In a spectrophotometry experiment, (34.1±0.2)% of the light (=I/Io) was transmitted through the sample. The molar absorptivity, e, of the analyzed was measured separately and determined to be (1.47±0.03)×104M' cm', and the path length of light, b, was (1.00 + 0.01) cm. Determine what is the concentration and the uncertainty in concentration of the analyte, with proper units. Be sure to show your work. In this problem you will need to use Beer's Law.
In a spectrophotometry experiment, (34.1±0.2)% of the light (=I/Io) was transmitted through the sample. The molar absorptivity, e, of the analyzed was measured separately and determined to be (1.47±0.03)×104M' cm', and the path length of light, b, was (1.00 + 0.01) cm. Determine what is the concentration and the uncertainty in concentration of the analyte, with proper units. Be sure to show your work. In this problem you will need to use Beer's Law.
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter1: Introduction
Section: Chapter Questions
Problem 1.9QAP
Related questions
Question
100%
Do you have any clues??
Transcribed Image Text:Styles Styles
Pane
Dicm
6
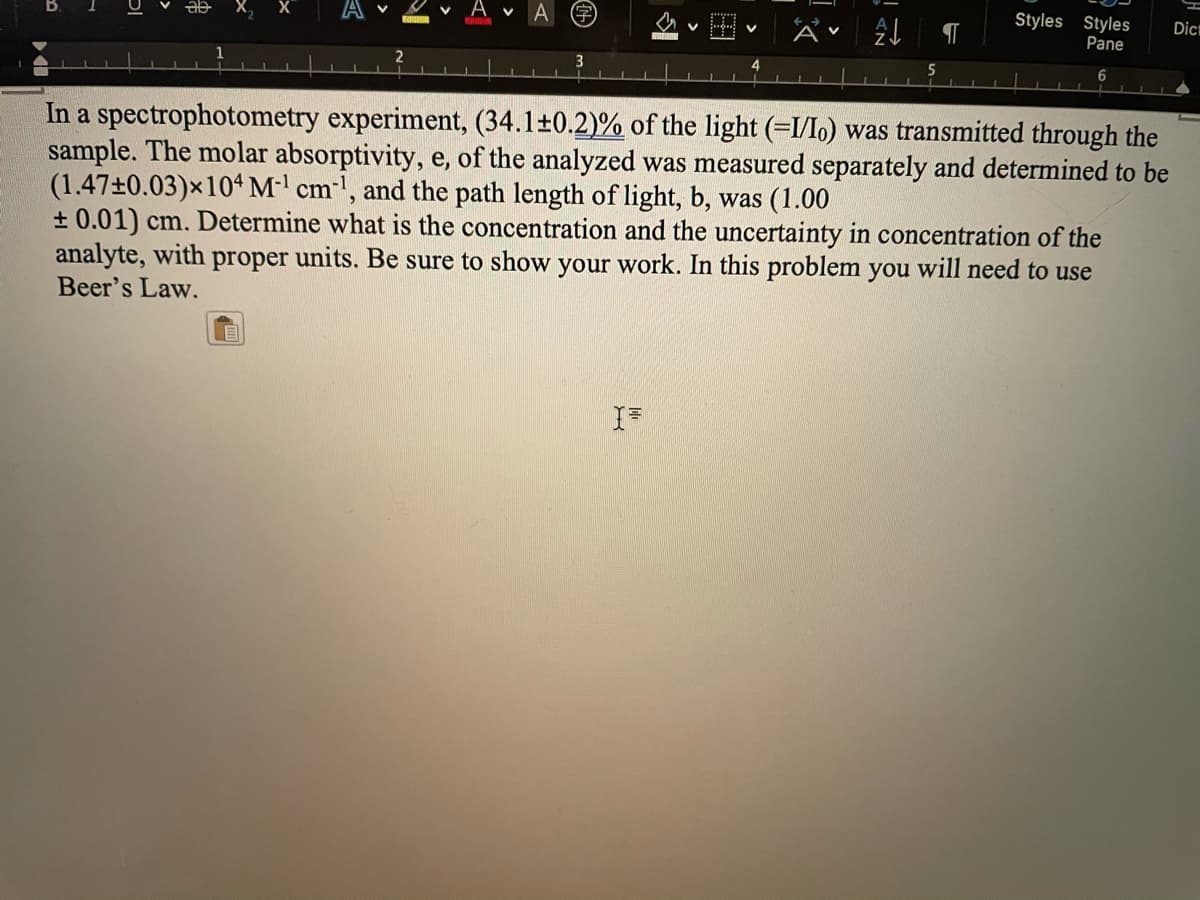
In a spectrophotometry experiment, (34.1±0.2)% of the light (=I/lo) was transmitted through the
sample. The molar absorptivity, e, of the analyzed was measured separately and determined to be
(1.47±0.03)×104M' cm', and the path length of light, b, was (1.00
+ 0.01) cm. Determine what is the concentration and the uncertainty in concentration of the
analyte, with proper units. Be sure to show your work. In this problem you will need to use
Beer's Law.

Transcribed Image Text:e oiar absorptivity, ɛ, of the analyzed was mea
±0.03)×10ª M-' cm', and the path length of lig
e what is the concentration and the uncertainty in
its. Be sure to show your work. In this problem
-1
A = –log10 ()
-log10 2.303
=,207 In (-)
ɛbc
ed to reduce the uncertainty in your final result, w
ments should you focus on making better? Or equi
nty in fraction of light absorbed, uncertainty in mo
oth) cotributes most to the overall uncertainty in t
Expert Solution
Step 1
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT