Newman as drawn: A HO. conformation: back carbon front carbon rotate back carbon 60 degrees counterclockwise Newman after rotation: C rotate front Newman after rotation: B carbon 180 degrees conformation: conformation: rotate back carbon 120 degrees clockwise Newman after rotation: D Wedge-dash redraw as wedge-dash conformation: Most stable : (least)
Newman as drawn: A HO. conformation: back carbon front carbon rotate back carbon 60 degrees counterclockwise Newman after rotation: C rotate front Newman after rotation: B carbon 180 degrees conformation: conformation: rotate back carbon 120 degrees clockwise Newman after rotation: D Wedge-dash redraw as wedge-dash conformation: Most stable : (least)
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter7: Cycloalkanes
Section: Chapter Questions
Problem 17E: Build a model of methylcyclohexane, and use the model to complete the following Newmanprojections of...
Related questions
Question
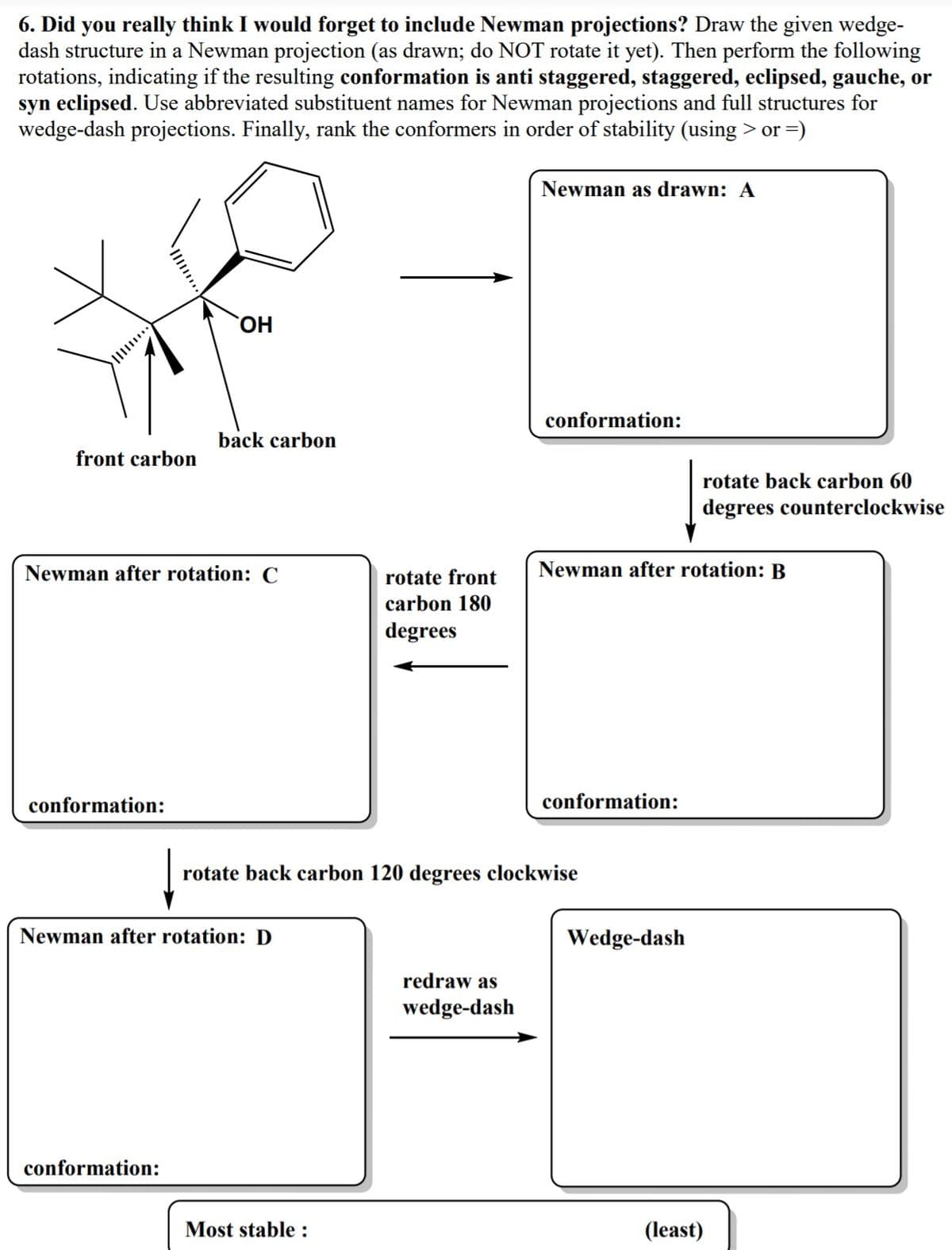
Transcribed Image Text:6. Did you really think I would forget to include Newman projections? Draw the given wedge-
dash structure in a Newman projection (as drawn; do NOT rotate it yet). Then perform the following
rotations, indicating if the resulting conformation is anti staggered, staggered, eclipsed, gauche, or
syn eclipsed. Use abbreviated substituent names for Newman projections and full structures for
wedge-dash projections. Finally, rank the conformers in order of stability (using > or =)
Newman as drawn: A
conformation:
back carbon
front carbon
rotate back carbon 60
degrees counterclockwise
Newman after rotation: C
rotate front
Newman after rotation: B
carbon 180
degrees
conformation:
conformation:
rotate back carbon 120 degrees clockwise
Newman after rotation: D
Wedge-dash
redraw as
wedge-dash
conformation:
Most stable:
(least)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning