In the carbonate diagram shown (same as in previous figure), report the approximate values for K₂1 and K₂2. Recall that K₁1 governs the equilibrium between H₂CO3 and HCO3, and K₂2 governs the equilibrium between HCO3 and CO3²-. Species Fraction 1 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0 0 DXC 6 7 1 2 3 4 Ka1 is 4.5E-7, and Ka2 is 4.7E-11 pka1 is 9.1 and pka2 is 5.9 Ka1 is 1.15E-6 and Ka2 is 7.5E-10 pka1 is 6.3, pka2 is 10.3 5 pH 8 9 10 11 12 13 B C
In the carbonate diagram shown (same as in previous figure), report the approximate values for K₂1 and K₂2. Recall that K₁1 governs the equilibrium between H₂CO3 and HCO3, and K₂2 governs the equilibrium between HCO3 and CO3²-. Species Fraction 1 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0 0 DXC 6 7 1 2 3 4 Ka1 is 4.5E-7, and Ka2 is 4.7E-11 pka1 is 9.1 and pka2 is 5.9 Ka1 is 1.15E-6 and Ka2 is 7.5E-10 pka1 is 6.3, pka2 is 10.3 5 pH 8 9 10 11 12 13 B C
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter11: Solutions
Section: Chapter Questions
Problem 75AP
Related questions
Question
Answer correct with explain
Please don't answer short.
Full details with correct.
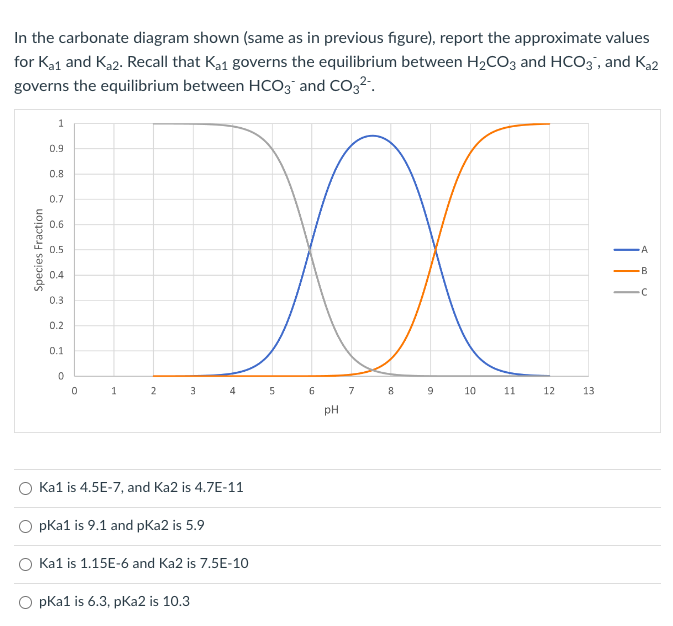
Transcribed Image Text:In the carbonate diagram shown (same as in previous figure), report the approximate values
for K₂1 and K₂2. Recall that K₂1 governs the equilibrium between H₂CO3 and HCO3, and Ka2
governs the equilibrium between HCO3 and CO3²-.
Species Fraction
1
0.9
0.8
0.7
0.6
0.5
0.4
0.3
0.2
0.1
0
0
1
2
XXX
8
3
4
Ka1 is 4.5E-7, and Ka2 is 4.7E-11
O pka1 is 9.1 and pka2 is 5.9
pKa1 is 6.3, pka2 is 10.3
Ka1 is 1.15E-6 and Ka2 is 7.5E-10
5
6
pH
7
10
11
12
13
A
B
C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 8 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning