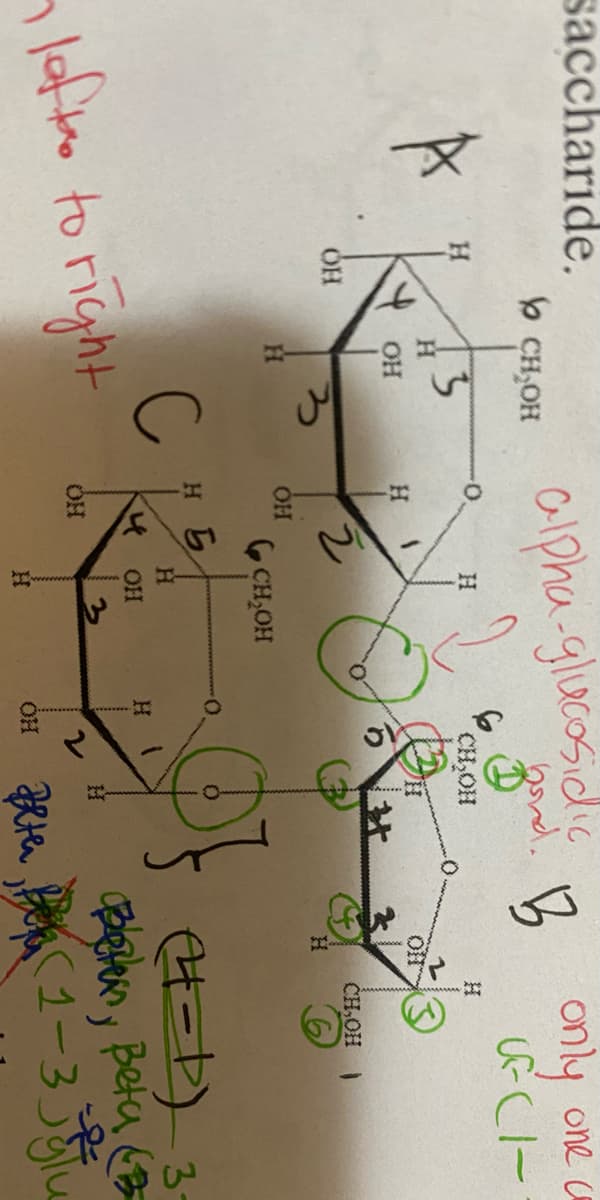
Is B monomer a beta fructose or alpha fructose. How do you know?
Q: true or false: A high dTTP concentration shifts the specificity of ribonucleotide reductase…
A: Ribonuclotide reductase is the enzyme that catalyses the conversion of NTPs to dNTPs. Ribonuclotide…
Q: In animal tissues, the ratio of active, unphosphorylated to inactive, phosphorylated pyruvate…
A: Cellular respiration is a collection of three metabolic pathways that generate ATP the energy…
Q: Biological value of cerebrosides and gangliosides.
A: The glycosphingolipid family includes the cerebrosides, which are crucial parts of the membranes of…
Q: Over the course of glycolysis and the citric acid cycle, there are 10 NADH and 2 FADH₂ produced per…
A: Number of ATP formed from each molecule of NADH and FADH2 through oxidative phosphorylation helps in…
Q: why is enzyme efficiency represented by kcat/Km rather than Vmax
A: Enzyme kinetics is the study of the rates of chemical processes that are catalysed by enzymes. The…
Q: An infant was admitted to hospital for failure to thrive. It was discovered that the infant lacks…
A: Saturated fatty acids undergo beta oxidation in the mitochondria. A fatty acids with n carbons…
Q: A total of 2.5 x 105 cells were harvested from the patient. You need the cells to fill a scaffold…
A: For calculation of volume we need to know the basic formula of l×b×h
Q: Why does glutamate the only amino acid used in oxidative deamination
A: Glutamate is an acidic amino acid that acts as the only amino acid used in oxidative deamination.…
Q: The key regulatory enzymes in glycolysis are: A) hexokinase, phosphofructokinase, and pyruvate…
A: In the anaerobic process of glycolysis, one glucose molecule is transformed into two pyruvate…
Q: Which of the following statements is true about the quaternary structure of protein? A) The…
A: There are 4 different types of proteins structures: 1. primary: It is the sequence of amino acids…
Q: Scheme of galactose catabolism. Enzymopathies
A: Galactose (Gal), also known as "milk sugar," is a monosaccharide sugar that is about as sweet as…
Q: Age-related macular degeneration (ARMD) within the eye is a disease that is closely related to…
A: Age-related macular degeneration (ARMD) is an ocular disease causing damage to the retinal macula,…
Q: 3. Why can DNA adopt both A- and B- forms, while RNA is restricted to the A-form?
A: DNA is two strands of polynucleotide linked to each other in an antiparallel direction by hydrogen…
Q: 1 what is the common intermediary in the catabolism of carbohydrates, proteins and lipids? 2 What…
A: Metabolism is the process of breakdown of food into energy. Carbohydrates, proteins, and lipids are…
Q: draw a guanine nucleobase and label all possible H bond donor and H bond acceptor?
A: H bond donor are those groups containing a H bonded to a electronegative atom (F,O,N) and H bond…
Q: Determine how many water molecules are used to completely oxidize Lignocerate (24:0) 8 10 014 011 9…
A: Fatty acids are long chain hydrocarbons with a carboxyl end. Glycogen is the storage form of…
Q: Enzymes increase the rate of a reaction by which contributing factor? - Increasing Keq -Formation…
A: Enzymes as biocatalysts catalyse the biochemical reactions by decreasing the activation energy so…
Q: Describe the mechanism for moving acetyl-CoA produced in the mitochondrial matrix into the cytosol…
A: An enzyme called acetyl CoA carboxylase in the cytoplasm catalyzes the carboxylation of Acetyl CoA…
Q: 1 the overall structure of DNA is double helical and the bases between each strand form watson-…
A: DNA or deoxyribonucleic acid is the fundamental component of all cells. It constitutes the genetic…
Q: Review what is enzymes/catalysts? How do you increase/ decrease the reaction? What happens to the…
A: Introduction: Enzymes are proteins in nature and their action is specific. It functions…
Q: What is the name of this cofactor related to niacin? 0
A: Enzymes are proteins that catalyse biochemical reaction. Sometimes enzymes require a non protein…
Q: Citric Acid Cycle Notes? C² 1000 K
A: In aerobic condition, pyruvate in the presence of pyruvate dehydrogenase complex produces Acetyl…
Q: Identify which of the following pairs of amino acid residues can have hydrogen bonding between their…
A: Amino acids are the building blocks of proteins. The structure of protein is maintained by many non…
Q: Which of the following statements regarding CoQ is false a. It resides in the inner…
A: ETC consist of four protein complexes called Complex I, II, III and IV that transport electron from…
Q: Urea is an ideal way to remove ammonia from the body because: OA) it does not affect the pH of…
A: Metabolism generates free ammonia.ammonia is toxic to cells so it must be eliminated from the cells…
Q: Tell how many ATPs result from from oxidative phosphorylation of one NADH; tell how many ATPs result…
A: Oxidative phosphorylation is oxygen dependent cellular process to generate energy in terms of ATPs…
Q: Can all abnormal hemoglobin be diagnosed by electrophoresis ?Explain why or why not .
A: Electrophoresis means migration of charged particles under the influence of an electric field.…
Q: a. What is the name of metabolite 1? b. What is the name of metabolite 2? c. What molecule a is…
A: Purines are heterocyclic organic compound that has 5-member rings fused to a 6-member ring.…
Q: Which of the following is a substrate from primase?
A: DNA/RNA are nucleic acids, the molecules responsible for carrying genetic information from one…
Q: How much ATP would be generated by having one molecule of oxaloacetate being completely oxidized to…
A: In glycolysis, a 6-carbon molecule of glucose-6-phosphate is broken down into 3-carbon pyruvate by…
Q: In extracellular proteins the amino acid Cys can interact with another Cys to form a bond sometimes…
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: True or False: Passive-mediated transport proteins lower the delta G of transport to create the…
A: Introduction :- The question is all about transport of molecules by diffusion I. e. Active and…
Q: Structure and biological role of phosphatidic acid.
A: The smallest and most basic phospholipid, phosphatidic acid (PA), serves as a precursor for other,…
Q: Meselson-Stahl Experiment showed that DNA replication is semi-conservative. In the experiment, DNA…
A: DNA replication is the biological process by which two identical replicas of DNA from one original…
Q: Which of the following statements concerning insulin is NOT true? a. Insulin can increase glycogen…
A: Glycolysis is the metabolic process that breaks down glucose into pyruvate. Gluconeogenesis if the…
Q: 1. You order two primers from a company that synthesizes oligonucleotides. The primers have the…
A: Primers are short stretches of nucleotides that are used to initiate the synthesis of nucleic acids.…
Q: Discuss how the shuttle mechanisms for cytoplasmic NADH operate in eukaryotes.
A: Most textbooks mention that under aerobic conditions, NAD+ is regenerated in the ETC. But the…
Q: What must happen first in order for dietary fats (triacylglycerols) to be digested? REME A) They…
A: Unlike carbohydrates and protein, lipids are a class of biological macromolecules that are not…
Q: The B-oxidation of the saturated 14-carbon fatty acid myristic acid yields the following NET amount…
A: In beta oxidation, large fatty acids that have been transformed into acyl-CoA chains are gradually…
Q: In its non-phosphorylated state, glycogen phosphorylase can be activated by which of the following…
A: Glycogen phosphorylase is the regulatory enzyme of the glycogenolysis pathway. Glycogenolysis is the…
Q: 16. The overall reaction for the glycolysis reaction is C6H₁2O6(aq) + 2NAD+ (aq) + 2ADP³- (aq) +…
A: Glycolysis is the breakdown (catabolism) of glucose into pyruvate with net generation of 2ATP and…
Q: Complex 2 oxidizes ___ and reduces __ a. FAD, COQ b. COQ , Cytochrome C C NADH, COQ d.…
A: Aerobic metabolism of 1 molecule of glucose can produce 10 NADH (6 from acetyl CoA in TCA cycle + 2…
Q: does BPG bind to deoxyhemoglobin only?why BPG does not bind to oxyhemoglobin? what is chemical…
A: Hemoglobin is a globular protein, ie it is roughly spherical. It is a tetramer of two types of…
Q: 1 Draw the following polypeptide: Met- Tyr-Val-Ser-Asn 2A Draw the hydrolysis of a dipeptide…
A: A peptide is a chain of amino acids, in which the amino acids are joined together through peptide…
Q: *While acetyl-SCOA activates pyruvate carboxylase, it inhibits pyruvate kinase. Explain how this is…
A: Complementary control is a type of metabolic regulation by which 2 complementary pathways are…
Q: BIOC 385 Biochemistry of Protein Synthesis Q11.4: Considering that binding of the correct tRNA to…
A: Translation is the process of protein synthesis. It occurs in the cytoplasm. Ribosomes have peptidyl…
Q: The mismatch DNA repair mechanism is based on the fact that: O a. O b. O C. O d. The parental DNA…
A: Mismatch repair is a process in which mismatched nucleotides present in the complementary paired…
Q: The term that refers to the light-dependent process in plants in which O₂ is consumed and CO₂ is…
A: Plants need energy to perform the functions that keep them alive, just like all other living…
Q: A patient with chronic pancreatitis and hence decreased secretory function of pancreas was…
A: Individual having an inflammation in the pancreas which does not improve over a long period of time…
Q: In term of the sugar identity , what is the difference between DNA and RNA. 2 What nucleobases are…
A: Deoxyribonucleic acid (DNA) and ribonucleic acid (RNA) are two of the most important molecules in…
Step by step
Solved in 2 steps with 1 images

- Energetic of Fructose-1 ,6-bis P Hydrolysis (Integrates with Chapter 3.) The standard free energy change (G) for hydrolysis of fructose-1. 6-bisphosphate (FBP) to fructose-S-phosphate (F-6-P) and P: is -16.7 KJ/mol: FBP + H2O fructose-6-P + Pi The standard free energy change (G) for ATP hydrolysis is -30.5 KJ/mol: ATP + H2O ADP + Pj What is the standard free energy change for the phosphofructokinase reaction: ATP + fructose-6-P ADP + FBP b. What is the equilibrium constant for this reaction? c. Assuming the intracellular concentrations of [ATP] and (ADP] are maintained constant at 4 mM and 1.6 mM, respectively, in a rat liver cell, what will be the ratio of [FBP]/[fructose-6-P] when the phosphofructokinase reaction reaches equilibrium?BIOMOLECULES - MULTIPLE CHOICE - Please answer properly QUESTION : The coenzyme biocytin is associated with which of the following vitamins? A. pyridoxine B. thiamine C. biotin D. nicotinic acidTrue or False e. Triacylglycerols and glycerophospholipids both contain fatty acids and saponifiable. True False f. Pyruvate serves as the precursor of the glycerol backbone during TAG biosynthesis. True False
- Briefly explain why chicken fat, which largely consists of various triacylglycerols, has such a higher energy content per mole compared to sucrose.-CHOOSE THE CORRECT LETTER- 1.What major metabolic substrate do ketone bodies convert into so that they enter the metabolic pathway?A. pyruvateB. acetyl-CoAC. c02D. oxaloacetateCompare and contrast glycogen synthesis/degradation in muscles as compared with the liver.
- Assignment on KetohexosesConstruct the structure of the following enantiomers or mirror images:1. D and L Fructose2. D and L Allulose3. D and L Sorbose4. D and L TagatoseRole of Carnitine in fatty acid β-oxidationPalmitoleic acid, 16:1Δ⁹ hexadecaenoic acid, (16 carbon FA with one double bond )is an important fatty acid component of TAGs and cell membranes. Briefly explain the process of beta oxidation of this fatty acid and the number (only) of FADH, NADH and acetyl CoA outcome. What is the total ATP (only number) generated from this fatty acid after beta oxidation.



