Sample "Lodi" was subjected to different chemical tests and the following data were obtained: Test Ninhydrin Biuret heating +/- Observations colordess spot appearance of violet -colored solution formation of coagulated product + + From the choices below, what is the most possible identity of sample "Lodi"? (A amino acid (B) amine © peptide D protein
Sample "Lodi" was subjected to different chemical tests and the following data were obtained: Test Ninhydrin Biuret heating +/- Observations colordess spot appearance of violet -colored solution formation of coagulated product + + From the choices below, what is the most possible identity of sample "Lodi"? (A amino acid (B) amine © peptide D protein
Chapter28: Atomic Spectroscopy
Section: Chapter Questions
Problem 28.13QAP
Related questions
Question
this is
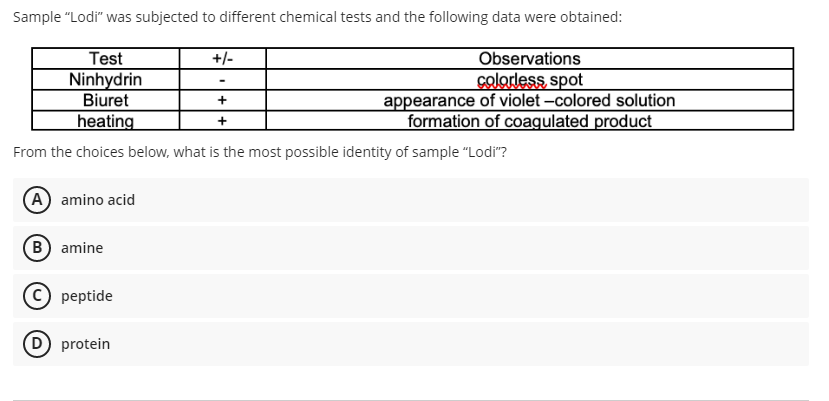
Transcribed Image Text:Sample "Lodi" was subjected to different chemical tests and the following data were obtained:
Test
Ninhydrin
Biuret
heating
+/-
Observations
colordess spot
appearance of violet -colored solution
formation of coagulated product
+
+
From the choices below, what is the most possible identity of sample "Lodi"?
(A amino acid
(B) amine
© peptide
D protein
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

