In which of the following pairs Is the first eleme than the second? (1 Point) O 0, P O Cs, Rb O I, Br O Al, P
In which of the following pairs Is the first eleme than the second? (1 Point) O 0, P O Cs, Rb O I, Br O Al, P
Principles Of Radiographic Imaging: An Art And A Science
6th Edition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Chapter2: Radiation Concepts
Section: Chapter Questions
Problem 6RQ
Related questions
Question
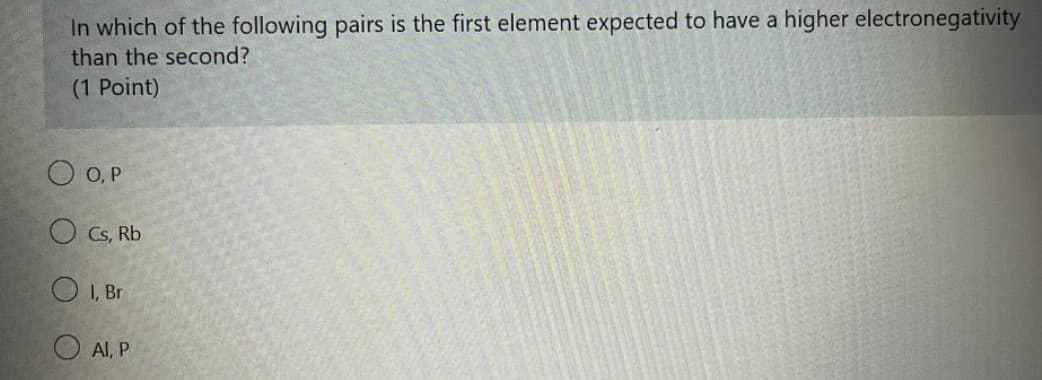
Transcribed Image Text:In which of the following pairs is the first element expected to have a higher electronegativity
than the second?
(1 Point)
O O, P
O Cs, Rb
O I, Br
Al, P
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning


Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning


Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning