Molecular Shape Number of Unshared Electron Pairs Around the Central Number of Bond Groups Around Central Atom Atom Linear Bent Trigonal Planar Trigonal Pyramidal Tetrahedral 2. What is the electrostatic charge of all ele ctron groups, either shared (Le. bonds) or unshared? 3. Since all electron groups have this same charge, do they attract or repel each other? 4. How many unshared electron pairs are present in the carbon atom in CO,? 5. Considering your answers to the previous three questions, explain why Co, is not bent? 6. Explain why H,O is not linear.
Molecular Shape Number of Unshared Electron Pairs Around the Central Number of Bond Groups Around Central Atom Atom Linear Bent Trigonal Planar Trigonal Pyramidal Tetrahedral 2. What is the electrostatic charge of all ele ctron groups, either shared (Le. bonds) or unshared? 3. Since all electron groups have this same charge, do they attract or repel each other? 4. How many unshared electron pairs are present in the carbon atom in CO,? 5. Considering your answers to the previous three questions, explain why Co, is not bent? 6. Explain why H,O is not linear.
Chapter9: Covalent Bonding: Orbitals
Section: Chapter Questions
Problem 57E: Use Figs. 4-54 and 4-55 to answer the following questions. a. Would the bonding molecular orbital in...
Related questions
Question
Answer question 2,3,4,5 and 6
Transcribed Image Text:-- - -- -
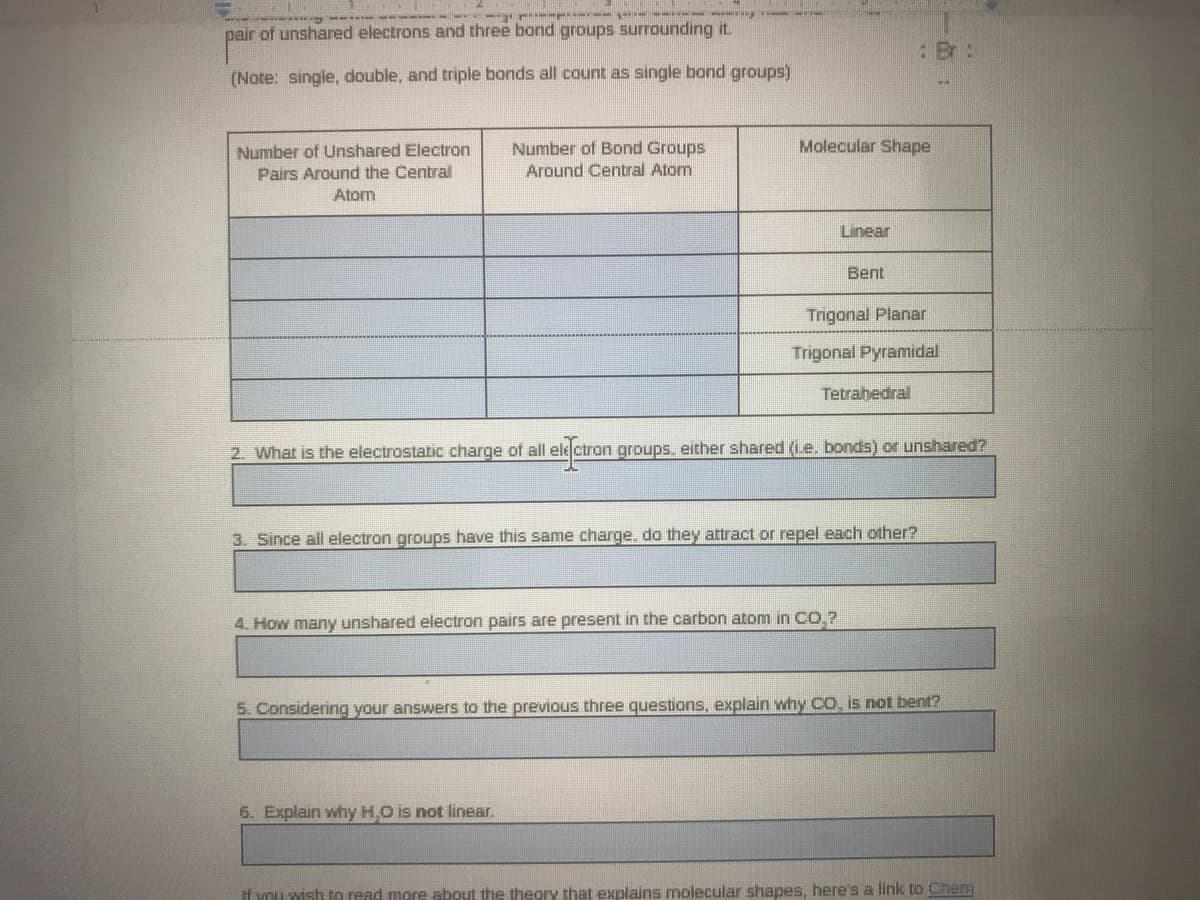
pair of unshared electrons and three bond groups surrounding it.
: Br:
(Note: single, double, and triple bonds all count as single bond groups)
Number of Bond Groups
Molecular Shape
Number of Unshared Electron
Pairs Around the Central
Around Central Atom
Atom
Linear
Bent
Trigonal Planar
Trigonal Pyramidal
Tetrahedral
2. What is the electrostatic charge of all ele ctron groups. either shared (Le. bonds) or unshared?
3. Since all electron groups have this same charge, do they attract or repel each other?
4. How many unshared electron pairs are present in the carbon atom in CO,?
5. Considering your answers to the previous three questions, explain why co, is not bent?
6. Explain why H.O is not linear.
If you wish to read more about the thegry that explains molecular shapes, here's a link to Che
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning