In each case below select the synthetic procedure/s that could be used to carry out the transformation, giving the alcohol shown as the single major product. The procedures are: Hydroboration/oxidation: alkene + BH3; then H,O2, "OH. Oxymercuration: alkene + Hg(OAc)2, H,0; then NaBH4 CH2CH3 CH2CH3 Но. CH3 CH3 `CH3 `CH3
In each case below select the synthetic procedure/s that could be used to carry out the transformation, giving the alcohol shown as the single major product. The procedures are: Hydroboration/oxidation: alkene + BH3; then H,O2, "OH. Oxymercuration: alkene + Hg(OAc)2, H,0; then NaBH4 CH2CH3 CH2CH3 Но. CH3 CH3 `CH3 `CH3
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter24: Catalytic Carbon-carbon Bond Formation
Section: Chapter Questions
Problem 24.18P: Show the sequence of Heck reactions by which the following conversion takes place. Note from the...
Related questions
Question
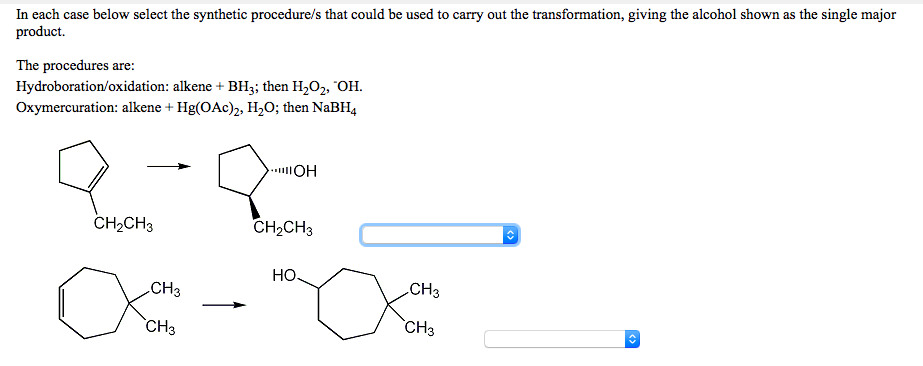
Transcribed Image Text:In each case below select the synthetic procedure/s that could be used to carry out the transformation, giving the alcohol shown as the single major
product.
The procedures are:
Hydroboration/oxidation: alkene + BH3; then H2O2, "OH.
Oxymercuration: alkene + Hg(OAc)2, H,O; then NaBH4
CH2CH3
CH2CH3
НО.
CH3
CH3
`CH3
`CH3
<>
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning