Part A Classify each orbital diagram for ground-state electron configurations by the rule or principle it violates. Drag the appropriate items to their respective bins. • View Available Hint(s) Reset Help 4p 1 2p 11 11 2p 111 34 1 4s 1 4p 1L 34 1 1 1 1 1 ad 1 1 1 1 1 | 111| 2s 11 2s 4s 1 Aufbau violation Hund violation Pauli violation
Part A Classify each orbital diagram for ground-state electron configurations by the rule or principle it violates. Drag the appropriate items to their respective bins. • View Available Hint(s) Reset Help 4p 1 2p 11 11 2p 111 34 1 4s 1 4p 1L 34 1 1 1 1 1 ad 1 1 1 1 1 | 111| 2s 11 2s 4s 1 Aufbau violation Hund violation Pauli violation
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter11: Quantum Mechanics: Model Systems And The Hydrogen Atom
Section: Chapter Questions
Problem 11.69E: Construct an energy level diagram showing all orbitals for the hydrogen atom up to n=5, labeling...
Related questions
Question
7ab ASAP
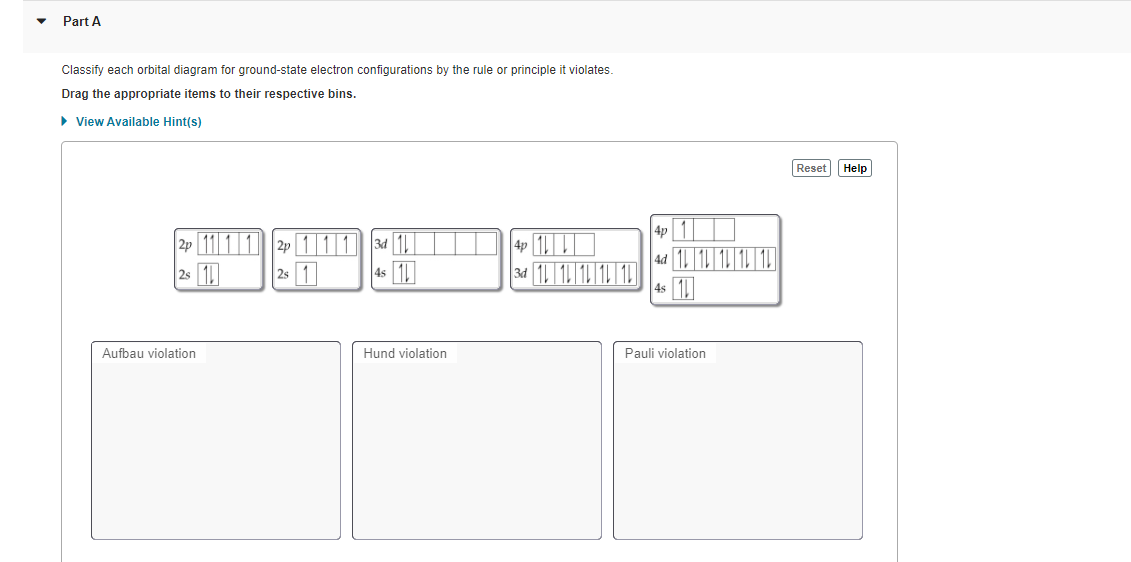
Transcribed Image Text:Part A
Classify each orbital diagram for ground-state electron configurations by the rule or principle it violates.
Drag the appropriate items to their respective bins.
> View Available Hint(s)
Reset Help
4p 1|
2p 11 11 2p 111 34 1
25 1
ad 1 1 1 1 1
25 1
4s 1
34 1 1 1 1 1
4s
Aufbau violation
Hund violation
Pauli violation
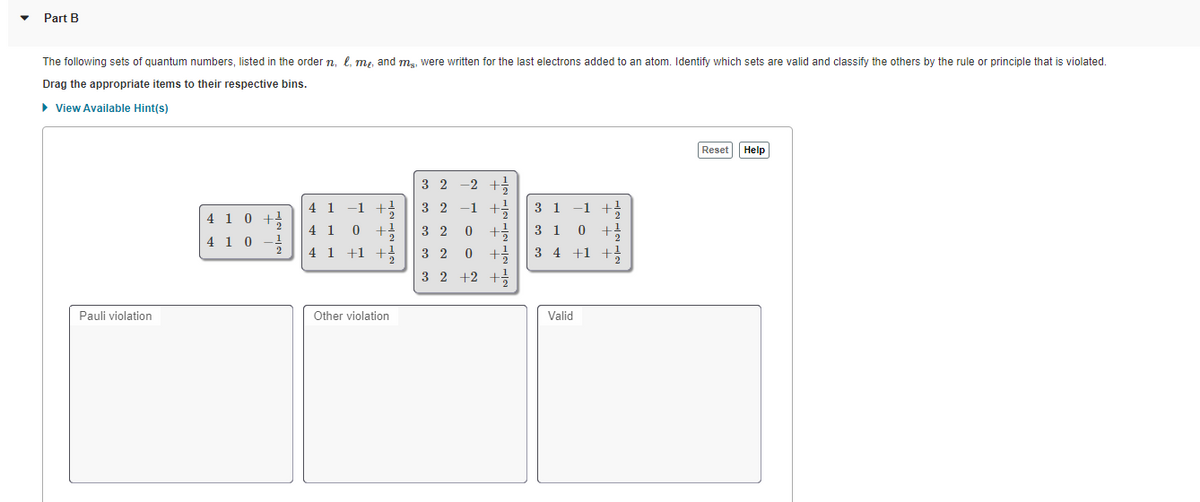
Transcribed Image Text:Part B
The following sets of quantum numbers, listed in the order n, l, m,, and mg, were written for the last electrons added to an atom. Identify which sets are valid and classify the others by the rule or principle that is violated.
Drag the appropriate items to their respective bins.
> View Available Hint(s)
Reset Help
3 2
-2 +
4 1
-1
3 2
-1
3 1
-1
+1
4 1 0
4 1
3 2
3 1
4 1 0
2
4 1 +1 +
3 2
3 4 +1 +!
3 2
+2
Pauli violation
Other violation
Valid
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning