Q: Time's Up! Provide the correct IUPAC name for the compound shown here. ОН F 5- 6- 2- 1- 3- 4- сyclo…
A:
Q: Calculate the partial pressure in [bar] of N; in air consisting of N3, Oz, and Ar assuming that the…
A: Answer: This question is based on the density form of ideal gas equation which is shown below:…
Q: If atoms have mostly empty space, so how does matter stay strong? please explain
A: The empty space in an atom is the space where the nucleus (proton+neutron) is absent.
Q: Which statement is true regarding drugs given intramuscularly? A. The drug goes directly to the…
A: Drugs can be given different ways- 1- orally it can be given 2- it can be given intramuscularly…
Q: 1. Calculate the volume of 0.050OM EDTA needed to thrate 26.37 m of 0.074IM MaNO Molar Mass MglNO-…
A: Answer: By using Eriochrome Black T indicator Mg2+ ion can be easily estimated by titrating them…
Q: Which of the following do you expect to be optically active? Encircle the letter(s) corresponding to…
A:
Q: Which molecule(s) contain(s) ketone functional group? a. CH3 OH H. 0=C осн OH CH3 C. CH3 OH O none…
A: Anwer of the question given below,
Q: Part B Calculate the entropy of each of the following states. (Figure 1) State (b). S = 0 J/K Submit…
A:
Q: If you have 4.7 moles of KBr in a 0.520 M solution of KBr, what is the volume o f your solution?
A:
Q: Provide the suitable reagent / reaction conditions for the following chemical transformation: 1) 2)…
A:
Q: A 100.0-mL sample of spring water was treated to convert any iron present to Fe. Titration with…
A:
Q: 11. Draw the following galvanic (voltaic) cells (salt bridge design) and label the following: i)…
A: Galvanic cell is also known as electrochemical cell . The electrochemical cell in which the chemical…
Q: (a) How many milliliters of 0.145 M HCl are needed to neutralize completely 45.0 mL of 0.101 M…
A:
Q: 3. Which process is expected to involve the highest energy? Transition from n=5 to n=1 in hydrogen…
A:
Q: We want to use Epsom salt (magnesium sulfate; bath salt) instead of common salt (sodium chloride)…
A: In order to produce the same effect of salting out, we have to produce a solution with the same…
Q: Consider the equilibrium NO2(g) = NO(g) + 1/2 O2(g). One mole of NO2(g) is placed in a vessel and…
A: Ans.
Q: A 2.16 g sample of phosphorus was burned in a large excess of chlorine, and the phosphorus chloride…
A: To Calculate molar mass , we can use the relation that rate of effusion is inversely proportional to…
Q: Draw three resonance structures for CS,. This species has its three atoms bonded sequentially in the…
A:
Q: What is the pH of a solution that has 0.22 M fluoride anion and 0.28 M hydrofluoric acid? Ka…
A: Given data,Concentration of F-=0.22MConcentration of HF=0.28MKa of HF=5.62×10-4
Q: ne standard reaction entropy of
A:
Q: b) Use the following radial probability function to justify i. why the B than has a first lower…
A: A question based on atomic structure that is to be accomplished.
Q: The mole fraction of silver acetate, AgCH3COO, in an aqueous solution is 7.96x10-2. The percent by…
A:
Q: What do we mean by the term electronic structure? Select all that apply. a) The arrangement of the…
A: Matter is made up of atoms. Atoms consists of sub atomic particles like electrons , protons and…
Q: If you have 35.4 mL of a 4.55% NaNO3 solution, what is the mass of NaNO3?
A: Answer: In this question (w/v)% of NaNO3 solution is given to us and we have to find out the mass of…
Q: VISUALIZATION Modes of Radioactive Decay 140 130 120 110 100 90 80 70 60 50 • Stable • Radioactive…
A: Here we have to predict the neutrons to proton ratio for atomic number 20 or less when graph is…
Q: A mixture containing 2.21 g each of CH4(g), C,Ha(g) and CaH,o(g) is contained in a 1.50 L flask at a…
A:
Q: The solubility of Ca(IO3)2 Woud be in a solution of 4M iodate ion thán in pure water at the same…
A: From common ion effect we found that the solubility of a sparingly soluble salt decrease when a…
Q: The emulsifying agent, a salt, is prepared from 2 chemicals. A) write the structure of stearic acid…
A:
Q: An uninsulated steam pipe passes through a room in which the air and walls are at 28°C. The outside…
A:
Q: Give the clear handwritten answer..
A:
Q: Potassium dichromate is often used to standardize reducing agents like Fe2** Cr207 + 6Fe2+ + 14 H* →…
A: it is an example of redox titration where potassium dichromate is standardized using reducing agents…
Q: 2. Are the following compounds likely to dissociate, partially dissociate, or not dissociate when…
A:
Q: Reduction is the ___________ of electrons. Select one: a. loss b. share c. transfer d. gain
A: ->Reduction in process in which the oxidation state of substance decreases. ->This decrease in…
Q: • Determine the number of grams of H2SO4 that would be contained in 800.0 mL of a 1.5 M H2SO4…
A: Given-> Volume b= 800 ml = 0.800 L (1L =1000 ml) Molarity = 1.5 M
Q: Write the chemical equation for the reaction between MgCl2 and the soap prepared. (Soap prepared is…
A:
Q: 1. Electrical current is carried by a flow of charged particles, which often occurs through…
A: Electrical conductivity of a material is based on the presence of charge carriers inside that…
Q: Draw the Newman and Sawhorse representation of ethane with the LOWEST POTENTIAL ENERGY.
A: Ethan has two different possible Newman and Sawhorse projections which are eclipsed and staggered.
Q: There are 14.5 psi per bar. Calculate the pressure in psi at 2.8 bar. Answer:
A: It is an example of conversion factor We are required to convert bar to psi
Q: D. SOs Е. Н:О
A:
Q: Show the two (neutral) organic starting materials that would be used to synthesize the compound…
A:
Q: Mg + O2 -------> MgO Na2CO3 -------> Na2O + CO2 C3H8 + O2 -------> CO2 + H20
A: Note : As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost…
Q: 2. Are the following compounds likely to dissociate, partially dissociate, or not dissociate when…
A: There are different type of substances with different physical and chemical properties. Dissolution…
Q: A water main that carries a flow of 0.5 MGD has been repaired after a break. The required chlorine…
A: Volume flowing = 0.5 MGD = 0.5 million gallon / day = 2273045 L/day (1 gallon = 4.546 L)
Q: Calculate the pH of a 5.3 x 10-3-M solution of H2S04 (Ka, = 1.2 x 10-2). pH =
A: Given-> Concentration of H2SO4 = 5.3 × 10-3 M
Q: 460 410 360 310 260 210 160 110 60 -10 -510 4 5 10 15 20 25 30 35 40 45 50 55 1/[S] per mM 1. What…
A: Michaelis Menten equation shows the effect of the concentration of the substrate on the rate of an…
Q: An electrochemical cell is based on the following two half-reactions: oxidation: Sn (s) →Sn²+ (aq,…
A:
Q: POST-LABORATORY QUESTIONS Due after Laboratory Experiment 1. While performing the Dumas experiment,…
A: ¶1):-> Step 1 Dumas experiment is used to determine the molecular weight of a substance which are…
Q: What is the pH of 0.024M acetic acid solution at 25°C? (Ka=1.8×10^-5)
A: Given, [CH3COOH] = 0.024 M Ka = 1.8 × 10-5
Q: Find the normalization constant, N, for the antibonding orbital wave function formed fi orbitals,…
A:
Q: A titration is carried out where 3 milliliters of 0.70M NaOH is added to 15 milliliters of 0.150M…
A: Volume of NaOH= 3 ml molarity of NaOH 0.70M Volume of HCl =15 ml Concentration of HCl= 0.150M pH…
Predict the product of the following reaction.
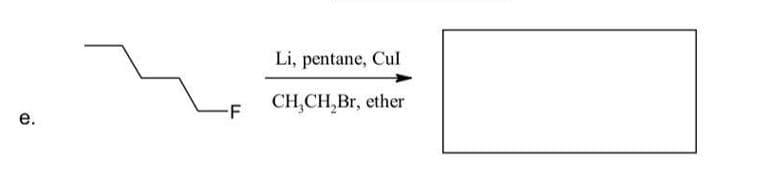
Step by step
Solved in 2 steps with 1 images


