
Q: b) 1,3, 5-trichloro benzene (Planar) CI CI
A: Symetry elements which are mirror image to each other. A plane which have plane of symetry that is…
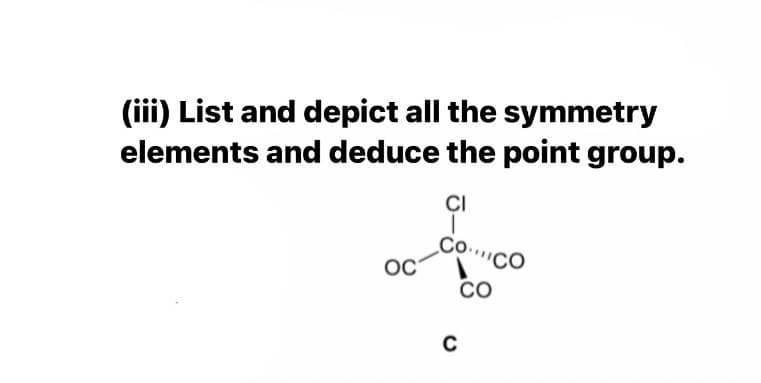
Q: Determine the point group for the following molecule: OCM CO CO
A: The organometallic compound contains the principle axis C3.
Q: What is the difference between the symmetry operators and point groups of NF3 and NF2CI?
A:
Q: identify the symmetry elements for each isomer of Ma3b2.
A:
Q: Sketch the following molecules or ions, include the important symmetry elements on the drawing and…
A: The molecular structure of a molecule/ion provides information about the arrangement of the…
Q: 1. For the following molecules determine the point group: CI H Fe ClC-CI
A:
Q: point groups and symmetry elements
A:
Q: (ii) List and depict all the symmetry elements and deduce the point group. NH2 N' NH2 B
A: There are several rules applied to determine the point group of a compound depending on the symmetry…
Q: 1. Using VSEPR, determine the shape of the molecules, symmetry elements/operation and assign the…
A: a) CCl4 i) The Name of CCl4 molecules : Carbon tetrachloride. ii) The molecular…
Q: List the symmetry elements of the following molecules and name the point groups to which they…
A: A point group can be defined as a set of all the symmetry operations, the action of such operations…
Q: 6- What is the difference between the symmetry operators and point groups of NF3 and NF2CI
A: In the given question, we have to differentiate between NF3 and NF2Cl on the basis of there point…
Q: how to determine irreducible representations of pyrazine? point group d2h
A:
Q: What is the basis of C versus D point group classification?
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: All mirror plane of HOCI, PTCI2B12 and the other symmetry elements Or Determination of the point…
A: HOCl doesn't contain mirror plane. PtCl2Br2 contain mirror plane CCl4 point group- Td. BCl3 point…
Q: What is the point group of the molecule SOF4 (S is the central atom)?
A:
Q: Why are there only 10 2D plane point groups n crystallography?
A:
Q: Identify the point group to which each of the possible isomers of dichloroanthracene belong.
A:
Q: 5. Use the character table of the Cav point group to assign the nine "s", "p", and d" orbitals to…
A: C3v point group is very common, its character table should be known specially Area IV, containing…
Q: cis [Fe(NH3)2(CO)2Br2] symmetry elements and point group?
A: Structure of cis Fe(NH3)2(CO)2Br2 is
Q: The CH3Cl molecule belongs to the point group C3v. List the symmetry elements of the group and…
A: In chloromethane (CH3Cl), the central carbon is bonded to three hydrogen atoms and a chlorine atom.…
Q: 2) Use the C2 point group to illustrate that the irreducible representations in a character table…
A: The character table for the C2 point group is shown below.
Q: What is the point group of CIOF4 (Cl is the central atom)?
A:
Q: 1. For each of the following molecules, list the symmetry elements present and assign its point…
A: As per our company guidelines we are supposed to answer only first 3subparts.kindly repost other…
Step by step
Solved in 2 steps with 2 images

- Determine which single symmetry operation of the following point groups is equivalent to the given combination of multiple symmetry operations. a In C2v, C2v=? b In C2h, iC2=? c In D6h, C6h=? d In D2d, C2C2=? e In Oh, iS4=?a In the Td point group, an S41 improper rotation is equivalent to what other improper rotation? b In the D6h point group, the symmetry operation labeled C21 is equivalent to what other symmetry operation?Without using the great orthogonality theorem, reduce the given irreducible representation in C2v symmetry. Does your answer make sense? EC25555
- Explain why this proposed irreducible representation for C2v is impossible. EC2A?1100Reduce the following reducible representations using the great orthogonality theorem. a In the C2 point group: EC251 b In the C3v point group: E2C23v600 c In the D4 point group: E2C4C22C22C262224 d In the Td point group: E8C33C26S46d72311Point groups are called such because all of the symmetry elements in the group intersect at one point in space. For point groups that have i as a symmetry operation, why must i be at that point?
- Construct the symmetry-adapted linear combination molecular orbitals for hydrogen sulfide, H2S.Linearly polarized light can be assigned a specific irreducible representation of a symmetry point group. If the electronic ground state of methane has A1g symmetry and x- polarized light has the label T2, what are the symmetry labels of allowed excited electronic states? Use the Td character table in Appendix 3.Structural isomers can have very different point groups. Determine the point groups of 1,4cyclohexadiene and 1,3cyclohexadiene, which both have the molecular formula C6H8.
- Determine the point groups of the following molecules. a Hydrogen selenide, H2Se b Partially deuterated hydrogen sulfide, or HDS c The chair conformer of cyclohexane, C6H12 d The boat conformer of cyclohexane, C6H12Why is it unnecessary to consider whether an irreducible representation from C4h is orthogonal to an irreducible representation of D6h?Show that any irreducible representation of these point groups is normalized. a C2 b D2d c Oh d C4h e C6v

