Name Formula lonic/Covalent Soluble/Insoluble Electrolyte/Precipitate Ammonium Sulfide (NH4)2S Phosphorus Pentachloride PCI5 Silver Bromide AgBr Barium Sulfide BaS Strontium Sulfate SrSO4 Lithium Chlorite LICIO2 Iron (II) Sulfate FESO4 Sodium Na2Cr207 Dichromate Silver Acetate AGC2H3O2 Dinitrogen Pentahydride N2HS
Name Formula lonic/Covalent Soluble/Insoluble Electrolyte/Precipitate Ammonium Sulfide (NH4)2S Phosphorus Pentachloride PCI5 Silver Bromide AgBr Barium Sulfide BaS Strontium Sulfate SrSO4 Lithium Chlorite LICIO2 Iron (II) Sulfate FESO4 Sodium Na2Cr207 Dichromate Silver Acetate AGC2H3O2 Dinitrogen Pentahydride N2HS
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 80AP
Related questions
Question
100%
Help plz thankyou
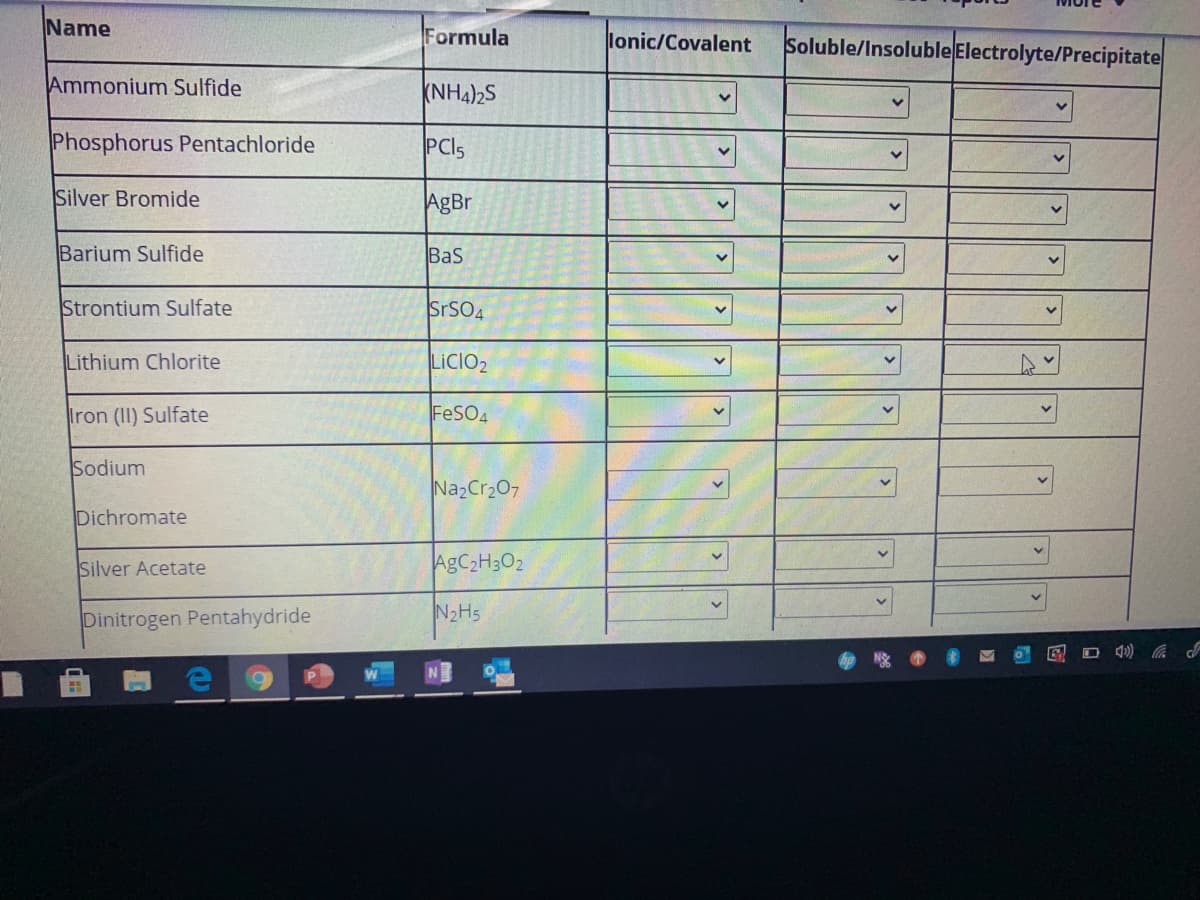
Transcribed Image Text:Name
Formula
lonic/Covalent
Soluble/Insoluble Electrolyte/Precipitate
Ammonium Sulfide
(NH4)2S
Phosphorus Pentachloride
PCI5
Silver Bromide
AgBr
Barium Sulfide
Bas
Strontium Sulfate
SrSO4
Lithium Chlorite
LICIO2
Iron (II) Sulfate
FeSO4
Sodium
NazCr207
Dichromate
Silver Acetate
AGC2H3O2
Dinitrogen Pentahydride
N2H5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning