Q: it is the correct order of stability of the following radicals (more stable > less stable)?
A: Carbon forms four bonds to complete its octet. In the formation of free radical,carbon has formed…
Q: Rank the carbocation compounds below from most stable to least stable.
A:
Q: Which Is the most stable radical? Cllck on a letter A through D to answer. A. В. С. B. D.
A: So the question has asked to determine the stability of free radical among the following four…
Q: 2. Zn, H30* CH3 7. Draw the other resonance contributor to this radical:
A:
Q: Draw curved arrows to show the movement of the electrons as one resonance contributor is converted…
A: Resonance contributors are the ones which are formed by moving a single electron resulting in…
Q: Rank the following from most to least stable: OH ОН OH C A,
A: Applying concept of stability of alkene with the help of hyperconjugation and stereochemistry of…
Q: んnん or
A: NOTE : Since you've asked multiple questions,we'll solve the first question for you.If you want any…
Q: The least Stable carbocation is
A: Given that : We have to identify the least stable carbocation out of the following :
Q: a. HN02 H12504 b. draw the resonance C. Which resonance Structure is most stable ? would react…
A: Ketone group is an ElectronWithdrawing group and Electrophile Attach at meta position .
Q: 1. Rank the stability of the carbon radicals below from least stable to most stable. В
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: • Draw resonance structures for a conjugated alkene, ion, or radical, and use an MO diagram to…
A: The energy of a system which forms resonance structure is lowered by interaction of pi bond with the…
Q: Which SN1 raction is faster in the given pair?
A: In the reaction it is said that reaction proceed by SN1 mechanism means there must be formation of…
Q: 14. What is the pro 15. What is the nar A) Fischer B) Friedel-c C) Diels-Ald D) Wittig E) Grignarc…
A:
Q: Rank the following in order of stability, from most stable -1 to least stable - 4
A:
Q: What is the correct order of stability of the following radicals (more stable > less stable)?
A:
Q: lentify the 3 major contributors to the resonance hybrids A B C E OZI ZI ZI ZI OZI OZI
A:
Q: 5. Circle the most stable one in each pair: or or
A:
Q: Which resonance contributor in each pair makes the greater contribution to the resonance hybrid?
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: Rank the following radicals in order of decreasing stability. most stable least stable (CHдС-снсн,…
A: Free radicals are electron deficeint. the stability of tertiary radicals is more than that of…
Q: Why is the 2 and 4 positions electrophilic, but the 3 position is not electrophilic ?
A:
Q: Between the following radicals; which one is more stable? I or II PHHC(*)CH2ET (1) PHCH2C(*)HEt (1I)…
A: Given free radicals are PhHC(•)CH2Et (I) PhCH2C(•)HEt (II) The free radical with more…
Q: ving free radicals is the MOST stable? C. D.
A:
Q: Rank the molecules below from least stable to most stable
A: we have been asked the stability of given ring structure
Q: Rank the attached radicals in order of increasing stability
A: Free radicals refer to the type of reactive intermediate in which a homolytic cleavage of bond takes…
Q: Which properly shows resonance of an allylic system? B C H. O: D
A: Resonating structures are the different structures which are obtained by the movement of pi of…
Q: See attachment
A: If more numbers of alkyl substituents are present in the radical carbon atom then it will be more…
Q: 13) Explain why (A) is more stable than (B) in the following two radicals (use resonance structures)…
A:
Q: Rank order the following in terms of relative reactivity (most reactive on the left, least reactive…
A:
Q: 1. Draw the resonance structures obtained by delocalising the lone pair on heteroatom X onto the…
A: Since you have posted multiple unrelated questions, we are entitled to answer the first only.
Q: Which one is more stable S2-, S4-, S6-?
A: The given ions of Sulphur are S2-, S4-, and S6-.
Q: 4. In each pair, circle the anion that is more stable. CI
A: An ion bearing a positive charge is called anion. Cations and anions are present in ionic compounds.…
Q: Given the compounds below, which is the more stable one?
A:
Q: a.What carbon radical is formed by homolysis of the C–Ha bond in propylbenzene? Draw all reasonable…
A: a. The structure of propylbenzene is, The structure of propylbenzene indicates that there are…
Q: Rank the following radicals in order of increasing stability.
A: Since the stability of free radicals depends on two things 1) resonance => more is the resonance…
Q: Which of the following is the most stable radical? CH,CHCH(CH,), CH,CH,CH(CH,), CH,CH,C(CH,),
A:
Q: Rosmarinic acid is an antioxidant isolated from rosemary. Draw resonance structures for the radical…
A: The removal of hydrogen atom in rosmarinic acid results in the formation of radical as shown below,
Q: Can you explain how they got the minor product, show the movement of electrons etc.
A:
Q: 4) Assign E /Z designation to each double bond where applicable. a) b) N, NH Br c)
A: Stereoisomers are the molecules having same molecular formula but differ only on the spatial…
Q: Rank the following 4 radicals in order of stability (1 = most stable, 4 = least stable).
A: see answer below.
Q: 2. Rank the following anions from most to least stable. a. b. d.
A: . Stability of anion depends upon the following factor :-a Resonance stabilize the anion .b Lower…
Q: Which one is the most stable?
A: The more substituents the alkenes have, the more stable they are. Thus, a tetra substituted alkene…
Q: Rank the free radicals shown below in order of increasing stability:
A:
Q: Arrange the following radicals in order of decreasing stability (most stable first). B>D>A>C e)…
A: radical A is stabilised by resonance + 3 alpha hydrogen (hyperconjugaton).
Q: how all the α-H's in the molecules below.
A: Given compounds,
Q: Rank the following radicals in order of increasing stability.
A: The stability of free radical is governed by various factors such as: Allylic / benzylic radical 3°…
Q: 2. Arrange the structures below in terms of most stable to least stable. Ans a. A B b. Y Ans
A: The stability of carbocations can be explained by Inductive effect and Hyper conjunction . The…
Q: Illustrate the Reaction of a Radical X• with a C=C ?
A: The free radical X·with an alkene (C=C).
Q: 23. Draw a resonance structure for the radical below and circle the one that is the most stable.
A:
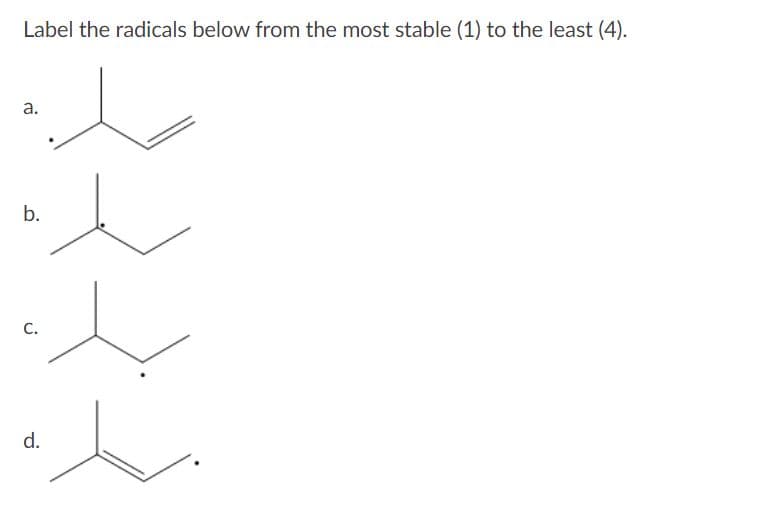
Label the radicals below from the most stable (1) to the least (4).
Step by step
Solved in 2 steps with 2 images

- Could you explain why the highlighted group on the right side of the double bond has a higher priority than the lower group on the right side of the double bond. I use this information to determine E/Z assigmentsI need alot of help on this section too!!5. Circle the most reactive with water and put an X through the least reactive
- Circle the molecule in each pair that is more stable, and write the reason why you selected that moleculeA A c-) For each pair of resonance structures shown below (A and B), circle which resonance contributor makes a greater contribution to the resonance hybrid. Briefly explain your answer. O.. :0: B m N:Please give the best resonance and second best resonance for SO2. Please explain why the best resonance is the "best" and why the second best resonance is the "second best".
- Rank the following radicals from least to most stable.Woodward and Fieser : Parent C=C-C=O Could you draw out how Alkyl/ring residue of a alpha vs beta vs sigma would look?a.What carbon radical is formed by homolysis of the C–Ha bond in propylbenzene? Draw all reasonable resonance structures for this radical. b.What carbon radical is formed by homolysis of the C–Hb bond in propylbenzene? Draw all reasonable resonance structures for this radical. c. The bond dissociation energy of one of the C–H bonds is considerably less than the bond dissociation energy of the other. Which C–H bond is weaker? Offer an explanation.

