MInbox (5) - bubbatx1998@gmail.cx A ALEKS Daniel Estrella - Pre-Clas X bAnswered: NAverage Bond Entha My Drive - Google Drive X X X www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-lgNslkr7j8P3jH -IBiKFdvJdkR4Qg D Fu FbLE9J HQJOYSYezxnZjy7ErUilWNytDwYCaGIl7ay3s981 NTPxuxnt8g-2ai_6UhWKeYIDgamBWqio?1oBw7QYjlbavbS... AMy Application Stat... IT Welcome Aggie C... Hazlewood Act- Te... New Tab a Amazon.com: Gene... AVeterans - Addition... T Aggie Transition Ca... StudentLoans.gov |... Apps http://bookshelf.vi... Pre-Class Quiz #17- 10/11/19 Question 3 of 3 (1 point) Daniel 2 = 3 = 1 (H,0). If 0.540 g Aqueous hydrochloric acid (HCI) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium chloride (NaCl) and liquid water of sodium chloride is produced from the reaction of 0.73 g of hydrochloric acid and 0.56 g of sodium hydroxide, calculate the percent yield of sodium chloride. dlo Be sure your answer has the correct number of significant digits in it. ? X Submit Assignment 2019 McGraw-Hill Education. All Rights Reserved Terms of Use Privacy 1:33 PM PType here to search 13 17 10/10/2019 ... г
MInbox (5) - bubbatx1998@gmail.cx A ALEKS Daniel Estrella - Pre-Clas X bAnswered: NAverage Bond Entha My Drive - Google Drive X X X www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-lgNslkr7j8P3jH -IBiKFdvJdkR4Qg D Fu FbLE9J HQJOYSYezxnZjy7ErUilWNytDwYCaGIl7ay3s981 NTPxuxnt8g-2ai_6UhWKeYIDgamBWqio?1oBw7QYjlbavbS... AMy Application Stat... IT Welcome Aggie C... Hazlewood Act- Te... New Tab a Amazon.com: Gene... AVeterans - Addition... T Aggie Transition Ca... StudentLoans.gov |... Apps http://bookshelf.vi... Pre-Class Quiz #17- 10/11/19 Question 3 of 3 (1 point) Daniel 2 = 3 = 1 (H,0). If 0.540 g Aqueous hydrochloric acid (HCI) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium chloride (NaCl) and liquid water of sodium chloride is produced from the reaction of 0.73 g of hydrochloric acid and 0.56 g of sodium hydroxide, calculate the percent yield of sodium chloride. dlo Be sure your answer has the correct number of significant digits in it. ? X Submit Assignment 2019 McGraw-Hill Education. All Rights Reserved Terms of Use Privacy 1:33 PM PType here to search 13 17 10/10/2019 ... г
Chapter31: Synthetic Polymers
Section31.SE: Something Extra
Problem 21AP
Related questions
Question
Transcribed Image Text:MInbox (5) - bubbatx1998@gmail.cx
A ALEKS Daniel Estrella - Pre-Clas X
bAnswered: NAverage Bond Entha
My Drive - Google Drive
X
X
X
www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-lgNslkr7j8P3jH -IBiKFdvJdkR4Qg D Fu FbLE9J HQJOYSYezxnZjy7ErUilWNytDwYCaGIl7ay3s981 NTPxuxnt8g-2ai_6UhWKeYIDgamBWqio?1oBw7QYjlbavbS...
AMy Application Stat...
IT Welcome Aggie C...
Hazlewood Act- Te...
New Tab
a Amazon.com: Gene...
AVeterans - Addition...
T Aggie Transition Ca...
StudentLoans.gov |...
Apps
http://bookshelf.vi...
Pre-Class Quiz #17- 10/11/19
Question 3 of 3 (1 point)
Daniel
2
= 3
= 1
(H,0). If 0.540 g
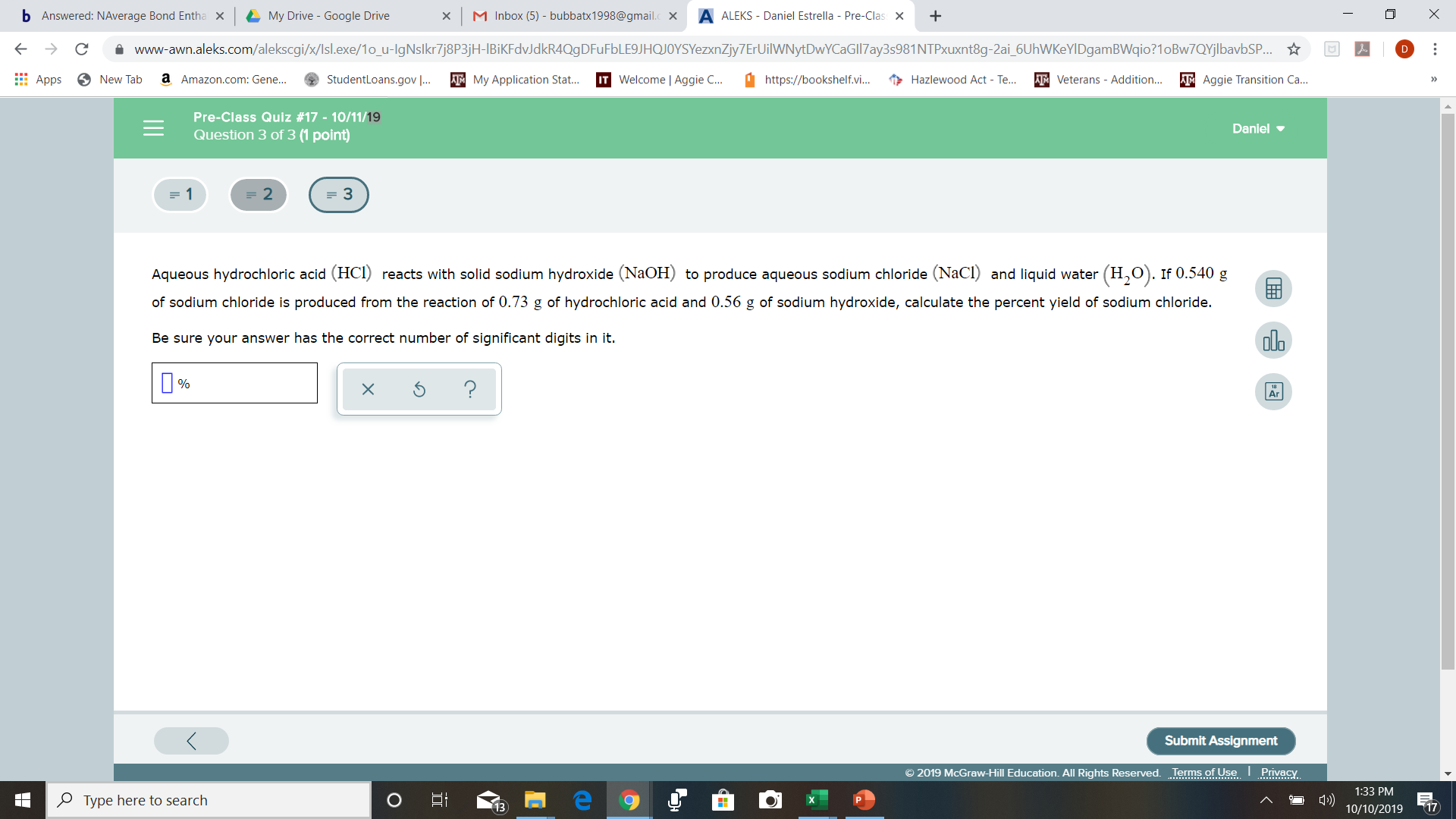
Aqueous hydrochloric acid (HCI) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium chloride (NaCl) and liquid water
of sodium chloride is produced from the reaction of 0.73 g of hydrochloric acid and 0.56 g of sodium hydroxide, calculate the percent yield of sodium chloride.
dlo
Be sure your answer has the correct number of significant digits in it.
?
X
Submit Assignment
2019 McGraw-Hill Education. All Rights Reserved
Terms of Use
Privacy
1:33 PM
PType here to search
13
17
10/10/2019
...
г
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

