lonic Size of Stable Fourth Second lonization Third Atomic Size (pm) First lonization Energy (k/mol) 9543 5877 10,543 6491 lonization lonization lon Element Energy (k/mol) 496 Energy (ki/mol) 4562 Energy (kl/mol) (pm) 116 Sodium 6910 154 152 196 130 Potassium 4420 7732 4912 419 3052 86 Magnesium Calcium 738 1451 114 590 1145 174 Justify the following using Coulomb's law and the data from the table a) Potassium is a larger atom than sodium. b) Sodium is a larger atom than magnesium. c) The first ionization energy of magnesium is larger than that of sodium. d) The first ionization energy of magnesium is larger than that of calcium. e) The second ionization of Mg is larger than its first ionization ) The second ionization of Na is larger than the second ionization of Mg g) KCI, is not known to exist. h) CaCl, is not known to exist. i) Sodium ion is smaller than the neutral sodium atom. j) Calcium ion is larger than the magnesium ion. k) Sodium ion is larger than the magnesium ion.
lonic Size of Stable Fourth Second lonization Third Atomic Size (pm) First lonization Energy (k/mol) 9543 5877 10,543 6491 lonization lonization lon Element Energy (k/mol) 496 Energy (ki/mol) 4562 Energy (kl/mol) (pm) 116 Sodium 6910 154 152 196 130 Potassium 4420 7732 4912 419 3052 86 Magnesium Calcium 738 1451 114 590 1145 174 Justify the following using Coulomb's law and the data from the table a) Potassium is a larger atom than sodium. b) Sodium is a larger atom than magnesium. c) The first ionization energy of magnesium is larger than that of sodium. d) The first ionization energy of magnesium is larger than that of calcium. e) The second ionization of Mg is larger than its first ionization ) The second ionization of Na is larger than the second ionization of Mg g) KCI, is not known to exist. h) CaCl, is not known to exist. i) Sodium ion is smaller than the neutral sodium atom. j) Calcium ion is larger than the magnesium ion. k) Sodium ion is larger than the magnesium ion.
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter12: The Solid State
Section: Chapter Questions
Problem 16PS
Related questions
Question
Please answer d and e and f
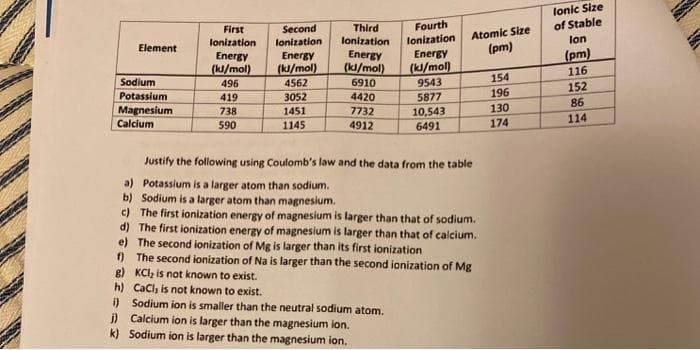
Transcribed Image Text:lonic Size
of Stable
Fourth
Second
lonization
Atomic Size
(pm)
First
Third
lonization
lonization
lonization
lon
Element
Energy
(k/mol)
496
Energy
(k/mol)
Energy
(kl/mol)
(pm)
116
Energy
(kl/mol)
4562
3052
Sodium
6910
9543
154
152
Potassium
419
4420
5877
196
86
130
Magnesium
Calcium
738
1451
7732
10,543
174
114
590
1145
4912
6491
Justify the following using Coulomb's law and the data from the table
a) Potassium is a larger atom than sodium.
b) Sodium is a larger atom than magnesium.
c) The first ionization energy of magnesium is larger than that of sodium.
d) The first ionization energy of magnesium is larger than that of calcium.
e) The second ionization of Mg is larger than its first ionization
) The second ionization of Na is larger than the second ionization of Mg
g) KCl, is not known to exist.
h) CaCl, is not known to exist.
i) Sodium ion is smaller than the neutral sodium atom.
j) Calcium ion is larger than the magnesium lon.
k) Sodium ion is larger than the magnesium ion.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning