Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter5: Resonance
Section: Chapter Questions
Problem 10E: Phenol (shown below) has a pKa10 . a. Based on pKa data, is phenol a stronger or weaker acid than an...
Related questions
Question
100%
Name each acids
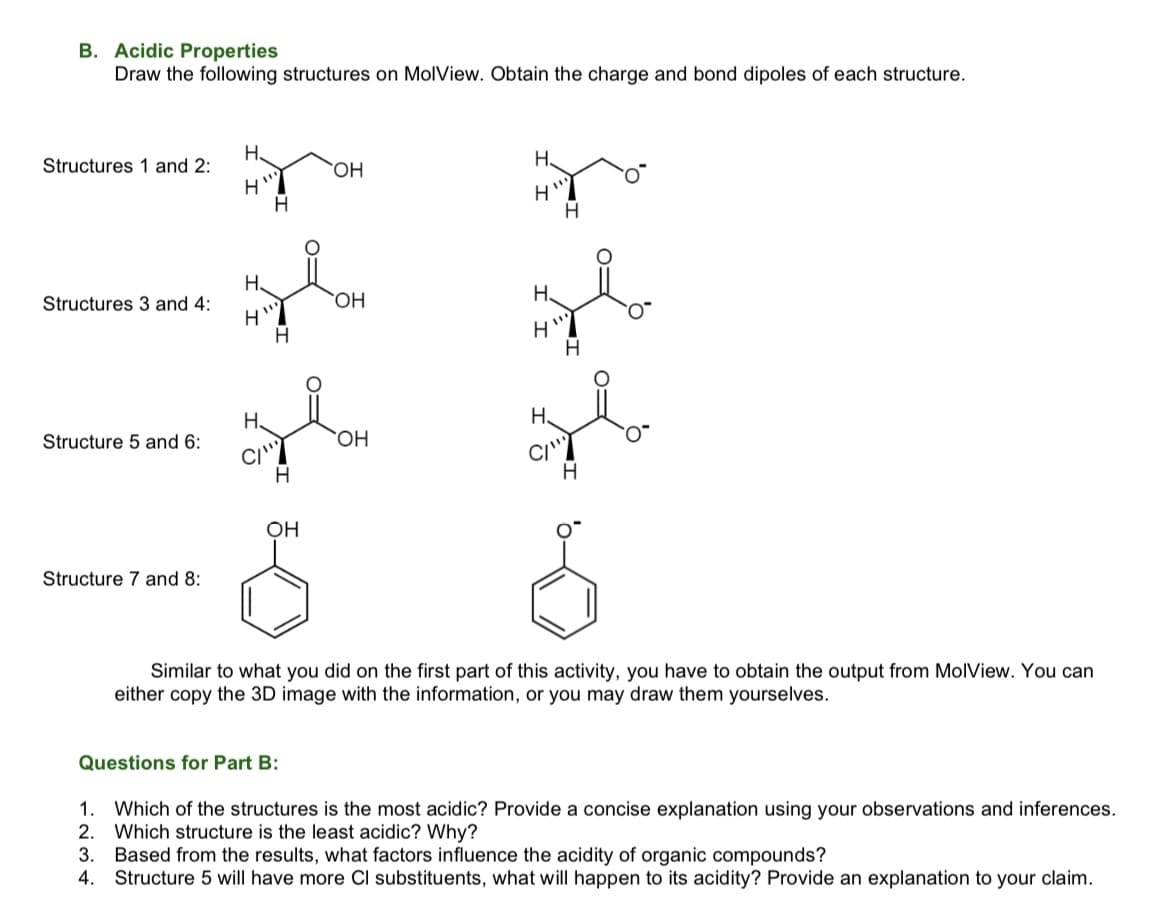
Transcribed Image Text:B. Acidic Properties
Draw the following structures on MolView. Obtain the charge and bond dipoles of each structure.
H.
H.
Structures 1 and 2:
HO.
H.
H.
Structures 3 and 4:
H.
H.
H.
Structure 5 and 6:
CI'
OH
Structure 7 and 8:
Similar to what you did on the first part of this activity, you have to obtain the output from MolView. You can
either copy the 3D image with the information, or you may draw them yourselves.
Questions for Part B:
1.
Which of the structures is the most acidic? Provide a concise explanation using your observations and inferences.
2.
Which structure is the least acidic? Why?
3.
Based from the results, what factors influence the acidity of organic compounds?
4. Structure 5 will have more Cl substituents, what will happen to its acidity? Provide an explanation to your claim.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT