Q: How can you use ion exchange Chromatography to de-ionize water? Please shortly write at your own…
A: Answer - Ion exchange Chromatography - Ion chromatography (or ion-exchange chromatography)…
Q: In a normal phase chromatography setup, which would be the predicted characteristics of the eluates…
A:
Q: 2. A solution prepared by combining 90% pentane and 10% heptane is passed through a gas…
A: In Gas Chromatography (GC), the carrier gas stream transports the injected sample solution into a…
Q: What factor allows molecules to be separated by chromatography?
A: Chromatography is a method which is used to separate, identify and purify the components of a…
Q: Is the paper chromatography used normal or reverse?. Explain why
A: With the help of this chromatography, we can separate colored compound or substances.
Q: A student analyzes sample 1, and sample 2 chromatograms of a mixture containing two compounds (A +…
A: The two samples( Compound A+Compound B ) are undergone chromatography : Compound A and Compound B
Q: Chromatography
A:
Q: When using gas chromatography, how is optimum column efficiency obtained? A. When the samples are…
A: Gas chromatography is analytical technique that used in analytical chemistry for separating and…
Q: Using the chromatogram shown here, which was obtained on a 2-m column, determine values for tr, w,…
A:
Q: Which of the following could be a consequence of an open and uncovered chromatographic chamber?…
A: TLC is an analytical technique which can be used to monitor the organic reaction transformations.…
Q: For any chromatographic technique, tall and narrow peaks are preferred to short and broad peaks.
A: Given statement: For any chromatographic technique, tall and narrow peaks are preferred to short and…
Q: Red, blue, and yellow spots were found in the chromatogram. Red spot traveled twice the distance…
A:
Q: Can paper chromatography be used to separate and identify very volatile substances? How do I prove…
A: Mss
Q: Gas chromatography requires a sample size.
A: Dear Subscriber! You have posted multiple questions. As per our policy, we answered only first…
Q: Are there any difference between using a 2% NaCl and using a water in the paper chromatography. If…
A: Chromatography is defined as the process of separation of components on the basis of color.
Q: what do the different colors in the result of paper chromatography experiment indicates? 2. Why do…
A: Paper chromatography is a technique in which colour chemicals or substances are separated.
Q: In a normal phase chromatography set up, the component that yields the highest Rf value is likely to…
A: In a normal phase chromatography set up , the conponent that yields the highest Rf value is likely…
Q: 2. What are mobile and Stationary phases in a chromatogram? Site their nature.
A: The solution is given below -
Q: 24 45 11 4 16 19 21 12 15 3 6 8 9 10 23 13 14 17 18 20 22 1 2 6. 8. 10 12 14 16 18 20 22 24 26 Time…
A:
Q: Explain why a chromatographic separation normally has an optimum flow rate that gives the best…
A: When an fast flow rate is maintained, the band is not allowed to spend much time within the column…
Q: A student accidentally started the chromatographic run without saturating the beaker with the…
A: Answer - The correct option is (d) All of the above Explanation - According to the question -…
Q: How can you use ion exchange Chromatography to de-ionize water? Please shortly answer at your own…
A: Chromatography is used to separate compounds into their various components. There are different…
Q: consequence of an open and uncovered chromatographic chamber? The travel rate of a component…
A: Chromatography is based on the principle where molecules in mixture applied onto the surface or into…
Q: Gestation time arrangement of Hegran, Hexanol, and Benzene compounds in the reverse phase of column…
A: A question based on analytical process that is to be accomplished.
Q: Which of the following chromatographic methods cannot use a mixture of liquid solvents (eluents)…
A: Chromatographic separation porocess and eluent for chromatographic separation.
Q: 1. State the reason why we need to compare the observed retention time values to the standard values…
A: As per the rules, only the first question can be answered. Retention time is the time spent by the…
Q: In a normal phase chromatography setup, which would be the predicted characteristics of the eluates…
A: Ans : Eluates will be in decreasing polarity ( Benzoic acid is more polar than benzaldehyde.…
Q: Question: What will happen to the result of chromatography if both the stationary and mobile…
A: Chromatography is the technique in which the mixture are separated using mobile and stationary…
Q: What are your observations performing paper chromatography
A: Paper chromatography is of two types . Thin layer chromatography Preparative TLC
Q: In one paper chromatography, the Rf for spots X and Y are 0.5 and 0.35 respectively. The solvent…
A: In paper chromatography Rf value is the ratio of distance travelled by compound to distance…
Q: Is the mobile phase for your TLC and column chromatography more or less polar than the stationary…
A: Mobile phase: It is the phase that is getting movement in the column. The sample that has to be…
Q: the efficiency of a chromatographic column improves the measure that a.increases the height of the…
A: We know that, In chromatography, column efficiency is measured in terms of number of theoretical…
Q: m doing lab report for thin layer chromatography right now, but I'm struggling with a question about…
A: TLC is used in organic synthesis to know whether the reaction is completed or not . And the no of…
Q: Here, you can find an example of a paper chromatography experiment. Use the two Chromatograms below…
A: Given that , There are two chromatograms. Following are the appropriate answers of the given…
Q: 1. Given the following chromatogram, mark the middle of each cation spot and calculate the Ry value…
A:
Q: Aside from TLC and paper chromatography, what are the other types of chromatography. Discuss their…
A: Answer - chromatography - Chromatography is a laboratory technique for the separation of a mixture…
Q: Gas chromatography cannot analyze mixtures due to the sensitivity of the instrument. True…
A:
Q: paper chromatography
A: In this question we have to explain that the arrangement of the above compounds in increasing Rf if…
Q: Which of the following statements about the mobile phase in Supercritical fluid chromatography (SFC)…
A: Gas chromatography is a type of chromatography used in analytical chemistry used for separating and…
Q: Which of the following statements is false? * O Chromatographic analysis is independent of film…
A:
Q: 4) If the classification of the chromatography run is a reverse-phase chromatography, discuss the…
A:
Q: In thin layer chromatography, the stationary phase is made of and the mobile phase is made of solid,…
A: Thin layer chromatography: TLC is a chromatography technique used to seperate non-volatile mixture.…
Q: Peak area is used for quantitative analysis in gas-liquid chromatography. True False
A: Is the given statement true or false has to be identified.
Q: First you drop a plastic bead that has a density of 0.32 g/cm into the column. What do you expect to…
A: Density is given by the formula d = m/V where d stands…
Q: One of the main differences between TLC and column chromatography is the direction in which the…
A: Purification is one of the key step in the isolation of organic or inorganic compounds. Different…
Q: Sketch a chromatogram for a compound with a stronger interaction with the stationary phase than the…
A:
Q: What weight of mercury bichloride should be taken as sample, assuming 100% purity, so that 0.5 g og…
A: Weight is calculated as, (weight of sample * GF)/ Weight of analyte.
Q: Match Column A with Column B. Mobile phase is more polar than the stationary phase A. Reverse Phase…
A: Explanation is on the image. 1)-‐---A 2)-----A 3)-----B 4)-----B
Q: 1- What term refers to the ability to distinguish two peaks from one another in a chromatogram? two…
A: Chromatography is a technique used to seperate components from a mixture. Here we have to answer…
I'm not sure how to approach this problem because the ruler on the chromatogram starts at 2. I need help finding the distance for the 3 species traveled.
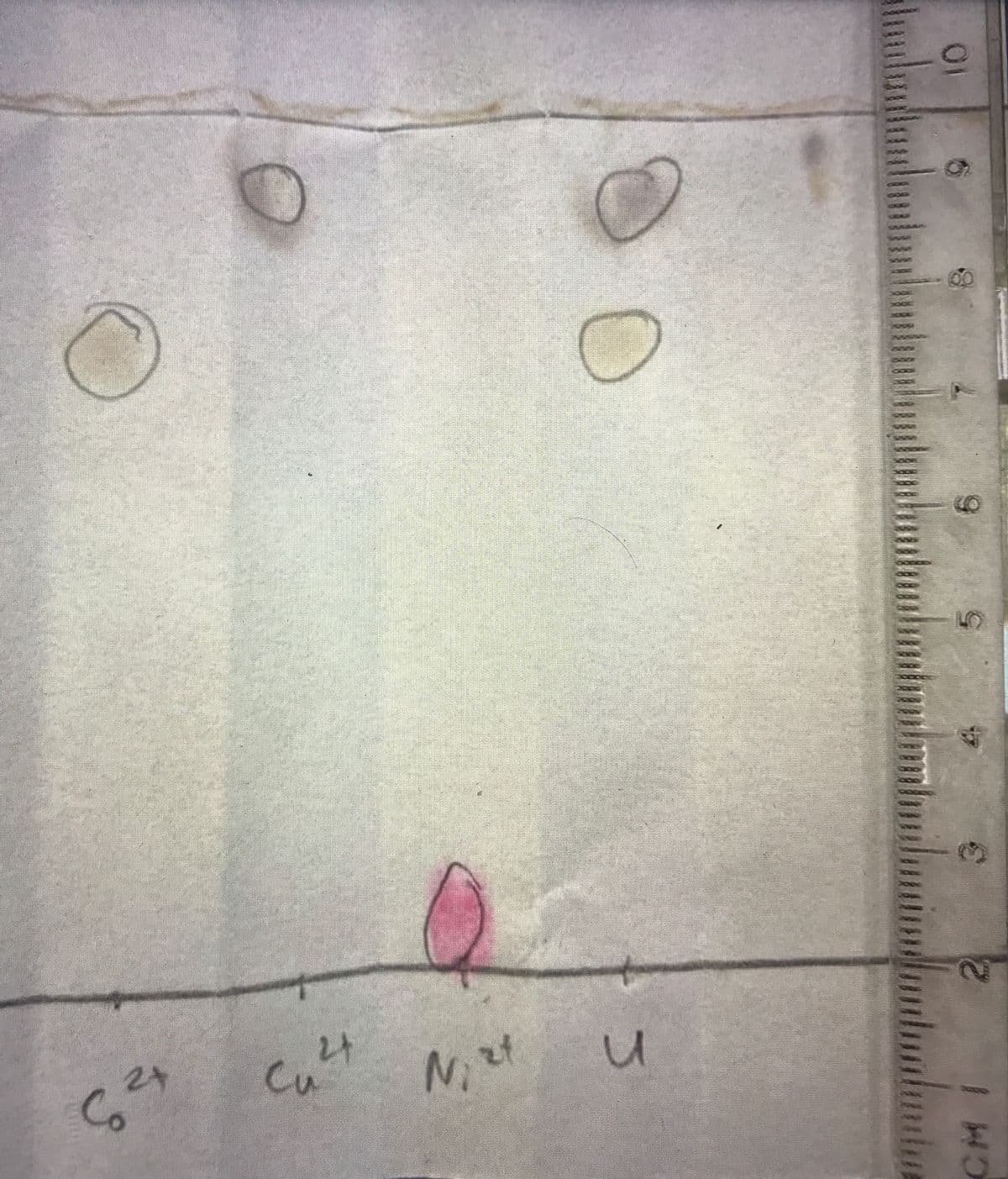

Rf value in chromatography, is the ratio of distance travelled by solute to distance travelled by solvent .
As you have measured distance of solvent to be 7.6cm
Step by step
Solved in 2 steps

- What effect will the following have on plate height (H). Make sure to explain your full and complete reasoning. Reducing the rate of sample injection.1. The properties that must be possessed by the adsorption reagent in air sampling are…a. Reagents must be resistant to oxidationb. Reagents must be qualitativec. Reagents must be applicable to all gaseous analytesd. Reagents must be in large quantitiesDetermine the relationship between the observed/apparent value (EX) VERSUS that of the true value (ET) for the quantity being sought by writing either <, >, or = on the space provided Determination of the true concentration (ppm Fe) in a water sample using the iron-phenanthroline method. Dilution factor was not taken into account. EX _____ 1ET
- For Indirect Iodometric Analysis of Copper... ~0.0896g KIO3 necessary to consume 350mL of 0.1 M Na2S2O3, Na2S2O3 is stored in an amber glass bottle until ready for use. Primary Standard KIO3 has 2g of KI, 50mL of DI water, and 10 mL of 1.0M HCl is added then immediately titrated with Na2S2O3 until medium yellow or straw... then 5mL of starch indicator is added and titrated again until blue black color turns clear. Unknown CuO use 1.2G of Unknown, 20mL of HNO3 heated until sample dissolved, 25 mL of DI water added and boiled until clear light blue color, after cooling 1:1 NH3 added (~34.47 mL of NH4OH reagent) until permanent deep blue color amine complex, 2g of NH4HF2 added and swirled until dissolved, 3 g of KI is added then titrated immediately with Na2S2O3 until brown color of iodide is nearly gone (brown milk color), 2 g of KSCN and 3 mL of starch indicator is then added with titration continuing until disappearance of new blue black color. 1. Na2CO3 is often added to thiosulfate…For Indirect Iodometric Analysis of Copper... ~0.0896g KIO3 necessary to consume 350mL of 0.1 M Na2S2O3, Na2S2O3 is stored in an amber glass bottle until ready for use. Primary Standard KIO3 has 2g of KI, 50mL of DI water, and 10 mL of 1.0M HCl is added then immediately titrated with Na2S2O3 until medium yellow or straw... then 5mL of starch indicator is added and titrated again until blue black color turns clear. Unknown CuO use 1.2G of Unknown, 20mL of HNO3 heated until sample dissolved, 25 mL of DI water added and boiled until clear light blue color, after cooling 1:1 NH3 added (~34.47 mL of NH4OH reagent) until permanent deep blue color amine complex, 2g of NH4HF2 added and swirled until dissolved, 3 g of KI is added then titrated immediately with Na2S2O3 until brown color of iodide is nearly gone (brown milk color), 2 g of KSCN and 3 mL of starch indicator is then added with titration continuing until disappearance of new blue black color. 4. Why is the starch indicator solution…correct this scheme and highlight what needs to be corrected
- Using your graph from the previous question and referring to the equation below, use the trendline equation to determine the molar absorptivity (e) of FD&C Yellow #5 at 430 nm. Remember that you used 1 cm cuvettes. A = ε l C (Hint: y = mx + b and b = 0) My equation was 1342.5x from my graph.Diprivan® (propofol) is a commonly used sedative-hypnotic agent used for sedation and anesthesia during minor as well as major surgical procedures. It is a liquid preparation administered through the IV route of drug administration. Based on what you see below, this prep is most likely a(an): Emulsion Microemulsion Suspension SolutionExplain how the Rf (Retention Factor)-values can be in agreement or disagreement with the “like dissolves like” principle?



