istory Bookmarks People Tab Window Help ** 1ON Wed 21 139 W2 X Chapter 14 Lecture Two or x O Intermolecular Forces and xG perodt sture.com/courses/1885023/quizzes/4613315 th charge X Consider the molecules shown below then answer the following questions. eject descriptie n.decx HC-CH2 H;C H:C. CH; H;C CH2 H,C-CH2 НС HC D. A. Which of the following molecules has the strongest attraction to a moleculełike itself? D B. What is/are the major type of intermolecular forces that are involved in all of the given molecules? Dispersion forces C. A container filled with which of the following molecules will have the highest vapor pressure? C D. A container filled with which of the following molecules will have the highest boiling point? A MAR MacBook Pro FB F7 F6 F5 F4 F3 & 8 %23 4 т R.
istory Bookmarks People Tab Window Help ** 1ON Wed 21 139 W2 X Chapter 14 Lecture Two or x O Intermolecular Forces and xG perodt sture.com/courses/1885023/quizzes/4613315 th charge X Consider the molecules shown below then answer the following questions. eject descriptie n.decx HC-CH2 H;C H:C. CH; H;C CH2 H,C-CH2 НС HC D. A. Which of the following molecules has the strongest attraction to a moleculełike itself? D B. What is/are the major type of intermolecular forces that are involved in all of the given molecules? Dispersion forces C. A container filled with which of the following molecules will have the highest vapor pressure? C D. A container filled with which of the following molecules will have the highest boiling point? A MAR MacBook Pro FB F7 F6 F5 F4 F3 & 8 %23 4 т R.
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter11: Intermolecular Forces And Liquids
Section: Chapter Questions
Problem 51SCQ
Related questions
Question
Chemistry Help. Cant get the right answer I'm stuck. Thank you.
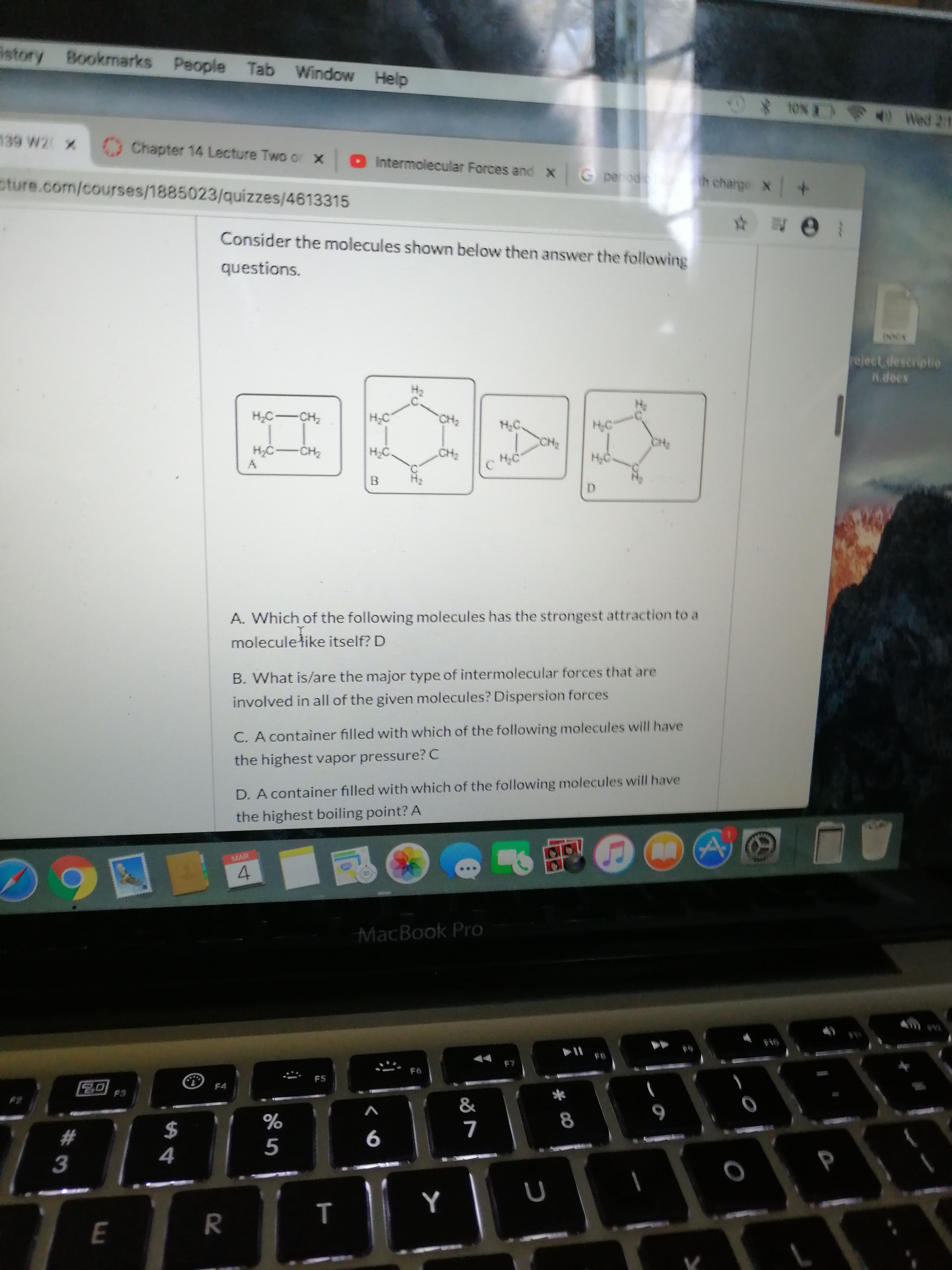
Transcribed Image Text:istory Bookmarks People Tab Window Help
** 1ON Wed 21
139 W2 X
Chapter 14 Lecture Two or x O Intermolecular Forces and xG perodt
sture.com/courses/1885023/quizzes/4613315
th charge X
Consider the molecules shown below then answer the following
questions.
eject descriptie
n.decx
HC-CH2
H;C
H:C.
CH;
H;C
CH2
H,C-CH2
НС
HC
D.
A. Which of the following molecules has the strongest attraction to a
moleculełike itself? D
B. What is/are the major type of intermolecular forces that are
involved in all of the given molecules? Dispersion forces
C. A container filled with which of the following molecules will have
the highest vapor pressure? C
D. A container filled with which of the following molecules will have
the highest boiling point? A
MAR
MacBook Pro
FB
F7
F6
F5
F4
F3
&
8
%23
4
т
R.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning