MISSED THIS? Read Section 6.6 (Pages 228-235); Watch KCV 6.6, IWE 6.10. A gas mixture contains 1.29 g N₂ and 0.76 g O₂ in a 1.64-L container at 15 °C. Mother to Son &....pdf A Part A Calculate the mole fraction of N₂. Express your answer to three significant figures. XN₂ = Submit ▾ Part B X₂ = 195] ΑΣΦ Submit Request Answer Calculate the mole fraction of O₂. Express your answer using two significant figures. 1951 ΑΣΦ → Request Answer ?
MISSED THIS? Read Section 6.6 (Pages 228-235); Watch KCV 6.6, IWE 6.10. A gas mixture contains 1.29 g N₂ and 0.76 g O₂ in a 1.64-L container at 15 °C. Mother to Son &....pdf A Part A Calculate the mole fraction of N₂. Express your answer to three significant figures. XN₂ = Submit ▾ Part B X₂ = 195] ΑΣΦ Submit Request Answer Calculate the mole fraction of O₂. Express your answer using two significant figures. 1951 ΑΣΦ → Request Answer ?
Chapter13: Titrations In Analytical Chemistry
Section: Chapter Questions
Problem 13.13QAP
Related questions
Question
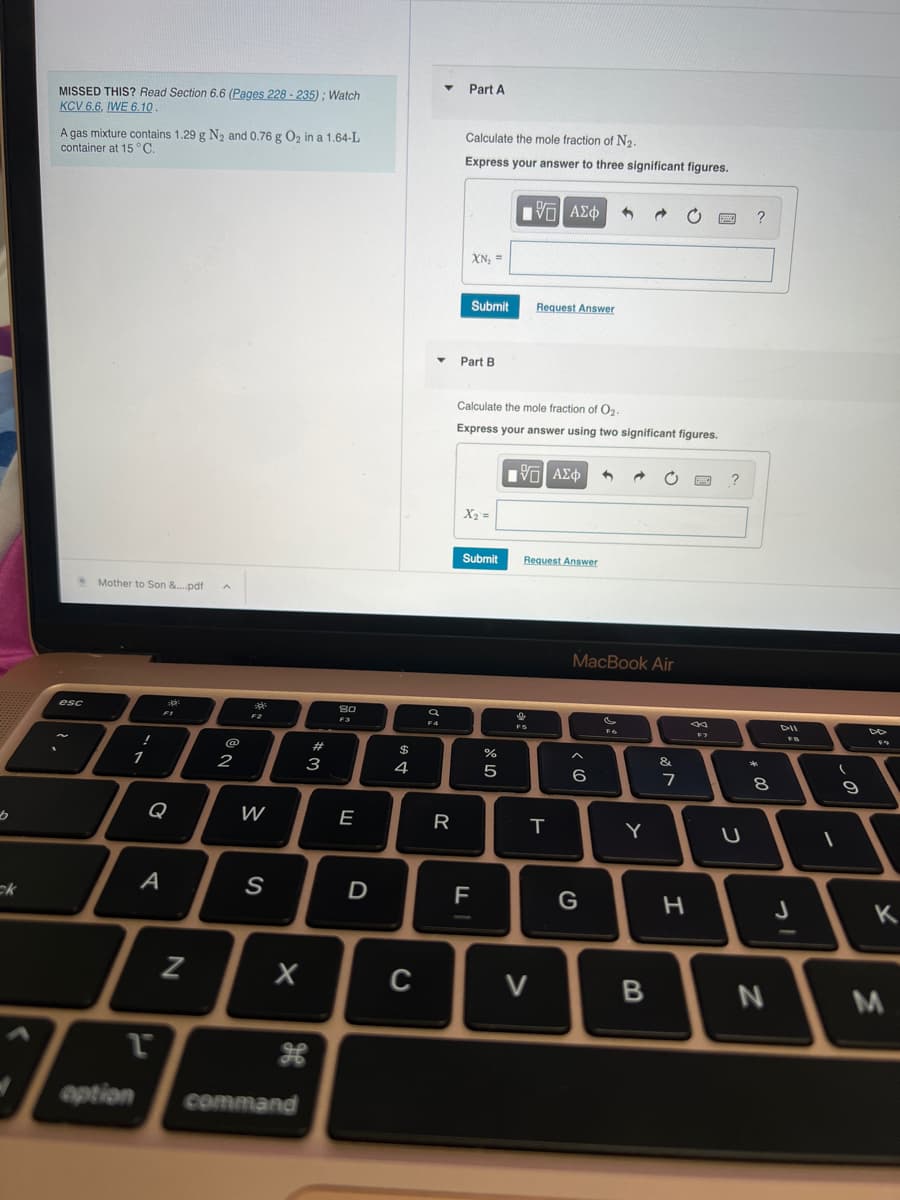
Transcribed Image Text:[b
ck
MISSED THIS? Read Section 6.6 (Pages 228-235); Watch
KCV 6.6, IWE 6.10
A gas mixture contains 1.29 g N₂ and 0.76 g O₂ in a 1.64-L
container at 15 °C.
esc
Mother to Son &....pdf
!
option
1
FI
G
1
A
1
N
@
2
300
W
S
X
command
#
3
8.0
F3
E
D
$
4
4
C
a
F4
R
Part A
Calculate the mole fraction of N₂.
Express your answer to three significant figures.
XN₂ =
Submit
Part B
X₂ =
Submit
Calculate the mole fraction of O₂.
Express your answer using two significant figures.
F
דן
olo 50
%
G| ΑΣΦ 1
Request Answer
IVE| ΑΣΦ 4
9
Request Answer
V
T
MacBook Air
< 10
^
6
G
FO
Y
B
&
7
H
PESCA
F7
?
U
?
00
8
N
DII
J
-
(
9
K
M
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

