Q: C. A +B e. a. d. C+D Progress of the Reaction 55) Which of the following terms best describes the fo...
A: We are to discuss what type of reaction is taking place in the given graph.
Q: Write the expression for the solubility product constant for each of the following a) Pb(NO3)2 b) ...
A: Note: As per our guidelines we are supposed to answer first 3 parts please repost the other parts as...
Q: Consider CH3-CH(OH)-CH(OH)(Br). a.How many stereogeniccenters are in the molecule? b.How many ster...
A: Stereoisomers are 3D representations oF molecule in space . It is shown on paper by Fischer projec...
Q: Find the total volume of atoms in this unit cell expressed in terms of "r". To do this, multiply the...
A:
Q: 5. Given the following percent composition and molecular weight determine the correct formula of eac...
A: Empirical formula :- A chemical formula with the shows only the relative number of atoms of differe...
Q: Consider the given Orgel diagram for d', d', d°, and d° ions in octahedral and tetrahedral fields, w...
A: 1) d1 and d6(high spin) configurations are equivalent.
Q: Acid anhydrides react with alcohols to give esters and carboxylic acids. Consider the formula of ace...
A: When two molecules of acetic acid reacts with each other then acetic anhydride is formed through the...
Q: The Haber Process synthesizes ammonia at elevated temperatures and pressures. Suppose you combine 15...
A: We have to calculate the volume of Reactant left.
Q: A sample of 10 ml of a solution of KBr has a mass of 12.60g. When the solution evaporates, the resid...
A:
Q: The chemist plans to try to identify the unknown liquid by measuring the density and comparing to kn...
A: Density of a liquid is represented by the mass of that liquid per unit volume of the liquid. ...
Q: CHEMICAL EQUILIBRIUM 1. Calculate the pH of the solution of 0.026M ammonia and what is its percent ...
A: Since, you have asked multiple question, so I will solved first question for you. You can see detai...
Q: What is the correct chemical name for Cu2O ? Copper oxide Copper (II) oxide Copper oxalate Copper (1...
A:
Q: Question Select your answer. Covalent compounds particles are held by ionic bonds while ionic compou...
A: Melting point is the temperature at which a solid melts and converts into liquid form.
Q: The best balanced chemical equation for the complete combustion of: 1. hexane 2. nonane
A: The best balanced chemical equation for thecomplete combustion of:1. hexane2. nonane
Q: H30: solved in CDCI3), shown below. What percentage of the total integrated spectrum is represented ...
A: Nuclear magnetic resonance is commonly known as NMR spectroscopy or Magnetic resonance spectroscopy....
Q: when necessary. a) b) Br2. H20 HCI d) H2 Ni MCPBA 1) ВН; THF 2) H2O2 NaOH f) H2SO4, HgSO4 H3O*
A:
Q: What would the growth curve if you grew E. coli at 5% dissolved 02 in solution? Explain your notatio...
A: Answer -
Q: Problem 3: Using curly arrows, draw both an acid- and a base-catalysed mechanism for the reaction be...
A: with alpha H caronyl compounds in presence of dilute base (or) acid gives beta-hydroxy carbonyl co...
Q: What would happen to the pH of your soap if you used 300 mL of saturated salt solution rather than t...
A: Carboxylic acids react with strong bases such as sodium hydroxide to produce carboxylate salts in ne...
Q: Draw the form of the following compound that will predominate at pH 13. pKa 1.84 NH2 pKa 9.13
A: We have to predict the structure of given Compound at pH 13
Q: Determine the indices for the planes in the cubic unit cell shown. (a) (b) (c) 2/3 (0,4/3,1) 1/2 1/3...
A: For determine Miller Indices- Determine the intercepts of the face along the crystallographic axes...
Q: What is the correct IUPAC name for the following compound? Br 3-bromo-2,3-dimethyl butane 2-bromo-2,...
A: Write IUPAC NAME OF THE GIVEN STRUCTURE--
Q: When a strong base titrant (e.g. NaOH) is added, the following reactions occur: (iv) H2A(aq) + OH(a...
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for you...
Q: When classifying, choose one or more of: combination, decomposition, replacement, ion exchange, and ...
A: Concept - 1) A combination reaction is a reaction where two or more reactants combine to form a sing...
Q: The oxygen mass transfer coefficient in fermentation vessels is determihed TPom measurements using t...
A: Given, Cal' =0.25 mol.m^-3 with uncertainty 4%, Cal = 0.183 mol.m^-3 with uncertainty 4%...
Q: Practice Problem 2.1 species by writing in the box provided. (1 point each) or a) Fe* protons electr...
A: We are authorised to solve only one question at a time. Please post rest of the questions separately...
Q: d. Below is a body-centered unit cell, write the letter "a" along the edge length. Calculate the len...
A: Bcc is a structure lattice in which atoms are present at the corners of the cube and at the body cen...
Q: 50 ml of HNO3 require 35.50mL of Ba(OH)2 0.159M to neutralize.Determine the molarity of the acidic s...
A: In a neutralization reaction an acid reacts with a base to form a salt and water. In a neutralizati...
Q: 3. Please write out the products or reagents (Note: stereochemistry ) H3C Br2 H3C Br/H20 (2) (1) H3C...
A:
Q: How many grams of Fe is needed to react with 35.0 g of sulfuric acid in the following reaction? 3H2S...
A:
Q: In fermentation distillation experiment, why does the boiling point increase, and the % ethanol decr...
A: A question based on physical chemistry, which is to be accomplished.
Q: ation. If it can rearrange draw the new carbocation obtained due to the rearrangement.
A:
Q: The markovnikov product for the reaction of methylcyclopentene with H2O)/H* is: Он А. В. HO C.
A: Markonikov addition reaction is the product form from the more substituted side of alkene.
Q: For the compound Pb(ClO4)2 what are the formulas of all the species you expect to be present in aque...
A: We are given an aqueous solution of Pb(ClO4)2, and we have to find all the species that are present ...
Q: What is the chemical formula of the solid represented by the vertical phase boundary passing through...
A: The given phase diagram is:
Q: Read the entire procedure and the recommended safety precautions. you think extra pieces of calcium ...
A: Detail explanation of the above question is given below.
Q: QUESTION 16 For the the reaction coordinate shown here, write the appropriate labels for the given d...
A: A question based on chemical kinetics, which is to be accomplished.
Q: Boron (B; Z=5) has two naturally occurring isotopes. Find the percent abundances of 1°B and 11B give...
A:
Q: Provide the balanced chemical equation for the undesirable reaction between HgCl2 and Sn2+ that will...
A: We are to write balanced equation for reacrion HgCl2 and Sn+2 , with excess Sn+2 .
Q: Does one bonding mode of DMSO favor on resonance structure in particular over the other? Please expl...
A: Resonance structure of DMSO
Q: For the cation Ti*: which of the following statements is false? Given: Ti (Z = 22) * The free ion ha...
A:
Q: Calculate the pH change [CH3COOH]=3.5*10^-3M, Ka of NaH2PO4/Na2HPO4 = 6.2×10^-8 A) add 0.5mL acetic ...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: Cis-1,4-polyisopreneis is Natural elastomers O Fiber with high crystanility O Thermoplastic O Synthe...
A: Answer - The correct option is - (a) Natural elastomers The cis-polyisoprene is natural elastomer...
Q: For a solution-phase dimerization reaction, the rate constants for the second-order forward reaction...
A: Equilibrium constant is ratio of rate constant of forward to backward
Q: Given that = -546.6kJ H,(g) +F,(g)→ 2 HF(g) rxn AHixn = -571.6 kJ (1DO H7 - (8) o+ (8) H calculate t...
A:
Q: Suppose you are titrating a sulfuric acid solution of unknown concentration with a sodium hydroxide ...
A:
Q: Circle anything and everything that applies for each of the following: (SS = soluble salt, IS = inso...
A: Carbon dioxide (CO2) , sodium oxide (Na2O) , nickel (II) phosphate [Ni3(PO4)2 ] are arranged accord...
Q: 31.01 OOne reaction scheme for producing ammonia, NH3, involves the reaction of methane (CH4), water...
A: Unbalanced chemical reaction equation is, CH4 + H2O + N2 -------> CO2 + NH3 Where, water = H2O...
Q: Gas A has a volume and pressure of 10.9 L and 201 KPa. Gas B has a volume and pressure of 8L and 29....
A: Given: For gas A : Volume = 10.9 L Pressure = 201 KPa For gas B : Volume = 8 L Pressure = 29.5...
Q: Consider CH3-CH(OH)-CH(OH)(Br). Determine the absolute configuration of each chiral center in one ...
A: We have to determine the absolute configuration of each chiral center in one pair of diastereomer of...
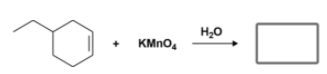
What is the organic product of the reaction in Figure 11?
Step by step
Solved in 3 steps with 1 images




