Chapter23: Carbonyl Condensation Reactions
Section23.SE: Something Extra
Problem 35MP: Isoleucine, another of the twenty amino acids found in proteins, is metabolized by a pathway that...
Related questions
Question
Provide a detailed mechanism for the complete reaction
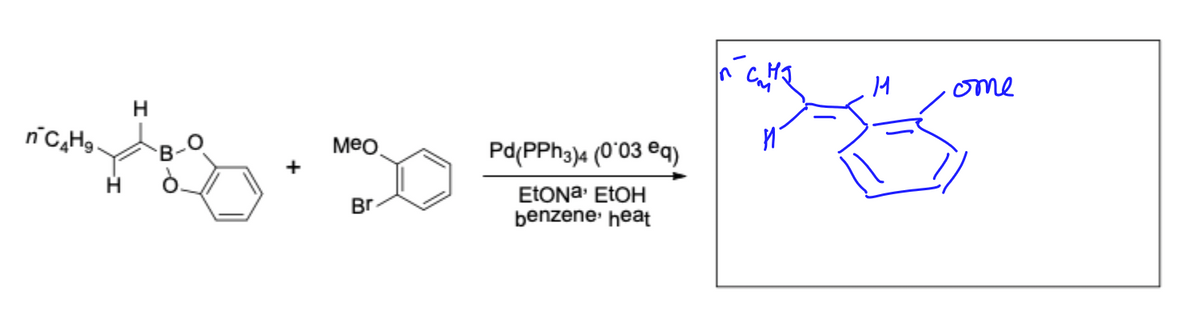
Transcribed Image Text:n C4H9.
H
B-
Meo
Br
Pd(PPh3)4 (0°03 eq)
Etona, EtOH
benzene, heat
H
ome
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning