In RbF(s), the Rb* ions occupy octahedral sites in a face-centred cubic lattice of Fions. Use the data below to estimate the size of lattice parameter of the unit cell, the mass of the unit cell, the volume of the unit cell, and the density of the solid. lon Rb F Mass 85.47 g mol-¹ 19.00 g mol-1 Radius 149 pm 133 pm Enter your answers to the following questions in the specified units and with the specified number of digits. Do not include units as part of your answer. (a) What is the lattice parameter of the unit cell, in picometres (pm) ? Number rounded to the nearest picometre.) pm (Enter your answer
In RbF(s), the Rb* ions occupy octahedral sites in a face-centred cubic lattice of Fions. Use the data below to estimate the size of lattice parameter of the unit cell, the mass of the unit cell, the volume of the unit cell, and the density of the solid. lon Rb F Mass 85.47 g mol-¹ 19.00 g mol-1 Radius 149 pm 133 pm Enter your answers to the following questions in the specified units and with the specified number of digits. Do not include units as part of your answer. (a) What is the lattice parameter of the unit cell, in picometres (pm) ? Number rounded to the nearest picometre.) pm (Enter your answer
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter9: Liquids, Solids, And Materials
Section: Chapter Questions
Problem 115QRT
Related questions
Question
What is the lattice parameter of the unit cell, in picometres (pm) ?
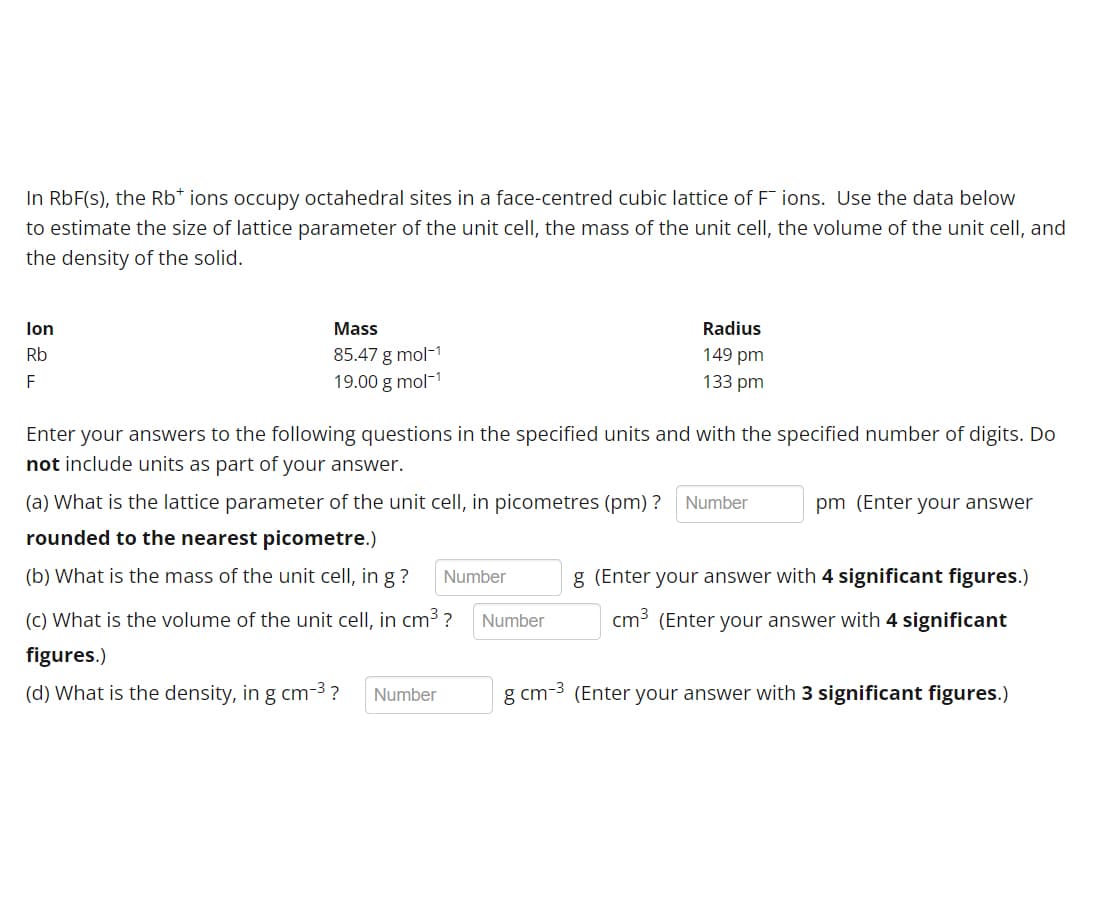
Transcribed Image Text:In RbF(s), the Rb* ions occupy octahedral sites in a face-centred cubic lattice of Fions. Use the data below
to estimate the size of lattice parameter of the unit cell, the mass of the unit cell, the volume of the unit cell, and
the density of the solid.
lon
Rb
F
Mass
85.47 g mol-1
19.00 g mol-¹
Enter your answers to the following questions in the specified units and with the specified number of digits. Do
not include units as part of your answer.
Radius
149 pm
133 pm
(a) What is the lattice parameter of the unit cell, in picometres (pm) ? Number
rounded to the nearest picometre.)
(b) What is the mass of the unit cell, in g?
(c) What is the volume of the unit cell, in cm³ ?
figures.)
(d) What is the density, in g cm-³? Number
Number
Number
pm (Enter your answer
g (Enter your answer with 4 significant figures.)
cm³ (Enter your answer with 4 significant
g cm-3 (Enter your answer with 3 significant figures.)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning