know there is only a limit to how many questions you can help me on and I don't know if each blank counts as one, I just have no idea how to start this.
know there is only a limit to how many questions you can help me on and I don't know if each blank counts as one, I just have no idea how to start this.
Chapter21: Potentiometry
Section: Chapter Questions
Problem 21.22QAP
Related questions
Question
I know there is only a limit to how many questions you can help me on and I don't know if each blank counts as one, I just have no idea how to start this.
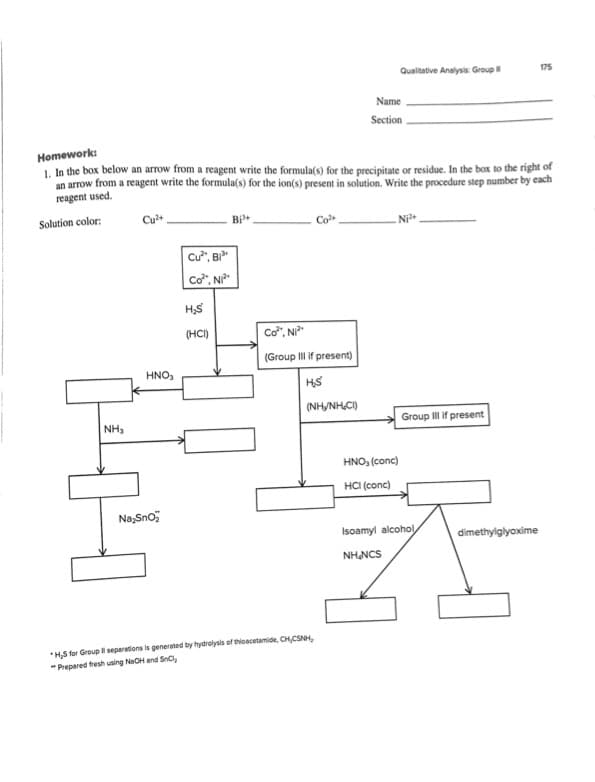
Transcribed Image Text:Qualitative Analysis Group
175
Name
Section
Homework:
1. In the box below an arrow from a reagent write the formula(s) for the precipitate or residue. In the box to the right of
an arrow from a reagent write the formula(s) for the ion(s) present in solution. Write the procedure step number by each
reagent used.
Solution color:
Cu
Bi
Co
Cư", Bì"
Cơ", NP"
(HCI)
Cơ". N
(Group II if present)
HNO
HS
(NH/NH,CI)
NH
Group III if present
HNO, (conc)
HCI (conc)
Na,Sno
Isoamyl alcohol
dimethylglyoxime
NH NCS
*HS for Group l separations is generated by hydrolysis of thioecetamide, CH,CSNH,
" Prepared fresh using NeOH and Snc,
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

