Q: Exp. 2:Determine the Enthalpy of Neutralization of HCI by NH3 Temp of empty calorimeter= 21.5C, calo...
A: a.) q (heat) can be calculated using formula : q = m × C × ∆T b.) ∆H for a reaction is written i...
Q: ature (Tg) is the temperature range where a polymer chain gains segm y of the polymer chains depends...
A: The glass transition temperature (Tg) is the temperature at which a polymer turns from a ductile mat...
Q: Lab. 7 1. ___medium and _____reagent are used to detect indole production. _____will react wit...
A: Ans. 1. medium : The medium used with this reagent is either motility- indole-ornithine agar, brot...
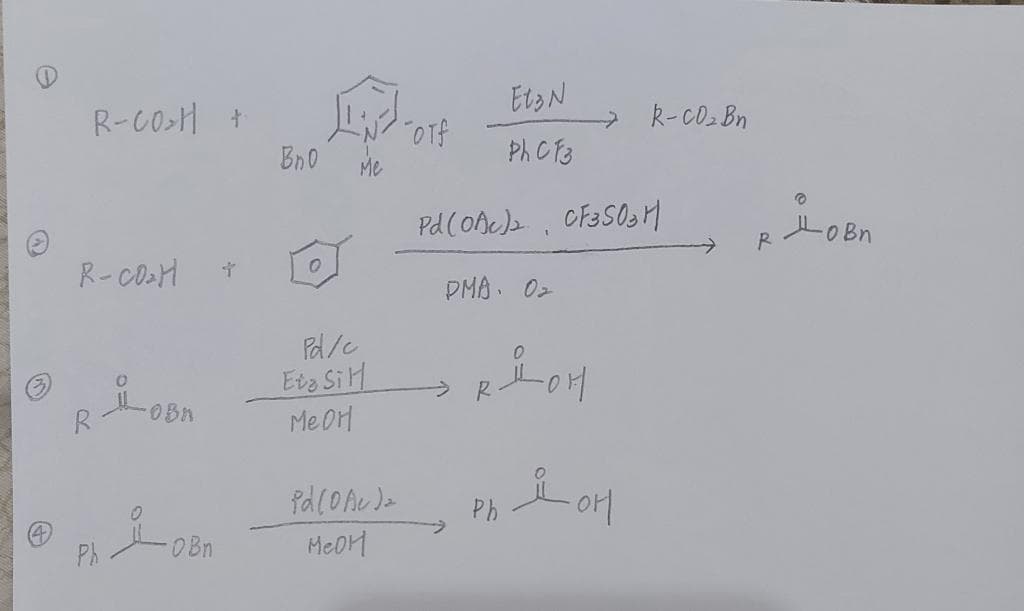
Q: Select the best answer for the most reasonable synthesis of the target molecule below from ethyl ace...
A: Here we have to predict the most reasonable synthesis method to convert ethyl acetate into 3-methylb...
Q: What is the chemical formula for a colorless, pungent gas that acts as a powerful preservative and d...
A: Preservatives: Preservatives are chemicals that are used to prevent the decomposition of food or bio...
Q: In acidic solution: 1. PbO2 + I2 → Pb2+ + IO3 Oxidation half-reaction: Reduction half-reaction: Comp...
A:
Q: Identify two (2) unwanted sources of heat loss and/or gain that may have contributed to the error in...
A: In calorimetry it is assumed that the whole amount of heat energy being released by the hot body is ...
Q: Suppose the galvanic cell sketched below is powered by the following reaction: Mn(s)+PdSO4(aq) MnSO,...
A: We have given a reaction- Mn(s) + PdSO4(aq)>>> MnSo4(aq) + Pd(s) The answers and explanatio...
Q: Problem #1: An electrochemical cell contains Fe3+ and is measured against a Ag/AgCl reference electr...
A: Given : Ecell = 0.703 V Since Ag/AgCl is reference electrode and present in standard condition so ...
Q: When a new chirality center is created, isomers can result. For the reaction below, draw all of the ...
A: -> NaCN has -CN nucleophile which can give addition reaction to carbony center which is electroph...
Q: Acid Names IUPAC HCN(aq) Classical Acid Names IUPAC HCIO2(aq) Classical Acid Name IUPAC CH3COOH(aq) ...
A: IUPAC name is the systematic name given to a chemical compound International Union of Pure and Appli...
Q: Which statement is correct? Dissolving table salt (NaCI) in water leads to a decrease in entropy bec...
A: Correct option is (C)
Q: Calculate the entropy change of the surroundings when 15.2 g of solid iodine sublimes at 25.0 °C. AH...
A:
Q: A chemistry graduate student is given 300. mL of a 1.30M diethylamine ((C,H5), NH) solution. Diethyl...
A:
Q: If 100.0 mL of potassium dichromate solution (10.0 g of K2Cr207 per liter) was mixed with 75.0 mL of...
A: Given : Volume of K2Cr2O7 = 100 ml = 0.10 litre Concentration of K2Cr2O7 = 10.0 g/litre Volume of ...
Q: D.I.2 Elechophilic Ar omatic fubs ( SEN)[ Comc eptual] CH3 C3 mol) ON ON Reactin i HンS0+ NOL ZON- CH...
A:
Q: Part A A current of 11.4 A is applied to 1.25 L of a solution of 0.557 mol L-1 converting some of th...
A: Given: Current = 11.4 A. Time passed = 78 min = 78 × 60 = 4680 s (si...
Q: For the central atom in each formula, draw the Lewis Structure with all valence electrons shown. 1. ...
A:
Q: For A → products, time and concentration data were collected and plotted as shown. [A] (M) t (s) 0.5...
A:
Q: Draw Lewis structures for bromine nitrate (BRONO2) and bromine monoxide (Bro). (Draw all major reson...
A: Lewis structure of Bromine nitrate:
Q: एकचर्चर नेकट ा ४ mssme missmg greagend ठेवतत A)1. Li Al Hy 2 द00 anके Bar (2 09) 2. H301 १. ५३० CH3I...
A: Organic reactions are those in which organic reactant react to form organic products. In this reacti...
Q: how many millimeters of 0.786M of sulphuric acid are needed to reduce a total of 0.345 of barium hyd...
A: Sulphuric acid reacts with barium hydroxide to form barium sulfate and water. H2SO4 + Ba(OH)2 ----&g...
Q: Provide one example for SN2 reaction of alkyl halide
A:
Q: Milk of magnesia is a suspension of magnesium hydroxide in water. Although Mg(OH)2 is relatively ins...
A:
Q: Lewis Dot Formulas for atoms: display the __________ electrons as dots around the chemical symbol.
A: Lewis Dot Formulas for atoms: display the Valence electrons as dots aroundthe chemical symbol. In Le...
Q: In each row, pick the compound with the bigger lattice energy. Note: lattice energy is always greate...
A:
Q: Four of the following five sets of quantum numbers are not allowed. Choose the a 4 3 -4 +1/2 in m 1 ...
A: Given : We have to tell which of the following is allowed set.
Q: A student measures the S2- concentration in a saturated aqueous solution of cobalt(II) sulfide to be...
A:
Q: Using photoelectron spectroscopy, the ionization energy of the least tightly bound valence electron ...
A: Given, Ionisation energy = 5.40 eV Electronic configuration of Li = 1s22s1 Principal quantum numbe...
Q: Sodium lauryl sulfate has the same use in embalming fluids as 1-sodium phosphate 2-citrates 3-su...
A: Sulfonates
Q: Part A etal plating is done by passing current through a metal solution. or example, an item can bec...
A:
Q: A student attempts to determine the buffer capacity of a solution by titrating 40.0 mL of solution w...
A: Given: Volume of buffer solution = 40.0 mL = 0.040 L (since 1 L =...
Q: Q2: OH OH Br Prepare CH;CH,CH-CHCH3 starting from CH;CH,CH-CH2CH3 And any other needed reagents.
A: We have to prepare given diol from alkyl halide.
Q: HIT-NO=15641 SCORE= (P-METHYLPHENOXY ) ACETIC ACIO SDBS-NO=31558 IR-NIDA-47630 : NUJOL MULL LOD SD 4...
A: (p - methyl phenoxy)acetic acids IR spectrum has some characteristic peaks as listed in the followin...
Q: QUESTION 8 The valence electron configuration ns-np corresponds to which group on the periodic table...
A: 8) Given general configuration--- ns2np5
Q: How many moles of sodium hydroxide would have to be added to 125 mL of a 0.339 M hypochlorous acid s...
A:
Q: An attempt was made to dissolve 5.893 grams of PbCl2 in 1.000 liters of water. The Ksp of PbCl2 is 1...
A:
Q: What simple characterization techniques can you say that your product is dibenzalacetone?
A: Characterization techniques are the methods to identify, analyze the chemical substance and determin...
Q: Under which conditions would you expect argon to behave ideally? In 150 K, 100 kPa or in 400 K, 100 ...
A: Generally a gas behave like ideal gas when intermolecular forces are less significant compare to kin...
Q: NaOH A 400 'C
A: The reaction given is,
Q: A possible set'of quantum numbers for an electron in the partially filled subshell in a potassium at...
A: Quantum numbers are the set of numbers which describe energy and position of electrons in atoms .
Q: Furnace 02 H,0 absorber CO, absorber Sample When 2.905 grams of a hydrocarbon, C,Hy, were burned in ...
A:
Q: predicting the IR spectrum for the product with specific absorbtion bands and for specific funstiona...
A:
Q: Calculate the half-life time & rate constant from the information below , the reaction is 1st order ...
A: For a first order reaction, A---------> Products Differential rate law is, Rate ...
Q: NH3 is more volatile than the ionized form, NH4+. The pKa of NH4+ is 9.3. Which pH leads you to have...
A:
Q: Unknown # Draw the flow chart for this experiment and record you observations for Use two different ...
A:
Q: Benzoic acid, HC6H5CO2, is a monoprotic acid (only one H* ionizes) with a Ka = 6.5 × 10-5. Calculate...
A: In this question, we have to find the [H+] and pH of a 0.016M solution of benzoic acid having Ka = 6...
Q: Three students are preparing alkoxides in the lab by mixing sodium metal with a suitable alcohol. St...
A: The rate of the formation of the product or the rate of disappearance of the reactants is known as t...
Q: Keeping pressure (P) and number of moles (n) constant, what happens to the temperature (T) if the vo...
A: Given:: V1= V V2=2V T1= T T2= ?
Q: When a new center of chirality is created, isomers can result. For the reaction below, specify the k...
A: A chiral centre is the carbon attached to four different atoms or group of atoms. A racemic mixture ...
Step by step
Solved in 2 steps with 2 images

- A monolayer of CO molecules is adsorbed on the surface of 1.00 g of an Fe/AI2O3 catalyst at 77 K, the boiling point of liquid nitrogen. Upon warming, the carbon monoxide occupies 4.25 cm3 at O oC and 1.00 bar. What is the surface area of the catalyst?Under what conditions does the extended Debye-Huckel law, equation 8.52, become the Debye-Hckel limiting law?Show all work pls
- 10.11/ A 1.5-kg specimen of a 90 wt% Pb-10 wt% Sn alloy is heated to 250°C (480°F), at which temperature it is entirely an a-phase solid solution . The alloy is to be melted to the extent that 50% of the speci- men is liquid, the remainder being the a phase. This may be accomplished either by heating the alloy or changing its composi- tion while holding the temperature con- stant. (a) To what temperature must the speci- men be heated? (b) How much tin must be added to the 1.5-kg specimen at 250°C to achieve this state? 10.15) A magnesium-lead alloy of mass 5.5 ka consists of a solid a phase that has a compo- sition that is just slightly below the solubility limit at 200°C (390°F). (a) What mass of lead is in the alloy? (b) If the alloy is heated to 350°C (660°F). how much more lead may be dissolved in the a phase without exceeding the solubility limit of this phase? 10.27 A hypothetical A-B alloy of composition 55 wt% B-45 wt% A at some temperature is found to consist of mass…Show all steps leading to the final answer po. Here’s a pdf file in accordance with the topic po: https://drive.google.com/file/d/1_FnDtXCrFKSol3RNWIG_9tNQ7IxgxD6t/view?usp=drivesdkBasis: https://youtu.be/Y4NMpO1xI8U
- https://m.youtube.com/watch?v=vM1SP346XBc&list=PLeJOSNLNZfHubfLdq0kOayASeUllMOGn4&index=4 I watched this the lecture video over and over and I am allowed to work with someone but I am having trouble with part B and I provided the YouTube link of the data or video attached to this labDetermine the boiling point of CS2, in °C, from the following data: CS2(g) CS2(l) ΔHf° / (kJ mol–1) 115.3 87.9 S° / (J mol–1 K–1) 237.8 151.0 Answer Options: 258 -14 116 78 43General Chemistry (Quesion -2) =====================



