Q: A certain compound (molar mass = 561.06 g/mol) has a ΔHvap = 18.6 kJ/mol. How much heat (in kJ) is…
A:
Q: decimal place. No unit or wrong unit means wrong answer. 1. It is desired to prepare cupric sulfate…
A:
Q: The synthesis of ammonia obeys the following reaction: N2(g) + 3H2(g) → 2 NH3(g) What is the…
A: Answer: Given chemical reaction is: N2(g)+3H2(g)→2NH3(g) Relation between average rate of reaction,…
Q: 5c) Write the rate equation for the following SN1 reaction:
A: 5c) Write the rate equation for the following SN1 reaction:
Q: A reaction has values ΔS = 60.3 J K-1 mol-1 and ΔH = 49.6 kJ mol-1. At what temperature (in °C) will…
A: A reac is spontenous if ∆G is negative. The relation is given as : ∆G = ∆H - T∆S So we would…
Q: 2. Rank the compounds in each group according to their reactivity to electrophilic substitution. (a)…
A:
Q: the pH of liquid bleach is 12.0, what is the hydroxide ion concentration in this solution? 1 ×…
A: Given-> pH = 12.0
Q: How many moles of argon are in a 1.80 L container at 5.84°C if the gas exerts a pressure of 55.7…
A:
Q: solution of weak acid ammonium bromide
A:
Q: Propose a two reaction sequence to convert each of the following substrates into the indicated…
A:
Q: ᴡʀɪᴛᴇ ᴛʜᴇ ᴄᴏᴍᴘʟᴇᴛᴇ ʙᴀʟᴀɴᴄᴇᴅ ᴇQᴜᴀᴛɪᴏɴꜱ ᴏꜰ ᴛʜᴇ ꜰᴏʟʟᴏᴡɪɴɢ ᴄʜᴇᴍɪᴄᴀʟ ʀᴇᴀᴄᴛɪᴏɴꜱ. ꜱʜᴏᴡ ʏᴏᴜʀ ꜱᴏʟᴜᴛɪᴏɴ
A:
Q: Brz -> la. Ph Br Br Br B. ph C. ph' Br A. Ph Br CHC13 (3. Ph h koH CI Cl Ci CI cl B. Ph D. C. A. Ph…
A: In this question we have to tell the product of the reaction.
Q: VI. Give IUPAC names for the following compounds: (c) (a) CH3 (ы) он он HOCH2CH2CHCH20H…
A: IUPAC name of the given organic compounds are -
Q: In what two ways does the Morse potential provide a more accurate description of molecular…
A:
Q: 1.Determine the number of lone pairs (if any) on each oxygen, nitrogen, and charged carbon atoms in…
A: To find the lone pair on oxygen and nitrogen atom we will use the following formula. FC= V – (N + B)…
Q: II. Predict the products of the ff: elimination reactions and show the mechanisms involv HO H2SO4…
A:
Q: Seléct the st one synthetic operation. HO OH HO. O D O A O E
A:
Q: The weak base dimethyl amine, (CH3)2NH, has a Kb = 5.9 x 10¬4 at 25 °C. Select all the correct…
A: Base can be defined as the species that give hydroxide ions in solution and acid is a species that…
Q: 4. You need to make a buffer with pH as close to 10.00 as possible. Two sets of weak acids and…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What are the pertinent chemical equations in the standardization of sodium thiosulfate solution?
A: In the determination of Biological Oxygen Demand using the Modified Winkler Method, the…
Q: A 500.00 mg vitamin C (MW 176.12 g/mol) tablet was ground, acidified and dissolved in H2O to make a…
A: here we are required to find the % vitamin C in the tablet.
Q: The product f Hhis Ea reaction is.
A: We know that , E2 mechanism is bimolecular elimination reaction in which 1,2 elimination takes place…
Q: Consider an unknown metal M. If the density of M is 19.3 g/cm3 and radius is 137 pm, find out the…
A: The correct answer is given below
Q: H2O ÇI (R) CH3CCH2CH2CH3 ČH2CH3 d) -> e) Br NaOH CH3 E2
A:
Q: Provide a multi-step synthesis to create the intended product for the listed original reactant. The…
A:
Q: 1) Consider the salt of the metal M and the anion A, of formula MA2. In the compound MA2 the…
A:
Q: Ammonium phosphate (NH,),PO,) is an important ingredient in many solid fertilizers. It can be made…
A:
Q: The decomposition of 2 mol of water to hydrogen and oxygen requires 137 kcal of energy. The reaction…
A: Start by taking a look at the thermochemical equation given to you 2 H2O ( l ) +…
Q: Provide the correct IUPAC name for the compound shown here.
A: IUPAC nomenclature rules to cycloalkanes : 1. The ring is the parent chain 2. While numbering…
Q: Posphate NaPOa Classify each reaction according to whether a precipitate forms. Pricipitate forms…
A:
Q: How to prepare 100 mL of 10% AlCl3
A: The mass of solute in grams present in 100 mL of solution is known as the mass percent concentration…
Q: 1. The oxalate ion concentration, C;0. of 0.20 M H;C0 is adjusted to a value of 3.00 x 10 M. What is…
A: As per the guideline, since you have asked multiple questions, we have solved the first question for…
Q: 80 CO AS = ΔΗ ||
A: Entropy is degree of randomness or disorderness in system. Order of Entropy of different phases…
Q: What is a correct name of the following compound ? NH O acetanilide O methylacetanilide…
A:
Q: 6. The mercury in a 0.839 g sample was precipitated with an excess paraperiodic acid, H¿IO, SHg* +…
A: Given reaction is : 5Hg2+ + 2H5IO6 -------> Hg5(IO6)2 + 10 H+ Given mass of the sample = 0.839…
Q: Please show complete solution and apply all units A gaseous mixture contains 18% oxygen, and 78%…
A: Given : Percentage of oxygen = 18 % Percentage of nitrogen = 78 % Mass fraction of oxygen gas =…
Q: What is the IUPAC name?
A:
Q: Which of the following substances has the highest boiling point? CH3CN CH3Cl HCOOH CH2O…
A: The boiling point compound is directly proportional to the strength of intermolecular forces of…
Q: In the following diagram, show the direction of electron flow, label the anode and cathode then…
A: Given,
Q: Provide a synthetic route for the 5-step synthesis problem shown below. но но Choose 5 sets of…
A: Bromination Elimination to form alkene Ozonolysis Reduction will give carbonyl Treatment with…
Q: How many grams of CuCl are formed from the complete reaction of0.814 mol Cu? 2 Cu + Cl2 --> 2CuCl
A:
Q: 7.0 QUESTIONS 1. Calculate the molarity of a NaOH solution if 32.02 mL. of the solution neutralizes…
A: Here we are required to find the Molarity of NaOH.
Q: In the p-type semiconductor the dopant and space in between answers) an electron the silica. (put a…
A: There are two type of semiconductor in solids. P type semiconductor and N type semiconductor.
Q: 1. How does the electron theory explain (a) why a rubber rod becomes negatively charged when rubbed…
A: When a rubber rod is rubbed with a fur the electron get transferred from the fur to the rod thus…
Q: CO reacts with Cl2 in the gas phase according to the following chemical equation: Co(g)…
A:
Q: You are tasked to prepare a 0.5 L of HCl solution with a concentration of 0.04 M. How will you do…
A: Given that - % HCl Solution = 35% Volume of new HCl Solution = 0.5 L = 500 mL (V2) Molarity of new…
Q: Identify the reagent that is needed to complete the transformation shown below. Assume an acidic…
A:
Q: SHOW SOLUTION A 0.7120 g of iron ore was brought into solution and passed through a Jones reductor.…
A: Given that - Balanced Chemical equation is - 5Fe+2 + MnO4 - + 8H+ → 5Fe+3 + Mn +2 + 4H2O Mass of…
Q: which compounds are soluble in water?
A: Here we are required to find the compound which are soluble in water
Q: ESTIONS 1. Acertain soft drink was analyzed and found to contain 0.080MH. Other ingredients of the…
A: We are authorised to solve only one question at a time. Please post rest of the questions…
5b) Draw the expected product(s) resulting from the following SN2 reaction:
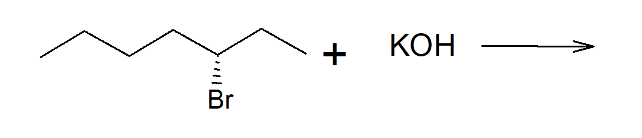
Step by step
Solved in 2 steps with 2 images


