Q: What is the main difference between space group and point group symmetries? Which one do we prefer…
A: Symmetry group: In group theory, a Symmetry group for an object is a group of all the…
Q: Determine the point groups of the following molecule: XeF4
A: A point group express all the symmetry operations that can be execute on a molecule that result in a…
Q: Define the term order in symmetry operations?
A: To find: The definition of order n symmetry operations
Q: Please explain Point group symmetry for a molecule with hexagonal symmetry such as bilateral…
A: The hexagonal symmetry means that the molecule must have the C6 rotation symmetry element. It is…
Q: Determine the point groups for Formaldehyde, H2CO .
A: Formaldehyde having Molecular formula H2CO and it has functional group of aldehyde. Structure of…
Q: Identify the point groups to which the following objects belong: (i) a sphere, (ii) an isosceles…
A: a. The circle has its origin fixed so that it belongs to the r3 point group which has its origin…
Q: - Determine the irreducible representations with reasons in the following character table. CE IR. R,…
A: When E=1 ,then one dimensional, representation may A or B If it is symmetric about principal axis…
Q: Determine the point groups for Chloroethylene.
A: Chloroethylene is the ethylene molecule attack with chlorine atom that is also known as…
Q: sO2 belongs to which point group? C2v .A O C2h .BO Doh.cO C2.DO
A:
Q: What is the point group of the following molecule
A: Molecular symmetry describes the kind of symmetry that exists in the organic molecules. It is…
Q: (i) What is the basis of C versus D point group classification?
A:
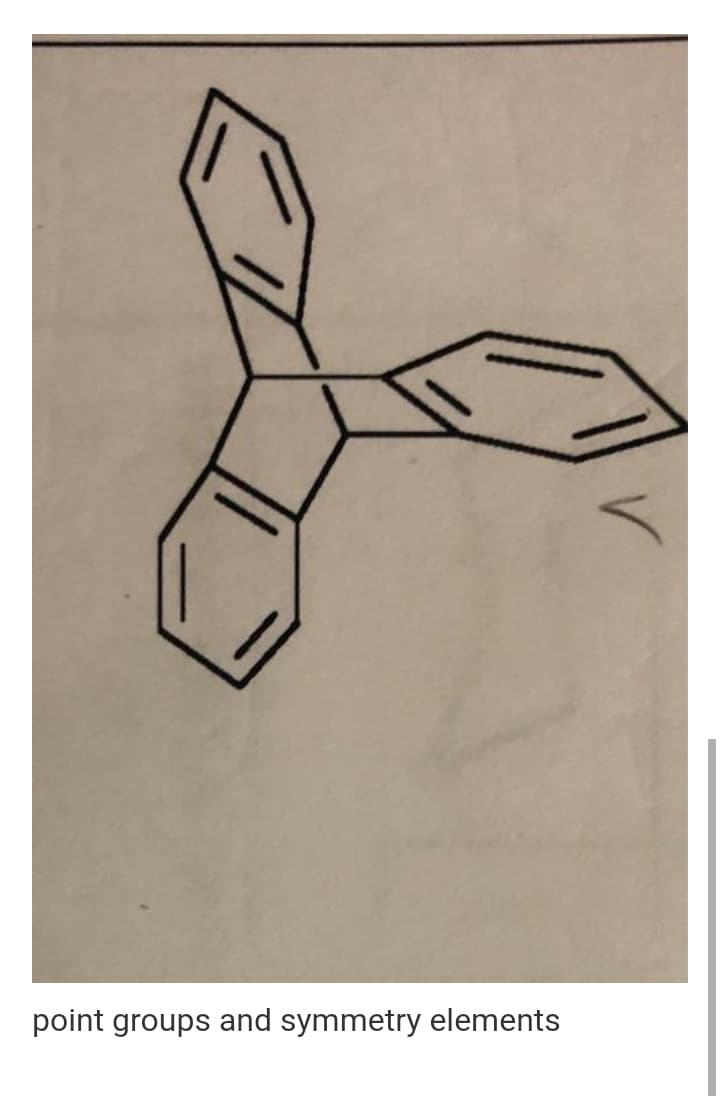
Q: Determine the point group of the molecule shown. The answer should have the correct formatting for…
A:
Q: What is the point group of SbF, ? C1 O C3h C3 O C3v
A: The given molecule is SbF3.
Q: An example of a real life object that has a point group of D6h is a snowflake. What are examples of…
A: A question about point group, which is to be accomplished.
Q: (b) Work out the point group of- (i) CIF;; (ü) IF5.
A:
Q: What point groups are represented by the symbols of chemical elements?
A: A point group is a collection of the symmetry elements present in a molecule that obeys the…
Q: What is the main difference between space group and point group symmetries? Which one do we prefer…
A: Following are the differences between space group and point group in a crystal lattice and group…
Q: Draw a pz and a dz2 orbital and assign their point groups
A: Px is symmetrical and dz2 is symmetrical
Q: Show that any irreducible representation of these point groups is normalized. (a) C4h (b) C6v
A:
Q: PF3 belong to: F: C3y point group A O C2v point group .BO C4v point group.c O Td point group.D O :i:
A: Point group to which the molecule belongs is identified based on the symmetry operations that can be…
Q: Identify the point group to which each of the possible isomers of dichloronaphthalene belong.
A: The possible isomers of given dichloronaphthalene compound are shown below,
Q: 3. Determine the multiplication table for the D2h point group.
A: Multiplication table: for any molecule as a group all the symmetry operations can be written in the…
Q: 1. For the following molecules determine the point group: CI H of Fe CIC-CI CI CI
A: The point group of a molecule gives the information about the symmetry operations that can be…
Q: 1) Using corresponding matrices for the operations of C3V point group, show that they fulfill the…
A:
Q: 8- How many classes are there in D3d point group. write them. Write one subgroup of this point…
A: Point groups are a set of symmetry elements for a molecule. Class is a set of theoretical collection…
Q: In which point group are the letters A, F, N respectively? A. C2v, Cs, C2h B. C2v, D2h, Cs…
A:
Q: In which point group are the letters A, F, N respectively?
A:
Q: (i) List and depict all the symmetry elements and deduce the point group. H CI CI H A
A:
Q: What point groups result from the combination of two mirror planes oriented at 90° with respect to…
A: Point group tells about the symmetry within a molecule. The following symmetries can be present…
Q: A point group has the following sets of elements E, 8 C3, 3C2, 6 S4, 6 od. The order and number of…
A: We know tetrahedral point group has E, 8 C3 , 3 C2 , 6 S4 and 6 σd. Where the numbers before…
Q: What is the point group of the following molecule
A: The symmetry elements present in Pt(CH2Cl)2 are E, 2C4, C2, 2C2’, 2C2”, i, 2S4, sigma h, 2 sigma v,…
Q: 1. Using VSEPR, determine the shape of the molecules, symmetry elements/operation and assign the…
A: a) CCl4 i) The Name of CCl4 molecules : Carbon tetrachloride. ii) The molecular…
Q: (b) Work out the point group of (i) CIF;; (ü) IF5.
A: To determine point group of a molecule , we first calculate the symmetry elements present in the 3D…
Q: HCN belong to: H- cEN Cs point group .A O Cooy point group.B C1 point group.C Doh point group.DO
A: HCN as shown in the figure is a linear molecule. There are only two point group possible for a…
Q: how to determine irreducible representations of pyrazine? point group d2h
A:
Q: How many classes are there in Dsa point group, write them. Write one subgroup of this point group.
A: For a Dsa group, ie, D3h group, total no. Of different symmetry operations of a molecule is given by…
Q: Which of the following is the point group for SF5Cl? A. C2v B. D2h C. D4v D. D4h E. C4v
A: For SF5Cl: The point group has to be determined.
Q: Reduce the following representations for C2h. C2v and Cav point groups into corresponding…
A:
Q: Show symmetry elements of cis-CHCl=CHCl and determine its point group
A: We have to predict symmetry elements and point group of given compound.
Q: Show that any irreducible representation of these point groups is normalized. (a) C2 (b) D2d (c) On…
A:
Q: (4) Determine the point group of CHa and show the symmetry elements/operations using 3D drawing. (5)…
A: In point group classification we have to find out different axis of rotation,plane of rotation ,if…
Q: O.
A: Pont group comes from the mathematical treatment to a molecule . By the point group we can explain…
Q: Determine the point group of the molecule shown. Be sure to rotate the molecule so that you can see…
A:
Q: Determine the point group of the pattern on the flag of Japan.
A: SYMMETRY OPERATIONS: In quantum chemistry, a symmetry operation is conducted by a geometrical…
Q: 2) Use the C2 point group to illustrate that the irreducible representations in a character table…
A: The character table for the C2 point group is shown below.
Q: Determine what point group does PF5 belong to, disscuss how that point group is constructed, and…
A: PF5 have D3h point group. PF5 has trigonal bipyramidal shape. Two fluorine atoms are present in…
Q: d) Borazina, HN `NH HB. BH e) [tris(pirazolil)borat]" H ZI
A: A symmetry operation is an action which when performed on a molecule yields a new orientation of it…
Q: How many classes are there in D3 point group, write them. Write one subgroup of this point group
A: Point group are the collection of symmetry operation that performed on a molecule results in…
4
Step by step
Solved in 3 steps with 2 images

- Show that any irreducible representation of these point groups is normalized. a C2 b D2d c Oh d C4h e C6vWhat are the number of classes and the order of the following point groups? a C2v b D2h c D6h d S4 e Cs.Structural isomers can have very different point groups. There are three isomers of dichlorobenzene. Identify the point groups of the three isomers.
- Why is it unnecessary to consider whether an irreducible representation from C4h is orthogonal to an irreducible representation of D6h?Determine the point group of the following molecules. a cis1,2 Dichloroethylene b trans1,2 Dichloroethylene c Toluene, C6H5CH3 d 1,3-Cyclohexadiene.In your own words, explain why an object that has more symmetry elements is said to have higher symmetry than an object with fewer symmetry elements.
- Explain why this proposed irreducible representation for C2v is impossible. EC2A?1100Show that the irreducible representations of the D2d point group satisfy the closure requirement. You will have to use the great orthogonality theorem to reduce one combination.Determine which single symmetry operation of the following point groups is equivalent to the given combination of multiple symmetry operations. a In C2v, C2v=? b In C2h, iC2=? c In D6h, C6h=? d In D2d, C2C2=? e In Oh, iS4=?
- Structural isomers can have very different point groups. Determine the point groups of 1,4cyclohexadiene and 1,3cyclohexadiene, which both have the molecular formula C6H8.Construct the symmetry-adapted linear combination molecular orbitals for hydrogen sulfide, H2S.Identify all the symmetry elements present in the tetrahedron, the cube, and the octahedron.

