Q: , draw its and then in the 2. For (1R, 2S, 4R)-4-tert-buyl-1-ethyl-2-methylcyclohexane: draw its cor...
A: Drawing skeletal structure of (1R,2S,4R)-4-tert-butyl-1-ethyl-2-methylcyclohexane.
Q: It is already known the specific heat of water is 4200 J/kg/°C. The vaporization energy of water is ...
A: Solution - According to the question - Given - To perform the conversion we need to do steps- 1) Hea...
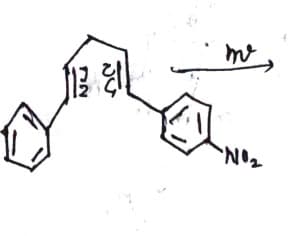
Q: Draw the product of this nydrogenation reaction. Use wedge and dash bonds to indicate stereochemistr...
A: Interpretation: We have to determine the product for the following given reaction.
Q: The reactant concentration in a first-order reaction was 6.80×10-2 M after 25.0 s and 2.10×10-3 Maft...
A: First order kinetic reaction are those in which the rate is direcdirectly proportional to the concen...
Q: 12 Belo e the processes that describe the first second, and third loniration energies of an aluminiu...
A: The crude defination of ionization energy is that the energy required to remove the electrons from t...
Q: Predict the solubility product constant of the given equation: MGF2(s) Mg*2(aq) + 2F(aq) Sample ans...
A:
Q: Consider the following elementary steps that make up the mechanism of a certain reaction: 1. 2A→B + ...
A: Given elementary steps are : 1. 2A --------> B + C 2. B + D -----> E + C a). What is the ove...
Q: When a chemical reaction occurs, atoms rearrange to form new compounds, but no new atoms are created...
A: Figure shows A is single element. There are 4 A atoms present in the reactant side. B is diatomic mo...
Q: Consider the reaction A + 2B = C whose rate at 25 °C was measured using three different sets of init...
A: A + 2B = C
Q: 12. Below are the processes that describe the first, second, and third ionization energies of an alu...
A: Ionization Energy The amount of energy required to remove the electron from the valance electron fro...
Q: Choices a. Solid b. Liquid c. Gas d. Solid and liquid e. Solid and Gas f. Liquid and Gas g. Solid, L...
A:
Q: Part A Consider the following reaction: 2CH3OH(g) +3O2(g) → 2CO2(g) + 4H2O(g) How many CO2 molecules...
A: If you see each reaction mixture then you see more number of reactants in ( b ) reaction mixture tha...
Q: An analyst wishes to add 256 mg of Cl-to to a reaction mixture. How many mL of 0,217 M BaCl is this?
A: Given, Mass of Cl- = 256 mg Molarity of BaCl2 = 0.217 M Volume of BaCl2 in mL = ?
Q: Calculate the amounts of Cu and Br: produced at the inert elecuvues by passing a current of 4.5 A th...
A: Given: Current = 4.5 A The atomic weight of Cu =63.55 gThe atomic weight of Br2=159.8 gCharge of 1 m...
Q: 17) An unknown hydrocarbon compound was analyzed for hydrogen by elemental analysis and results show...
A:
Q: This series demonstrates a progression as 0.5 M solution of sodium carbonate (?? ?? ) as it is added...
A: 1. Formation of white precipitate indicates that a chemical reaction has occurred 2. The balanced ch...
Q: how can you prepare diethyl ether from an alkyl halide and any other organic or inorganic compound?
A: Diethyl ether is prepared from an alkyl halide by Williamson's synthesis . Williamson's synthesis is...
Q: 3. What is the correct IUPAC name for the following compound? N. CH3 OA N-benzyl-N-methylamine O B. ...
A: 1- In this question, we will write the IUPAC name of the given compound. You can see below.
Q: (a) Which has the smaller fourth ionization energy, Sn or Sb? (b) Which has the larger sixth ionizat...
A: Ionisation energy is of energy required to remove an electron from the valence shell of an isolated ...
Q: 12. Why does Li have a larger first ionization enengy than Na? OA Because Li has only 3 electrons an...
A: Ionization energy :- The amount of energy required to remove an electron from an isolated neutral g...
Q: In an experiment, 2 grams of zinc and 150 mL aq CuSO. were made to react, then as observed, the temp...
A: Given the mass of Zn = 2 g Molar mass of Zn = 65.38 g/mol Conversion factor: 1 mol = 65.38 g Moles o...
Q: Give the values for all four quantum numbers for each electron in the ground state of the nitrogen ...
A: The solution is given below -
Q: Lewis
A:
Q: The equilibrium constant, K, for the following reaction is 3.92×10-2 at 531 K. PCl5(g) PCl3(g) + Cl2...
A: [PCl5] = 0.120 M [PCl3] = 0.0687 M or 6.87 x 10-2 M [Cl2] = 0.0687 M or 6.87 x 10-2 M
Q: Consider two solutions, solution A and solution B. [H+] in solution A is 450 times greater than that...
A: Ph difference between solution A and solution B is 2.6232
Q: Rashid has performed titrimetric analysis of antacid tablets which solely contain aluminum hydroxide...
A: To neutralize 100ml of stomach acid with a PH 1.50 we require 31.68 ml Of a 0.0998M NaOH solution.
Q: Considering molecule, th (or "g") indic change in si passing thro
A: In bonding molecular orbital, sigma is the cylindrically symmetric along the bond axis bonding molec...
Q: Ph H. PPH3 oxophosphetane complex I H. Ph EtO2C H. OYonhosnhetane complex II
A: The above reaction is a witting reaction in which the aldehyde or ketone reacts with phosphonium yli...
Q: Suppose that a semiconductor device is using a 9.17-mg sample of a hypothetical element By, that is ...
A:
Q: 8.97 x 106 J = kcal Incorrect
A:
Q: The concentration of calcium carbonate in a sample of water saturated with the solid was found to be...
A:
Q: Based on the thin layer chromatography experiment what should be considered when choosing the right ...
A: Thin layer chromatography is one of the conventional technique for the separation of constituents in...
Q: absorbance data that you have obtained while doing an experiment studying the diffusion of potassium...
A: A numerical problem based on quantitative analysis, which is to be accomplished.
Q: A 250,0 mL aqueous solution contains 45,1 micro gram of a pesticide. Expres the pesticide's concentr...
A: Given, Volume of aqueous solution of pesticide = 250.0 mL Mass of pesticide in μg = 45.1 μg a). Ca...
Q: What's More Activity 1. Stoich in Action! Direction: Use a separate sheet to show your computation f...
A: (1) Given Chemical equation is - Fe(s) + O2(g) ----> Fe2O3(s) (a) The balanced Chemical equat...
Q: (1/ The volume of an aqueous solution of KBr at 25 °C was measured at a series of molalities, m, and...
A:
Q: By fully describing the nature of each solid including a diagram for each, explain why solid HCl mel...
A: Melting point of a solid is quite dependent on the nature of the force between the atoms in a molecu...
Q: 8. Determine if the following compounds are soluble or insoluble (circle your answer): a. NaCl solub...
A: A cation and anion combination gives a salt .certain cation and anion combination results in precipi...
Q: Please answer serisously and provide link for validation. Thank you :) 1. Why can alcohols reac...
A: We can define esterification a reaction where carboxylic acid reacts with alcohol in presence of aci...
Q: Find the pH of a 0.200 M NH4Cl solution given that the Kb of NH3 = 1.8x10-5 at 25 °C.
A:
Q: Consider the following system at equilibrium where H° = -111 kJ/mol, and Kc = 0.159, at 723 K. N2(g)...
A: Given that ∆H° = -111 kJ/mol Hence the reaction is exothermic in nature. This means the forward rea...
Q: 5. Calculate the emf at 298 K generated by the cell: Cr20, (aq) + (aq) + 61 (aq) → 2Cr*(aq) + 312(s)...
A: Given : We have to calculate Ecell for the reaction.
Q: In the following chemical reaction, 2 mol of A will react with 1 mol of B to produce 1 mol of A2B wi...
A:
Q: Find the empirical formula of the following compounds: Compound made of a. 0.039 mol of iron atoms c...
A: Given : a) no. of moles of iron atoms = 0.039 mol no. of moles of oxygen atoms = 0.052 mol b) mas...
Q: which one of the Folow ing is NOT an exampie of an acidic acidl? o Sodium dinydrog en phosphate o ma...
A: If any compound have very low pH, then it's means the compound have higher acidic property, therefor...
Q: Not all unit cells have a cubic structure. Take for example the below image wherein the unit cell ta...
A: Given, density = 25.69 gm/cm3 a = 10.48 A b = 6.59 A Molar mass of Ro = 49.875 gm/mole
Q: 24) Which of the following compounds contains the largest number of atoms? A) 1.00 mole of H,PO, C) ...
A:
Q: How many lone pairs (total) around the molecule COCl2 ?
A: The total number of lone pairs around the molecule can be calculated by drawing the Lewis structure ...
Q: Shon how you wuld ynthesize to compound below using any reagents. More than one step may be required...
A:
Q: Based on the given figure, a. what is the possible identity of the amino acid? b. what is the isoele...
A: Since you have asked a question with multiple subparts, we will answer only first 3 subparts for you...
Step by step
Solved in 2 steps with 1 images




