Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.20QAP
Related questions
Question
Observe the diagram and describe the
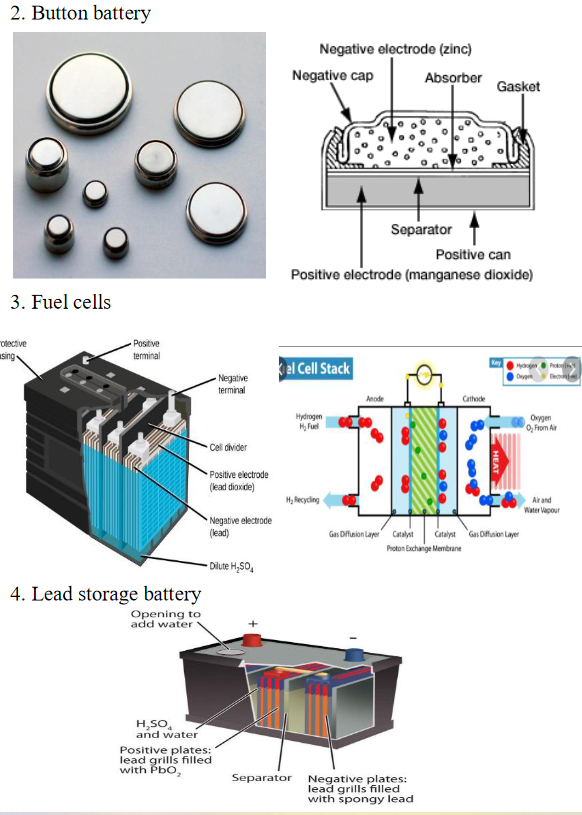
Transcribed Image Text:2. Button battery
Negative electrode (zinc)
Negative cap
Absorber
Gasket
Separator
Positive can
Positive electrode (manganese dioxide)
3. Fuel cells
otective
sing
Positive
terminal
3el Cell Stack
Key
Proto
et
Negative
terminal
Orygn
Anode
Cathode
Hydrogen
H, Fuel
Orygen
0, From Air
Cel divider
Positive electrode
(kead dioxde)
H, lecycing
Ar and
Water Vapour
Negative electrode
(lead)
Gas Difinion Layer Catalst
Catalynt Gas Dfusion Layer
Poton Exchange Membrane
- Dlute H,S0.
4. Lead storage battery
Opening to
add water
H,SO,
and water
Positive plates:
lead grills filled
with Pbo,
Separator
Negative plates:
lead grills filled
with spongy lead
HEAT
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning