The following instrumental readings were obtained for the determination of low concentrations of sodium by flame photometry. 175, 104, 164, 193, .131, 189, 155,133, 151 and 176. Ten blanks had a mean reading of 45.O. The slope of the calibration curve is 1.75x10 M :Answer the following questions (-1) Mean concentration of sodium (X Standard deviation of the sodium measurement Signal detection limit iv) Lowest detection limit in umole
The following instrumental readings were obtained for the determination of low concentrations of sodium by flame photometry. 175, 104, 164, 193, .131, 189, 155,133, 151 and 176. Ten blanks had a mean reading of 45.O. The slope of the calibration curve is 1.75x10 M :Answer the following questions (-1) Mean concentration of sodium (X Standard deviation of the sodium measurement Signal detection limit iv) Lowest detection limit in umole
Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.24QAP
Related questions
Question
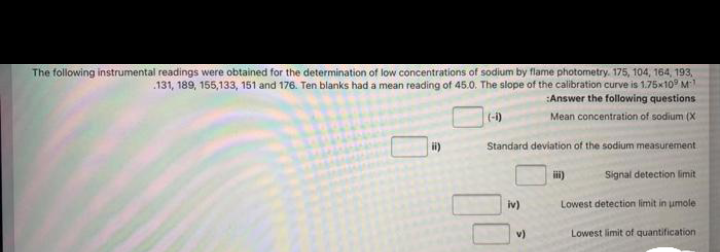
Transcribed Image Text:The following instrumental readings were obtained for the determination of low concentrations of sodium by flame photometry. 175, 104, 164, 193,
131, 189, 155,133, 151 and 176. Ten blanks had a mean reading of 45.0. The slope of the calibration curve is 1.75x10 M
Answer the following questions
(-1)
Mean concentration of sodium (X
Standard deviation of the sodium measurement
Signal detection limit
iv)
Lowest detection limit in umole
v)
Lowest limit of quantification
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



