In a series of seven steps, (S)-malic acid is converted to the bromoepoxide shown on the right in 50% overall yield. This synthesis is enantioselective-of the stereoisomers pos- sible for the bromoepoxide, only one is formed. OH COOH A (CgH1405) C (C,H1804) НООС (S)-Malic acid D →E F (C,H,OBr2) Br A bromoepoxide 3. LIAIH4, then H,O 4. TSCI, pyridine 5. NaBr, DMSO Steps/reagents: 1. CH,CH,OH, H+ 6. Н.О, СH,COOН 7. KOH 2. H+ In thinking about the chemistry of these steps, you will want to review the use of dihydropyran as an -OH protecting group (Section 16.7D) and the use of the p-toluenesulfonyl chloride to convert the -OH, a poor leaving group, into a tosylate, a good leaving group non
In a series of seven steps, (S)-malic acid is converted to the bromoepoxide shown on the right in 50% overall yield. This synthesis is enantioselective-of the stereoisomers pos- sible for the bromoepoxide, only one is formed. OH COOH A (CgH1405) C (C,H1804) НООС (S)-Malic acid D →E F (C,H,OBr2) Br A bromoepoxide 3. LIAIH4, then H,O 4. TSCI, pyridine 5. NaBr, DMSO Steps/reagents: 1. CH,CH,OH, H+ 6. Н.О, СH,COOН 7. KOH 2. H+ In thinking about the chemistry of these steps, you will want to review the use of dihydropyran as an -OH protecting group (Section 16.7D) and the use of the p-toluenesulfonyl chloride to convert the -OH, a poor leaving group, into a tosylate, a good leaving group non
Chapter19: Aldehydes And Ketones: Nucleophilic Addition Reactions
Section19.SE: Something Extra
Problem 50MP
Related questions
Question
What is the configuration of the chiral center in the bromoepoxide? How do you account for the stereoselectivity of this seven-step conversion?
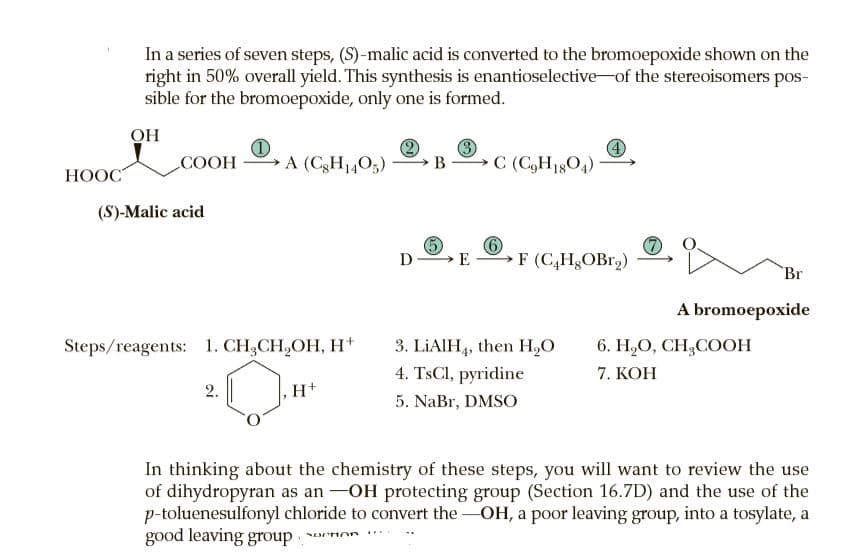
Transcribed Image Text:In a series of seven steps, (S)-malic acid is converted to the bromoepoxide shown on the
right in 50% overall yield. This synthesis is enantioselective-of the stereoisomers pos-
sible for the bromoepoxide, only one is formed.
OH
COOH
A (CgH1405)
C (C,H1804)
НООС
(S)-Malic acid
D
→E
F (C,H,OBr2)
Br
A bromoepoxide
3. LIAIH4, then H,O
4. TSCI, pyridine
5. NaBr, DMSO
Steps/reagents: 1. CH,CH,OH, H+
6. Н.О, СH,COOН
7. KOH
2.
H+
In thinking about the chemistry of these steps, you will want to review the use
of dihydropyran as an -OH protecting group (Section 16.7D) and the use of the
p-toluenesulfonyl chloride to convert the -OH, a poor leaving group, into a tosylate, a
good leaving group non
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning