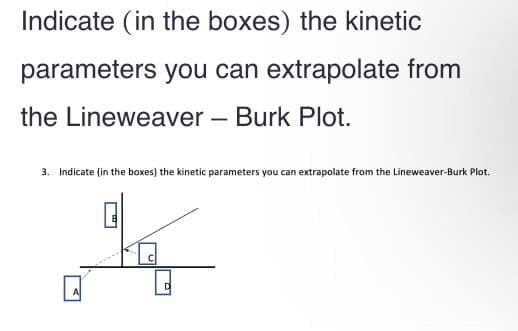
Indicate (in the boxes) the kinetic parameters you can extrapolate from the Lineweaver - Burk Plot. 3. Indicate (in the boxes) the kinetic parameters you can extrapolate from the Lineweaver-Burk Plot. D
Q: Provide a schematic representation of the reactions in the beta
A: Beta oxidation is a fundamental metabolic process involving catabolism of fatty acids into energy as…
Q: 2. Compare and contrast the biological roles of the following amino acids the following pairs of…
A: The objective of this question is to compare and contrast the biological roles of three pairs of…
Q: NO AI GENERATED RESPONSE I NEED EXPERTS!! using results for experiment below conduct 1 graph of the…
A: The objective of this question is to create a graph that visually represents the correlation between…
Q: B- Calculate the missing value of the amino acids listed in the table below. Amino acid…
A: Amino acids are the basic units of proteins. They have an amino group, a carboxyl group and a side…
Q: Provide a stepwise, arrow-pushing mechanism for the following transformation. You may use general…
A: The reaction in question involves a lysis reaction catalyzed by a lyase enzyme using the coenzyme…
Q: The authors in the abstract given above describe the mechanism for the activation of metallothionein…
A: Protein Phosphatase 2A (PP2A) Binding: Complexes of PP2A PR110 bind to MTF-1, the metal regulating…
Q: p53 is an activator of a miRNA called miR-145. miR-145 targets c-myc, a gene that promotes cellular…
A: Increased proliferation and survival.Explanation:Increased proliferation and survival would be the…
Q: 7. Why proteins prefer to have a-helices or ẞ-sheets in their interior core, instead of random coil…
A: The preference for alpha helices and beta sheets in the interior core of proteins is driven by a…
Q: 3. The image below depicts 2,3-bisphosphoglycerate depicted in dark gray, and ß subunits are…
A: The objective of the question is to understand the role of 2,3-bisphosphoglycerate (BPG) in the…
Q: Draw the structure of cholesterol and write the biosynthetic relations of steroid hormones starting…
A: Cholesterol, a 27-carbon molecule, is the most common steroid in the body and plays a crucial role…
Q: You have a crude lysate sample (CL) containing a mixture of six proteins (1, 2, 3, 4, 5, ẞ-…
A: Proteins precipitate at specific concentrations of various salts. The concentration of salt required…
Q: An enzyme has a rate enhancement of 1.3x106. Calculate the value of ΔΔG‡ at 25.0 °C in kJ mol-1.…
A: Step 1: Step 2: Step 3: Step 4:
Q: Which of the following is true about a mixed type inhibition? a. None of these is true b. A…
A: The enzyme kinetics, inhibition in particular, is vital to understanding and purposefully managing…
Q: To which class of lipid does the molecule below belong? OH HO OH H ...CH 3 H O eicosanoid O…
A: The objective of the question is to determine which category does the given molecule belongs.
Q: Draw the major organic product of the following reaction: 1. (CH3CH2)2NH, H+ (cat.) 2. о H 3. H3O+
A: In the reaction of Cyclopentanone the major product will be a ketone which is…
Q: None
A:
Q: Consider the following Peptide: Alanine-Lysine-Glutamine-Serine-Glycine Select all that applies Give…
A: Peptides are short chain of amino acids linked by peptide bonds. Amino acids that have been…
Q: draw the mechanism for the glyoxalate cycle enzyme malate synthase typed solution
A: The glyoxyate cycle is an alternative pathway that allows the organism to convert acetyl CoA to…
Q: In no more than two sentences each describe electroporation
A: There are several methods or techniques that allow us to introduce foreign DNA into the host cell,…
Q: Describe how the SCAM analysis was performed so that it maintains replicatability.
A: SCAM (Substituted Cysteine Accessibility Method) is an analytical technique which is used to study…
Q: Glycogen contains___ linkages that ___ be digested by animals. a α1 --> 4 glycosidic ; can b α1-->…
A: Glycogen is the storage sugar in animals. It is composed of units. There are two types of…
Q: 8. Write the systematic name for each glycosidic bond. HO CON OH 6-CH₂ OHHO OH 8-CH₂ O OH HO OH…
A: Glycosidic bonds are covalent bonds which connect monosaccharides to form disaccharides and…
Q: You are sprinting around a track and at about the 200 meter mark you start to feel a burning…
A: Fermentation is defined as an anaerobic process in which even during absence of oxygen, energy can…
Q: Under low tryptophanyl-tRNA (tRNAtrp), we expect the trp operon to be expressed because: Question…
A: Trp operon functioning in the bacterial system Escherichia coli consists of a set of genes that…
Q: Genetics Question 18
A: The question is asking whether the number of chromosomes in the cells produced at the end of meiosis…
Q: 2. Lactate dehydrogenase (LDH) catalyzes the reaction Ο ()) 0 NADH + H* NAD+ C=0 HO-C-H CH₁t…
A: Lactate dehydrogenase catalyzes the interconversion of pyruvate and lactate with concomitant…
Q: Tube # 2 3 4 6 5. THE EFFECT OF TEMPERATURE Temp. Abs. 0°C N/A 0°C 25°C 25°C 37°C 37°C 70°C 70°C 10…
A: Changes in temperature can affect the enzyme in different ways. When the temperature of the system…
Q: Draw the structure of phosphatidylcholine at pH 7 with the moiety on R₁ as 14:0 (C14) and R₂ as…
A: Glycerophospholipids, membrane lipids in which two fatty acids are attached in ester linkage to the…
Q: Genetics Q4
A: The objective of the question is to find the sequence of the opposite strand of a given DNA strand.…
Q: Genetics 8 Q4
A: The question is asking whether the process of meiosis, which is the division of a germ cell…
Q: serine proteases act via a two-step catalytic mechanism. However, as a critical scientist, you may…
A: Serine proteases are protein cleaving enzymes that have Serine, Histidine and Aspartate (the…
Q: Since two 19S regulatory subunits and a 20S catalytic unit constitute the 26S proteasome, why is the…
A: A peptide is a short chain of amino acid residues linked together via peptide bonds. A folded…
Q: Hyperacetylation of histone tails is often associated with: Question 22 options:…
A: Loose conformation of Chromatin Explanation:Histone connections with DNA become weaker by…
Q: Question #4 The graph on the left shows data for an experiment in which isolated mitochondria were…
A: The process of oxidative phosphorylation, which mitochondria use to produce energy, is essential to…
Q: What is the product of CH₂OH H-C-OH HO-C-H H-C-OH H-C-OH CH₂OH COOH H-C-OH HO-C-H I H-C-OH H-C-OH…
A: This is an example of reduction of a sugar with NADH/enzyme. The reaction of glucose with NADH is a…
Q: graph the data from the table after taking recipocals of [S], V without inhibitor and V with…
A: Plot between the reciprocal of V (velocity of reaction) and S (concentration of substrate) is known…
Q: please explain the question, i am unable to input values of x
A: The dependent variable, the reaction rate (µmol/L/min), is usually plotted on the y-axis and is…
Q: MSA: 1. Explain the incubation conditions 2. Explain the reagents being added 3. Explain the…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: I have glucoseoxidase 1000 Udl-1. What is the exact unit of the enzyme that could work for 1…
A: Specific activity of glucose oxidase (from literature) : 200 units/mg protein Calculation: First,…
Q: I need answer expert solutions please
A: Certainly! Let's break it down:1. Composition: The disaccharide consists of two glucose molecules…
Q: You need to make a protein buffer of: 100 mM NaCl ⚫ 25 mM Tris 8 • • 5% w/v glycerol 2 mM DTT Your…
A: In order to calculate how much of each component we should use to get the required buffer, we need…
Q: Label each Amine (A–D) in Table 1 as primary, secondary, or tertiary. Which classes of amines –…
A: Good evening,Hope this helps, Thank you!Explanation:Approach to solving the question: Detailed…
Q: [AktivGrid] Draw the product of the reaction of isocitrate catalyzed by isocitrate dehydrogenase in…
A: Isocitrate dehydrogenase catalyzes the irreversible decarboxylation of isocitrate to yield…
Q: If instead of using the pyruvate for gluconeogenesis, the pyruvate entered the mitochondria, what…
A: The question states that if pyruvate enters the mitochondria instead of being used for…
Q: If someone's pH increases above 7.8, they die. How much would the % buffer have to shift from normal…
A: The usual pH range for blood is 7.35 to 7.45. The body's buffering systems, such as the phosphate…
Q: pless dont proved hand ratting solution
A: Step 1:The compound is written in Newman Projection. The circle indicates that it is projected in…
Q: 48. A 58-year-old woman comes to the office because of a 1-month history of fatigue, fever,…
A: The objective of the question is to identify the cell type that is most likely being directly…
Q: 2.13 Calculate the reaction enthalpy, entropy, and free energy, AH°r, AS°r, and AGºr, for the…
A: Reaction thermodynamics is the energy things that occur during a chemical reaction together with a…
Q: The anticodon _______ can form a Wobble interaction with 5’ UAG 3’? Please note the directionality…
A: Codon is defined as the combination of three ribonucleotides of mRNA. These are complementary to…
Q: why do so many pharmaceutical drugs contain nitrogen? How would an NH2 group benefit this dimer…
A: The objective of the question is to find the reason why so many pharmaceutical drugs contain…
Step by step
Solved in 2 steps with 1 images

- Combustion of a fuel sample in a bomb calorimeter increases the temperature of the entire system by 5.10 °C if the calorimeter contains 1700 g of water, but only by 4.00 °C if the calorimeter contains 2200 g of water. What is the heat capacity of the dry bomb calorimeter assembly? Assume that the specific heat capacity of water is 4.18 J g–1 °C–1.In kool aid chromatography, what factors affect the efficiency of the column? Also discuss the Van-Deemter equation.Explain briefly effects and importance of following phenomena on bioreactor design a) oxygen demand b) foam production c) shear and viscosity d) growth rate e) pH
- My data is attached, please 1.) Create a scatter chart with all the data then **Using your graph - predict how many seconds it will take to dissolve an Alka-Seltzer® tablet in 200 mL of water at the following temperatures. Use the exponential trend line to find the function used in predicting the time. If needed, please refer to the Introduction to Graphing manual for guidance. 15°C, 35°C and 60°Ca. State TWO (2) advantages of Inductively coupled plasma-optical emission spectroscopy (ICP-OES) over Flame atomic absorption spectroscopy (FAAS).b. What are the TWO (2) advantages of graphite furnace AAS (GFAAS) over flame AAS (FAAS)?c. What is one of the main disadvantages that most flames used in AAShave?1) what is the equilibrium constant for the reaction A + CT AT+ C? Show calculations. 2) the image
- The column efficiency is constant for a column that uses a cons tant eluent composition. What does this say about the peak width of slowly eluting compounds relative to those of rapidly eluting compounds?Discuss the limitations of the lineweaver-burk plot and suggest alternative secondary plots and compare and contrast their advantages/disadvantages in analysis of kinetic data.When a mixture of various molecular sized dyes are run through a gel chromatography column, what is eluted in the Void Volume? A. Blue Dextran B. Flourescein C. Both D. Nothing
- A chemist is performing a precipitation titration on a 1.00 L sample containing an unknown concentration of the cadmium ion, Cd2+. If it takes 24.52 mL of a 0.250 M solution of Na2CO3 to reach the end point, calculate the concentration of the cadmium ion?Calculate the enthalpy of combustion for ethanol using bond energies and compare it with the other values.Thank you for helping me. I have to questions. In solution B, if we choose two other points on the graph for calculating the slope, wouldnt the values for Km/Vmax be different than 6.1? Why did you choose exactly those points on the graph? Another thing, I understand the method you used to get to Km/Vmax= slope, but how did you then get the value for Vmax to be 10 mM/s??



